Advertisements
Chapters
1: Solid State
2: Solutions
3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
▶ 9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 9 - Coordination Compounds Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 9 - Coordination Compounds - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 9: Coordination Compounds
Below listed, you can find solutions for Chapter 9 of CISCE Nootan for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds Review Exercises [Pages 517 - 541]
What are coordination compounds and how do they differ from double salts? Explain with examples.
Explain the following term with respect to a coordination compound:
Ligands
Explain the following term with respect to a coordination compound:
Coordination sphere
Explain the following term with respect to a coordination compound:
Coordination number
What is the oxidation state of Ni in Ni(CO)4?
Calculate the oxidation number of the central metal atom (underlined) in the following complex species:
K4[Fe(CN)6]
Calculate the oxidation number of the central metal atom (underlined) in the following complex species:
[Fe(H2O)6]Cl3
Calculate the oxidation number of the central metal atom (underlined) in the following complex species:
[Co(NH3)6]3+
Calculate the oxidation number of the central metal atom (underlined) in the following complex specie:
[Fe(C2O4)3]3−
In the complex ion [Co(NH3)3(H2O)2Cl]+:
Identify the ligands’ formulae and charge on each of them.
In the complex ion [Co(NH3)3(H2O)2Cl]+:
Write the geometry of the complex ion.
Define coordination number.
Find the coordination number of the central metal atom in [Co(NH3)5Cl]Cl2.
Find the coordination number of the central metal atom in K2[FeCl4].
What is the oxidation state of Co in the complex, [Co(NH3)2(NO2)Cl][Au(CN)2]?
What is the oxidation state of platinum in the complex, [Pt(NH3)2Cl2]Cl2?
What is the oxidation state of cobalt in the complex, [Co(NH3)4(H2O)Br](NO3)2?
What is the coordination number of Fe in the complex, K3[Fe(C2O4)3]?
What is the coordination number of cobalt in [Co(en)(H2O)(Br)(Cl2)]?
What is meant by a hexadentate ligand?
Give an example of hexadentate ligand.
What do you understand by chelating ligands?
What are chelates?
Give one example of a chelating ligand.
Give one example of a chelate.
Give one example of a neutral ligand.
Give one example of an anionic ligand.
Give one example of a cationic complex ion.
Give one example of an anionic complex.
Calculate the charge number of the complex ion:
[Co(NH3)4Cl2]+
Calculate the charge number of the complex ion:
[Pt(NH3)2Cl2]
What is the coordination number of central metal ion in [Fe(C2O4)3]3−?
Give an example of chelate complexes.
Why is ammonia molecule a good ligand?
Name a ligand which is bidentate and give an example of the complex formed by this ligand.
Name the following complex ion in the IUPAC system:
[Co(CN)6]3−
Name the following complex ion in the IUPAC system:
[HgI4]2−
Name the following complex ion in the IUPAC system:
[Fe(H2O)6]3+
Name the following complex ion in the IUPAC system:
[Cu(NH3)4]2+
Name the following complex ion in the IUPAC system:
[CoCl(en)2NH3]2+
Name the following complex ion in the IUPAC system:
[CoCl2(NH3)4]+
Name the following complex ion in the IUPAC system:
[Co(ONO)(NH3)5]2+
Give the IUPAC name of the following coordination compound:
K3[Al(C2O4)3]
Give the IUPAC name of the following coordination compound:
[CoCl(NH3)5]Cl2
Give the IUPAC name of the following coordination compound:
K3[Fe(CN)6]
Give the IUPAC name of the following coordination compound:
Na3[Co(NO2)6]
Give the IUPAC name of the following coordination compound:
[Cr(en)3]Cl3
Give the IUPAC name of the following coordination compound:
[PtCl3(NH3)3]Cl
Give the IUPAC name of the following coordination compound:
[Co(en)3][Cr(CN)6]
Give the IUPAC name of the following coordination compound:
Na2[Cr(EDTA)]
Give the IUPAC name of the following coordination compound:
[Co(NO2)(NH3)5]Cl2
Give the IUPAC name of the following coordination compound:
NH4[Cr(NH3)2(NCS)4]
Give the IUPAC name of the following coordination compound:
[Cu(H2O)2(NH3)4]SO4
Give the IUPAC name of the following coordination compound:
[Pt(py)4][PtCl4]
Give the IUPAC name of the following coordination compound:
[Co(ONO)(NH3)5]Cl2
Give the IUPAC name of the following coordination compound:
Na3[AIF6]
Give the chemical formula for the following compound:
copper (II) hexacyanidoferrate (II)
Give the chemical formula for the following compound:
potassium hexacyanidocobaltate (III)
Give the chemical formula for the following compound:
hexaammineplatinum (IV) chloride
Give the chemical formula for the following compound:
potassium hexacyanidoferrate (III)
Give the chemical formula for the following compound:
pentaamminenitrocobalt (III) chloride
Give the chemical formula for the following compound:
tetrachloridocuprate (II) ion
Give the chemical formula for the following compound:
chloridobis (ethylenediamine) nitrocobalt (III) ion
Give the chemical formula for the following compound:
hexaaquamanganese (II) ion
Give the chemical formula for the following compound:
potassium hexachloridoplatinate (IV)
Give the chemical formula for the following compound:
triamminebromidonitrocobalt (II)
Write the name of the following complex using IUPAC norms:
[Cu(NH3)4]SO4
Write the name of the following complex using IUPAC norms:
[Pt(NH3)2Cl2]
Write the name of the following complex using IUPAC norms:
[Ag(NH3)2Cl]
Give the IUPAC name of the following coordination compound:
Na3[AIF6]
Write the IUPAC names of the complexes [CoBr(NH3)5]SO4 and [Co(SO4)(NH3)5Br] and give a simple test to distinguish them.
Write the chemical formula of the following complex:
Hexaammineplatinum (IV) chloride
Write the chemical formula of the following complex:
Tetraamminedichloridocobalt (III) chloride
Write IUPAC name of the complex Na3[Cr(OH)2F4].
Name the following complex using IUPAC norms:
[Co(en)2(ONO)Cl]Cl
A coordination compound has the formula CoCl3.4NH3. It does not liberate ammonia but forms a precipitate with AgNO3. Write the structure and IUPAC name of the complex compound.
Give the IUPAC name of [PtCl(NH2CH3)(NH3)2Cl.
Write the formula of the following complex:
Hexaammineplatinum (IV) chloride
Write the formula of the following complex:
Dichloridotetraammine cobalt (III) ion
Illustrate:
Linkage isomerism in coordination compounds.
Illustrate:
Coordination isomerism in coordination compounds.
Explain with a suitable example geometrical isomerism in coordination compounds.
Illustrate coordination isomerism in coordination compounds giving one example.
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
Write the structures of a pair of complexes showing geometrical isomerism.
Give the IUPAC name of the linkage isomer of [(NH3)3PtNO2]Cl.
Give the IUPAC name of the linkage isomer of [(NH3)2(H2O)PtNO2]Br.
Give the IUPAC name of the linkage isomer of [(NH3)3PtNO2]Br.
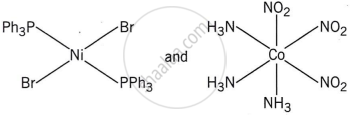
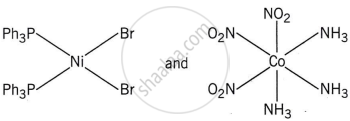
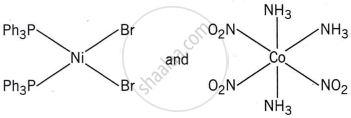
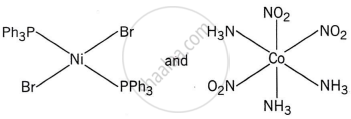
Write the name of the coordination isomer of the complex [Co(en)3][Cr(CN)6].
Write the IUPAC name of the linkage isomer of [Co(NH3)5NO2]Cl2.
Write the formula for the linkage isomer of [Cr(H2O)5SCN]2+.
How many isomers are there for the complex [Co(NH3)4Cl2]Cl?
A coordination compound has the formula CoCl3.4NH3. It does not liberate ammonia but forms a precipitate with AgNO3. Write the structure and IUPAC name of the complex compound.
Describe linkage isomerism with an example.
Draw the geometrical isomers of [Pt(NH3)4Cl2]2+.
What type of coordination compounds show optical isomerism?
Give two examples of types of complexes that exhibit optical isomerism.
What type of isomerism is exhibited by [CoCl2(NH3)4]+?
Write the structures of possible isomers [CoCl2(NH3)4]+.
Give the IUPAC name of the isomer of [CoCl2(NH3)4]+.
Name the type of isomerism exhibited by the following pair of compounds:
[Co(NH3)6][Cr(CN)6] and [Co(CN)6][Cr(NH3)6]
Name the type of isomerism exhibited by the following pair of compounds:
[Cr(H2O)6]Cl3 and Cr(H2O)5Cl]Cl2.H2O
Name the type of isomerism exhibited by the following pair of compounds:
[Co(NH3)5(ONO)]Cl2 and [Co(NH3)5(NO2)]Cl2
Write the structure of the geometrical isomer of the following complex:
[Pd(NO2)2(NH3)2]
Write the structure of the geometrical isomer of the following complex:
[Pt(gly)2]
Write the IUPAC name for any of the isomers with the molecular formula [Pt(NH3)2Cl2]Cl2.
Name the ionisation isomer of [Cr(H2O)3Br]SO4.
Write the IUPAC name for any one of the isomers of [Cr(NH3)4Cl2]+.
Give the IUPAC name of the ionisation isomer of [Cr(NH3)4Cl2]+.
Write the IUPAC name of [Co(en)2Cl2]+ ion.
Describe briefly the isomerism shown by [Cr(H2O)5(NCS)]2+.
Write the IUPAC name of [Co(en)2Cl2]Cl.
Draw the structures of all the isomers with this formula for the [Co(en)2Cl2]Cl complex.
Using valence bond theory of complexes explain the geometry and diamagnetic nature of ion [Co(NH3)6]3+.
Describe the hybridisation scheme, the resultant geometry and the magnetic behaviour of [Co(NH3)6]3+.
How would you account for the following?
[Ti(H2O)6]3+ is coloured, while [Sc(H2O)6]3+ is colourless.
How would you account for the following?
[Fe(CN)6]3− is weakly paramagnetic, while [Fe(CN)6]4− is diamagnetic.
How would you account for the following?
[Ni(CO)4] possesses tetrahedral geometry, while [Ni(CN)4]2− is square planar.
Using valence bond approach, predict the shape and magnetism (i.e., paramagnetic or diamagnetic) of [Co(CN)4]−.
Using valence bond approach, predict the shape and magnetism (paramagnetic or diamagnetic) of [Ni(CN)4]−.
Following valence bond scheme, explain the bonding in [Cr(H2O)6]3+.
What do you understand by the term stability constant, K of a complex?
Knowing that the value of K for [Cu(NH3)4]2+ is 4.5 × 1011 and for [Cu(CN)4]2− is 2.0 × 1027, suggest which complex species will furnish less Cu2+ ions in solution.
Knowing that the value of K for [Cu(NH3)4]2+ is 4.5 × 1011 and for [Cu(CN)4]2− is 2.0 × 1027, suggest which out of NH3 and CN− is a stronger ligand.
Select a complex formation reaction and write an expression for the stability constant of the complex species. What information is conveyed regarding the strengths of ligands from the stability values of their complexes with a metal ion? Illustrate your answer with examples of monodentate ligands.
Using the valence bond approach, explain the shape and magnetic behaviour of [Ni(NH3)6]2+.
On passing H2S through a solution containing Cu2+ and Cd2+ ion and excess of KCN solution, only Cd gets precipitated. Explain.
Draw the structure and write the hybridisation state of the central atom of the following specie:
[Co(NH3)6]3+
Draw the structure and write the hybridisation state of the central atom of the following specie:
[NiCl4]2−
Describe linkage isomerism with an example.
Describe the magnetic behaviour of [Ni(CO)4].
Explain the square planar shape of the tetracyanidonickelate (II) ion.
Account for the magnetic property of the tetracyanidonickelate (II) ion.
Using the valence bond approach, predict the shape and magnetic character of [Ni(CO)4].
Deduce the shape and magnetic behaviour of the complex ion [Co(NH3)5NO2]2+.
Explain the following observation:
Tetrahedral Ni(II) complexes are usually paramagnetic but square planar Ni(II) complexes are diamagnetic.
What magnetic behaviours are expected for [Ni(CO)4] and [NiCl4]2− and why?
Giving an example, describe how the formation of coordination compounds is useful in analytical chemistry.
Using the valence bond approach, deduce the shape and magnetic character of [Cr(CO)6].
Using the valence bond approach, predict the shape and magnetic character of [Fe(CN)6]3− ion.
Write the neutral molecule in which the central atom is sp3d2 hybridised.
Using valence bond theory, deduce the geometry and magnetic nature of [Ni(CN)4]2−.
Draw a figure to show splitting of degenerate d-orbitals in an octahedral crystal field. How does the magnitude of Δo decide the actual configuration of d-orbitals in an complex entity?
Compare the magnetic behaviour of the complex entities [Fe(CN)6]4− and [FeF6]3−.
The values of dissociation constant of [Cu(NH3)4]2+ and [Co(NH3)6]3+ are 1.0 × 10−12 and 6.2 × 10−34 respectively. Which complex would be more stable and why?
Using the valence bond theory of complexes, explain the geometry and magnetic nature of [Ni(NH3)6]2+.
Name two properties of the central metal ion which enable it to form stable complex entities.
The formation of complex compounds, finds application in the extraction of some metals. Furnish one example to support the above statement.
Draw a sketch to show the splitting of d-orbitals in an octahedral crystal field.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds VERY SHORT ANSWER TYPE QUESTIONS [Pages 544 - 545]
Sort out the complex salts and double salts among the following:
Pot. dicyanidoargentate (I), Mohr’s salt, Potash alum, Pot. ferrocyanide.
Does a solution of K4[Fe(CN)6] contain Fe2+ ions?
Name the ions present in the aqueous solution of K2SO4.Al2(SO4)3.24H2O.
What is a ligand?
Give two examples of ligands.
Sort out the cationic, anionic and neutral complexes among the following:
K2[HgI4], [Co(NH3)6]Cl3, K3[Fe(CN)6], [Ni(CO)4], [Pt(NH3)2Cl2], [Fe(H2O)6]Cl3.
Name the ligand/ligands present in [Pt(NH3)2Cl2]Br2.
What is the oxidation number of Co in [Co(NH3)5NO2]Cl2?
Give one example of a neutral ligand.
Give one example of an anion ligand.
Give one example of a cationic ligand.
Give two examples of monodentate ligands.
Give two examples of bidentate ligands.
Give the name of the OH− ligand.
Give the name of the H2O ligand.
Give the name of the NH3 ligand.
Give the name of the NH2CH2-CH2NH2 ligand.
Give the name of the CN− ligand.
Give the name of the NCS− ligand.
Give one example of a chelating ligand.
Write the structures of any two chelating ligands.
What is the number of coordination sites in oxalato?
What is the number of coordination sites in ammine?
What is the number of coordination sites in ethylenediamine?
What is the number of coordination sites in EDTA?
What is the number of ionisable chlorine atoms in the complex CoCl3.4NH3?
How many ions would be furnished by the complex CoCl3.6NH3 in aqueous solution?
Calculate the charge number of the complex ion furnished by the complex K2[HgI4].
Write the IUPAC name of the [Fe(H2O)6]Cl3 complex.
Write the IUPAC name of the [CoCl2(en)2] complex.
Write the IUPAC name of the complex [CoCl(NO2)(NH3)4]ClO4.
Write the structure of the ammonium diamminetetra (thiocyanato-N) chromate (III) complex.
Write the structure of the dichloridobis (ethylenediamine) cobalt (IV) sulphate complex.
What type of isomerism is exhibited by the following pair of isomers?
[Pt(OH)2(NH3)4]SO4 and [Pt(SO4)(NH3)4](OH)2
What type of isomerism is exhibited by the following pair of isomers?
[Cr(SCN)(H2O)5]2+ and [Cr(NCS)(H2O)5]2+
What type of isomerism is exhibited by the following pair of isomers?
[Cu(NH3)4][PtCl4] and [Pt(NH3)4[CuCl4]
Write the geometrical isomers of the complex [PtBrCl(NH3)2].
Write the cis isomers of the complex [CoCl2(en)(NH3)4]+.
Write the trans isomer of the complex [CoCl2(en)(NH3)4]+.
Write the geometrical isomers of [CoCl2(en)(NH3)2]+.
Do square planar complexes exhibit optical isomerism?
Write the optical isomers of [Cr(C2O4)3]3−.
Write the optically active isomers of [Co(en)2Cl2]+.
Why is the trans-isomer of [Co(en)2Cl2]+ not optically active?
Name the hybridisation state of the central metal ion in the [Co(NH3)6]Cl3 complex.
Name the hybridisation state of the central metal ion in the [Pt(NH3)2Cl2] complex.
State whether a complex formed by sp3d2 hybridisation is a low spin or a high spin complex.
The complex [Cr(NH3)6]3+ involves d2sp3 hybridisation of the central metal ion. State whether it is an inner orbital or an outer orbital complex.
How is the stability constant K of a complex related to its thermodynamic stability?
Among NH3 and CN−, which is a stronger ligand?
The ionic radii of Cu2+, Ni2+ and Fe2+ ions are 69, 78 and 83 pm respectively. Arrange these ions in the decreasing order of the stability of the complexes formed by them.
Name the reagent generally used to estimate the hardness of water.
Name a complex used in the treatment of cancer.
Name the main metal present in the following:
Haemoglobin
Name the metals present in chlorophyll.
Name the metals present in vitamin-B12.
Complete the following statement for the coordination entity (complex ion) [CrCl2(OX)2]3−.
OX is abbreviation for ______.
Complete the following statement for the coordination entity (complex ion) [CrCl2(OX)2]3−.
The oxidation number of chromium is ______.
Complete the following statement for the coordination entity (complex ion) [CrCl2(OX)2]3−.
The coordination number of chromium is ______.
Complete the following statement for the coordination entity (complex ion) [CrCl2(OX)2]3−.
______ is a bidentate ligand.
Specify the oxidation number of the metal in the following coordination entity:
[Co(H2O)(CN)(en)2]2+
Specify the oxidation number of the metal in the following coordination entity:
[PtCl4]2−
Specify the oxidation number of the metal in the following coordination entity:
[Cr(NH3)3Cl3]
Specify the oxidation number of the metal in the following coordination entity:
[CoBr2(en)2]+
Specify the oxidation number of the metal in the following coordination entity:
K3[Fe(CN)6]
Specify which out of the following complex structures exhibit geometrical isomerism:
Linear
Square planar
Tetrahedral
Octahedral
How many geometrical isomers are possible in the following coordination entity?
[Cr(OX)3]3−
How many geometrical isomers are possible in the following coordination entity?
[Co(NH3)3Cl3]
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds SHORT ANSWER TYPE QUESTIONS [Pages 545 - 547]
What are coordination compounds? Explain with suitable examples.
Give the ionisation behaviour of the following salt:
Potash alum
Give the ionisation behaviour of the following salt:
Potassium argentocyanide
Give the ionisation behaviour of the following salt:
Mohr’s salt
Define the following term in relation to coordination compounds:
Ligands
Define the following term in relation to coordination compounds:
Complex ion
Define the following term in relation to coordination compounds:
Coordination sphere
Define the following term in relation to coordination compounds:
Coordination number
What is a ligand?
How are ligands classified on the basis of charge? Give suitable examples.
How are ligands classified on the basis of the number of donor atoms? Give an example of each type.
What is meant by monodentate ligands? Illustrate with examples.
What is meant by bidentate ligands? Illustrate with examples.
What do you understand by chelating ligands?
What are chelates?
Write the structures of any two chelating ligands.
Illustrate chelates with a suitable example.
Write four postulates of Werner’s theory.
Write the structure of the following complex on the basis of Werner’s theory:
CoCl3·6NH3
Write the structure of the following complex on the basis of Werner’s theory:
CoCl3·5NH3
Write the structure of the following complex on the basis of Werner’s theory:
CoCl3·3NH3
Represent the following complex according to the IUPAC convention:
CoCl3·5NH3
Represent the following complex according to the IUPAC convention:
CoCl3·4NH3
Represent the following complex according to the IUPAC convention:
CoCl3·6NH3
Represent the following complex according to the IUPAC convention:
PtPyNH3ClBr
Calculate the oxidation number of Au in H[AuCl4].
Calculate the oxidation number of Ag in K[Ag(CN)2].
Specify the oxidation number of the metal in the following coordination entity:
K3[Fe(CN)6]
Calculate the oxidation number of Co in [Co(NH3)3(H2O)2Cl]+.
Give the name of the \[\ce{NH^{-}_2}\] ligand.
Give the name of the ONO− ligand.
Give the name of the [CH3]3P ligand.
Give the name of the \[\ce{NO^{+}_2}\] ligand.
Give the name of the NH2CH2CH2NH2 ligand.
Give the IUPAC name of the following complex:
[Cu(NH3)4]SO4
Give the IUPAC name of the following complex:
[CoCl2(en)(NH3)2]+
Give the IUPAC name of the following complex:
[Co(en)2Cl2]
Give the IUPAC name of the following complex:
K3[Al(C2O4)3]
Give the IUPAC name of the following complex:
[Co(NH3)6][CdCl5]
Write the formulae of the following complex:
potassium tetraiodidomercurate (II)
Write the formulae of the following complex:
hexaaquarion (II) sulphate
Write the formulae of the following complex:
sodium ethylenediaminetetraacetatonickelate (III)
Using IUPAC norms write the formulate for the following coordination compound:
Hexaamminecobalt(III) chloride
Explain and illustrate with an example of the following in relation to coordination compounds:
Ionisation isomerism
Explain and illustrate with an example of the following in relation to coordination compounds:
Hydrate isomerism
Explain the term ‘linkage isomerism’ with respect to coordination compounds and illustrate with two examples.
When does coordination compounds show coordination isomerism? Explain with suitable examples.
Define geometrical isomerism in relation to coordination compounds.
Write the geometrical isomers of [PtCl2(NH3)2].
Write the geometrical isomers of [Co(NH3)4Cl2].
Write the structure of the geometrical isomer of the following complex:
[Pd(NO2)2(NH3)2]
Write the structure of the geometrical isomer of the following complex:
[Pt(gly)2]
Write the structure of cis- and trans-isomers of the following complex:
[Co(en)2Cl2]−
Why is optical isomerism not common in square planar complexes?
Why is optical isomerism not common in tetrahedral complexes?
What type of coordination compounds show optical isomerism?
Draw optical isomers of [Co(en)3]3+.
Write the structures of optical isomers of the complex ion [Co(en)2Cl2]+.
Write the structures of optical isomers of the following complex:
[CoCl2(en)(NH3)2]+
Draw all the isomers (geometrical and optical) of [Co(NH3)Cl(en)2]2+.
What are the basic assumptions of valence bond theory in relation to coordination compounds?
Name the geometrical shape of the complex involving the following type of hybridisation:
sp3
Name the geometrical shape of the complex involving the following type of hybridisation:
dsp2
Name the geometrical shape of the complex involving the following type of hybridisation:
sp3d2
Name the geometrical shape of the complex involving the following type of hybridisation:
d2sp3
What do you understand by inner orbital complex?
What do you understand by outer orbital complex?
Give one example of an inner orbital.
Give one example of an outer orbital.
What do you understand by inner orbital complex?
What do you understand by outer orbital complex?
Illustrate high-spin complexes with a suitable example.
Illustrate low-spin complexes with a suitable example.
Explain the structure of K3[Fe(CN)6] on the basis of valence bond theory.
Explain the geometry of the complex K3[Fe(CN)6] on the basis of valence bond theory.
Explain the magnetic behaviour of the complex K3[Fe(CN)6] on the basis of valence bond theory.
Explain the formation of [Co(NH3)6]3+ ion on the basis of valence bond theory.
Based on valence bond theory, predict the magnetic behaviour of the [Fe(H2O)6]2+ complex.
Based on valence bond theory, predict the magnetic behaviour of the [Ni(NH3)4]2+ complex.
Discuss the formation of [Ni(CO)4] on the basis of valence bond theory.
[Ti(H2O)6]3+ is coloured, while [Sc(H2O)6]3+ is colourless – explain.
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[FeF6]3−
Explain the structure of [Cu(NH3)2]2+ on the basis of valence bond theory.
What are the limitations of VB theory?
Write a note on the stability of coordination compounds in solution.
State the factors which govern stability of complexes.
How does the presence of a particular ligand affect the thermodynamic stability of a complex? Illustrate your answer with a suitable example.
What is the role of KCN in the test of Cd2+ ion in the presence of Cu2+ ion?
Name the reagent used for the detection and estimation of Ni2+ ions. Write the equation.
Write the equation of the reagent used for the detection and estimation of Ni2+ ions.
How is the technique of complex formation useful in the extraction of metals? Explain with an example.
What is meant by the denticity of a ligand?
Give an example of unidentate ligand.
Give one example of didentate ligand.
Using IUPAC norms, write the formula for the following:
Tetrahydroxidozincate(II)
Using IUPAC norms, write the formula for the following:
Hexaamminecobalt (III) sulphate
Using IUPAC norms, write the formula for the following:
Potassium tetrachloridopalladate(II)
Using IUPAC norms, write the formula for the following:
Potassium tri(oxalato)chromate(III)
Using IUPAC norms write the formula for the following:
Diamminedichloridoplatinum(II)
Using IUPAC norms, write the formula for the following:
Hexaammineplatinum(IV)
Using IUPAC norms, write the formula for the following:
Potassium tetracyanidonickelate(II)
Using IUPAC norms, write the formula for the following:
Tetrabromidocuprate(II)
Write the formula for the following complex:
Pentaamminenitrito-O-Cobalt(III)
Using IUPAC norms, write the formula for the following:
Pentaamminenitrito-N-Cobalt (III)
Write the IUPAC name of the following coordination compound:
[Co(NH3)6]Cl3
Using IUPAC norms, write the systematic name of the following:
[Co(NH3)4Cl(NO2)]Cl
Using IUPAC norms, write the systematic name of the following:
[Ni(NH3)6]Cl2
Write the IUPAC name of the following coordination compound:
[Pt(NH3)2Cl(NH2CH3)]Cl
Using IUPAC norms, write the systematic name of the following:
[Mn(H2O)6]2+
Using IUPAC norms, write the systematic name of the following:
[Co(en)3]3+
Using IUPAC norms, write the systematic name of the following:
[Ti(H2O)6]3+
Using IUPAC norms, write the systematic name of the following:
[NiCl4]2−
Using IUPAC norms, write the systematic name of the following:
[Ni(CO)4]
Draw the structure of optical isomers of [Cr(OX)3]3−.
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [CoCl2(en)2]+.
Draw all the isomers (geometrical and optical) of [Co(NH3)Cl(en)2]2+.
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
Draw the structure of cis-dichloridotetracyanidochromate(III).
Draw the structure of Mer-triamminetrichloridocobalt (III).
Draw the structure of Fac-triaquatrinitro-N-cobalt (III).
Write the correct formula for the following coordination compound:
CrCl3·6H2O (violet, with 3 chloride ions/unit formula)
Write the correct formula for the following coordination compound:
CrCl3·6H2O (light green colour, with 2 chloride ions/unit formula)
Write the correct formula for the following coordination compound:
CrCl3.6H2O (dark green colour, with 1 chloride ion/unit formula)
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds LONG ANSWER TYPE QUESTIONS [Pages 547 - 548]
What are the coordination compounds?
Explain the following term in relation to coordination compounds:
Central metal ion
Explain the following term with respect to a coordination compound:
Coordination sphere
Explain the following term in relation to a coordination compound:
Ionic sphere
Explain the following term with respect to a coordination compound:
Coordination number
What is a ligand?
What are ligands and how are they classified? Give suitable examples.
Giving suitable examples, discuss the various types of structural isomerism exhibited by coordination compounds.
Giving suitable examples, discuss the geometrical isomerism shown by the following type of complex:
[Ma2b2]n±
Giving suitable examples, discuss the geometrical isomerism shown by the following type of complex:
[Ma2bc]n±
Giving suitable examples, discuss the geometrical isomerism shown by the following type of complex:
[M(AB)2]n±
What type of coordination compounds show optical isomerism?
Discuss the optical isomerism shown by the complex of the type [M(AA)2a2]n±. Give suitable examples.
Discuss the optical isomerism shown by the complex of the type [M(AA)a2b2]. Give suitable examples.
What are the salient features of the valence bond theory?
Explain the structure and magnetic behaviour of hexaamminenickel (II) ion on the basis of this theory.
Discuss the salient features of crystal field theory.
State the superiority of crystal field theory over valence bond theory.
Apply the crystal field theory to an octahedral complex.
Apply the crystal field theory to the tetrahedral complex.
What is meant by stability of a coordination compound in solution?
Explain its significance in predicting the thermodynamic stability of a complex in solution.
Discuss the factors which affect the stability of a complex.
Discuss the important applications of coordination compounds.
Explain on the basis of valence bond theory that [Ni(CN)4]2− ion with square planar structure is diamagnetic and the [NiCl4]2− ion with tetrahedral geometry is paramagnetic.
Explain the bonding in coordination compounds in terms of Werner’s postulates.
What are the main defects of Werner’s theory?
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Fe(CN)6]4−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[FeF6]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Co(OX)3]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[CoF6]3−
Discuss briefly, giving an example of the role of coordination compounds in biological systems.
Discuss briefly, giving an example of the role of coordination compounds in analytical chemistry.
Discuss briefly, giving an example of the role of coordination compounds in medicinal chemistry.
Discuss briefly, giving an example of the role of coordination compounds in extraction/metallurgy of metals.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds INTEGER TYPE QUESTIONS [Page 548]
For the octahedral complexes of Fe3+ in SCN− (thiocyanato-S) and in CN− ligand environments, the difference between the spin-only magnetic moments in Bohr magnetons (when approximated to the nearest integer) is ______ [Atomic number of Fe = 26].
If the freezing point of a 0.01 molal aqueous solution of a cobalt (III) chloride-ammonia complex (which behaves as a strong electrolyte) is −0.0558°C, the number of chloride(s) in the coordination sphere of the complex is ______ [Kt of water = 1.86 K kg mol−1].
The number of geometric isomers possible for the complex [CoL2Cl2]− (L = H2NCH2CH2O−) is ______.
The total number of possible isomers for [Pt(NH3)4Cl2]Br2 is ______ (nearest integer).
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 548 - 554]
Coordination number of Fe in K4[Fe(CN)6] is ______.
2
3
4
6
10
The complex [Co(NH3)5Br]SO4 will give white precipitate with ______.
BaCl2
AgNO3
KI
None
[Cr(NH3)6]3+ ion is ______.
paramagnetic
diamagnetic
square planar
tetrahedral
The coordination number and oxidation number of X in [X(SO4)(NH3)5]Cl are ______.
10 and 3
2 and 6
6 and 3
6 and 4
K3[CoF6] is a high spin complex. The hybridisation state of Co in this complex is ______.
sp3d
dsp2
sp3d2
d2sp3
Which of the following is the most likely structure of CrCl3.6H2O if `1/3` of total chlorine of the compound is precipitated by adding AgNO3 to its aqueous solution?
[Cr(H2O)6]Cl3
[Cr(H2O)4Cl2]Cl.2H2O
[Cr(H2O)5Cl]Cl2.H2O
[Cr(H2O)3Cl3](H2O)3
CrCl3.6H2O
Amongst Ni(CO)4], [Ni(CN)4]2− and [NiCl4]2−:
[Ni(CO)4] and [NiCl4]2− are diamagnetic and [Ni(CN)4]2− is paramagnetic.
[NiCl4]2− and [Ni(CN)4]2− are diamagnetic and [Ni(CO)4] is paramagnetic.
[Ni(CO)4] and [Ni(CN)4]2− are diamagnetic and [NiCl4]2− is paramagnetic.
[Ni(CO)4]is diamagnetic and [NiCl4]2− is paramagnetic.
The name of [Cr(NH3)4Cl2]NO3 is ______.
tetraamminodichloridochromium(III) nitrate
tetraaminodichloridochromium(II) nitrate
dichloridotetraamminechromium(II) nitrate
tetraamminedichloridochromium(III) nitrate
Which one of the following has tetrahedral geometry?
[Fe(CO)5]
[CO(NH3)6]2+
[NiCl4]2−
[Ni(CN)4]2−
The number of geometrical isomers for [Pt(NH3)2Cl2] is ______.
two
one
three
four
Which of the following complexes is likely to show optical activity?
[Cr(H2O)6]3+
trans-[Co(NH3)2(en)2]3+
trans-[Co(NH3)4Cl2]+
cis-[Co(NH3)2(en)2]3+
[Co(NH3)4Cl2]NO2 and [Co(NH3)4Cl(NO2)]Cl are ______.
geometrical isomers
optical isomers
linkage isomers
ionisation isomers
The number of ions formed when [Cu(NH3)4]SO4 is dissolved in water is ______.
1
2
4
zero
Metal present in chlorophyll is ______.
zinc
magnesium
calcium
sodium
Wilkinson’s catalyst is ______.
Ni
[(C6H5)3P]3 RhCl
LiAlH4
Fe2O3
The hardness of water is estimated by ______.
conductivity method
EDTA method
titrimetric method
distillation method
A complex involving dsp2 hybridisation has ______.
a square planar geometry
a tetrahedral geometry
an octahedral geometry
trigonalplanar geometry
In the compound, lithium tetrahydroaluminate, the ligand is ______.
H+
H
H−
None of these
Which of the following ligands does form a chelate?
Acetate
Oxalate
Cyanide
Ammonia
The geometry of Ni(CO)4 and Ni(PPh3)Cl2 are ______.
both square planar
tetrahedral and square planar respectively
both tetrahedral
square planar and tetrahedral respectively
Which of the following complex species does involve d2sp3 hybridisation?
Linkage
Ionisation
Hydrate
Coordination
Type of isomerism shown by [Cr(NH3)5NO2]Cl2 is ______.
linkage isomerism
hydrate isomerism
ligand isomerism
none of these
Coordination number of Fe in K4[Fe(CN)6] is ______.
2
3
4
6
10
The complex ion which has no ‘d’ electrons in the central metal atom is ______.
\[\ce{[MnO^{-}_4]}\]
[Co(NH3)6]3+
[Fe(CN)6]3−
[Cr(H2O)6]3+
The IUPAC name of K3[Co(NO2)5] is ______.
potassium (I) hexanitrocobaltate (II)
potassium (III) hexanitrocobaltate (III)
potassium hexanitrocobalt (0)
potassium hexanitrocobaltate (III)
Among the following ions, which one has the highest paramagnetism?
[Cr(H2O)6]3+
[Fe(H2O)6]2+
[Cu(H2O)6]2+
[Zn(H2O)2]2+
Which of the following give maximum number of isomers?
[Co(NH3)4Cl2]
[Ni(en)(NH3)4]2+
[Ni(C2O4)(en)2]
[Cr(SCN)2(NH3)4]2+
Coordination number of Ni in [Ni(C2O4)3]4− is ______.
3
6
4
5
The species having tetrahedral shape is ______.
[PdCl4]2−
[Ni(CN)4]2−
[Pd(CN)4]2−
[NiCl4]2−
Which of the following compounds is not coloured?
Na2[CuCl4]
Na3[AlF6]
K4[Fe(CN)6]
K3[Fe(CN)6]
The IUPAC name of [Co(en)2Cl2]Cl is ______.
bis(ethylenediamine)dichloridocobalt(III) chloride
dichloridobis(ethylenediamine)cobalt(III) chloride
dichloridobis(ethylenediamine)cobalt(I) chloride
trichloridobis(ethylenediamine)cobalt(0)
The correct order of hybridisation of the central atom in following species NH3, [PtCl4]2−, PCl5, BCl3 is: (At. No. Pt = 78)
dsp2, dsp3, sp2 and sp3
sp3, dsp2, dsp3, sp2
dsp2, sp2, dsp3, sp3
dsp2, sp3, sp2, dsp3
Consider the following complex:
[Co(NH3)5CO3]ClO4
The coordination number, oxidation number, number of d-electrons and number of unpaired d-electrons on the metal respectively are:
6, 3, 6, 0
7, 2, 7, 1
7, 1, 6, 4
6, 2, 7, 3
The most stable ion is ______.
[Fe(OH)5]3–
[FeCl6]3–
[Fe(CN)6]3–
[Fe(H2O)6]3+
One mole of the complex compound Co(NH3)5Cl3, gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles of AgNO3 solution to yield two moles of AgCl (s). The structure of the complex is ______.
[Co(NH3)5Cl]Cl2
[Co(NH3)3Cl3].2NH3
[Co(NH3)4Cl2]Cl.NH3
[Co(NH3)4Cl]Cl2.NH3
The coordination number of a central metal atom in a complex is determined by ______.
the number of ligands around a metal ion bonded by sigma bonds
the number of ligands around a metal ion bonded by pi-bonds
the number of ligands around a metal ion bonded by sigma and pi-bonds both
the number of only anionic ligands bonded to the metal ion
Which one of the following complexes is an outer orbital complex?
[Fe(CN)6]4−
[Mn(CN)6]4−
[Co(NH3)6]3+
[Ni(NH3)6]2+
Coordination compounds have great importance in biological systems. In this context which of the following statements is incorrect?
Chlorophylls are green pigments in plants and contain calcium.
Haemoglobin is the red pigment of blood and contains iron.
Cyanidocobalamin is vitamin-B12 and contains cobalt.
Carboxypeptidase-A is an enzyme and contains zinc.
Which one of the following has largest number of isomers?
[Ru(NH3)4Cl2]+
[Co(NH3)5Cl]2+
[Ir(PR3)2H(CO)]2+
[Co(en)2Cl2]+
The correct order of magnetic moments (spin-only values in BM) among the following is:
[MnCl4]2– > [CoCl4]2– > [Fe(CN)6]4–
[MnCl4]2– > [Fe(CN)6]4– > [CoCl4]2–
[Fe(CN)6]4– > [MnCl4]2– > [CoCl4]2–
[Fe(CN)6]4– > [CoCl4]2– > [MnCl4]2–
Which of the following compounds show optical isomerism?
[Cu(NH3)4]3−
[ZnCl4]2−
[Cr(C2O4)3]3−
[Co(CN)6]3−
[Cu(NH3)4]2+
Which one of the following cyanido complexes would exhibit the lowest value of paramagnetic behaviour?
(Atomic no. Cr = 24, Mn = 25, Fe = 26, Co = 27)
[Co(CN)6]3−
[Fe(C2O4)3]3−
[Mn(CN)6]3−
[Cr(CN)6]3−
The value of the spin only magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
d5 (in strong ligand field)
d3 (in weak as well as in strong fields)
d4 (in weak ligand fields)
d4 (in strong ligand fields)
The IUPAC name for the complex [Co(NO2)(NH3)5]Cl2 is ______.
nitrito-N-pentaaminecobalt (III) chloride
nitrito-N-pentaamminecobalt (II) chloride
pentaamminenitrito-N-cobalt (II) chloride
pentaamminenitrito-N-cobalt (III) chloride
Nickel (Z = 28) combines with a uninegative monodentate ligand X− to form a paramagnetic complex [NiX4]2−. The number of unpaired electron(s) in the nickel and geometry of this complex ion respectively are ______.
one, tetrahedral
two, tetrahedral
one, square planar
two, square planar
The coordination number and the oxidation state of the element ‘E’ in the complex [E(en)2(C2O4)]NO2 (where (en) is ethylenediamine) are, respectively ______.
6 and 2
4 and 2
4 and 3
6 and 3
In which of the following octahedral complexes of Co (at. no. 27), will the magnitude of Δ0 be the highest?
[Co(CN)6]3−
[Co(C2O4)3]3−
[Co(H2O)6]3+
[Co(NH3)6]3+
Which of the following complex ions is not expected to absorb visible light?
[Ni(CN)4]2−
[Cr(NH3)6]3+
[Fe(H2O)6]2+
[Ni(H2O)6]2+
The existence of two different coloured complexes with the composition of [Co(NH3)4Cl2]+ is due to ______.
linkage isomerism
geometrical isomerism
coordination isomerism
ionisation isomerism
The d-electron configurations of Cr2+, Mn2+, Fe2+ and Co2+ are d4, d5, d6 and d7 respectively. Which one of the following will exhibit minimum paramagnetic behaviour?
(At. no. Cr = 24, Mn = 25, Fe = 26, Co = 27)
[Fe(H2O)6]2+
[Co(H2O)6]2+
[Cr(H2O)6]2+
[Mn(H2O)6]2+
The complexes [Co(NH3)6][Cr(CN)6] and [Cr(NH3)6][Co(CN)6] are the examples of which type of isomerism?
Ionisation isomerism
Coordination isomerism
Geometrical isomerism
Linkage isomerism
The complex [(Pt(Py)(NH3)BrCl] will have how many geometrical isomers?
3
4
0
2
Which one of the following is an outer orbital complex and exhibits paramagnetic behaviour?
[Ni(NH3)6]2+
[Zn(NH3)6]2+
[Cr(NH3)6]3+
[Co(NH3)6]3+
Among the following complexes, the one which shows zero crystal field stabilisation energy (CFSE) is ______.
[Mn(H2O)6]3+
[Fe(H2O)6]3+
[Co(H2O)6]2+
[Co(H2O)6]3+
Which of the following complexes is used to be as an anticancer agent?
[mer-[Co(NH3)3Cl3]
cis-[PtCl2(NH3)2]
cis-K2[PtCl2Br2]
Na2CoCl4
Which of the following complex species is not expected to exhibit optical isomerism?
[Co(en)3]3+
[Co(en)2Cl2]+
[Co(NH3)3Cl3]
[Co(en)(NH3)Cl2)]+
The octahedral complex of a metal ion M3+ with four monodentate ligands L1, L2, L3, and L4 absorb wavelengths in the region of red, green, yellow and blue respectively. The increasing order of ligand strength of the four ligands is:
L4 < L3 < L2 < L1
L1 < L3 < L2 < L4
L3 < L2 < L4 < L1
L1 < L2 < L3 < L4
The complex ion [Ni(CN)4]2− is ______.
square planar and diamagnetic
tetrahedral and paramagnetic
square planar and paramagnetic
tetrahedral and diamagnetic
The number of geometric isomers that can exist for square planar [Pt(Cl)(py)(NH3)(NH2OH)]+ is (py = pyridine) ______.
2
3
4
6
Which of the following compounds is not coloured yellow?
Zn2[Fe(CN)6]
K3[Co(NO2)6]
(NH4)3[As(Mo3O10)4]
BaCrO4
The pair having the same magnetic moment is ______.
[At. No.: Cr = 24, Mn = 25, Fe = 26, Co = 27]
[Cr(H2O)6]2+ and [CoCl4]2−
[Cr(H2O)6]2+ and [Fe(H2O)6]2+
[Mn(H2O)6]2+ and [Cr(H2O)6]2+
[CoCl4]2− and [Fe(H2O)6]2+
Which one of the following complexes shows optical isomerism? (en = ethylenediamine)
[Co(NH3)3Cl3]
cis[Co(en)2Cl2]Cl
trans[Co(en)2Cl2]Cl
[Co(NH3)4Cl2]Cl
Among [Ni(CO)4], [NiCl4]2−, [Co(NH3)4Cl2]Cl, Na3[CoF6], Na2O2 and CsO2, the total number of paramagnetic compounds is ______.
2
3
4
5
Which of the following has longest C−O bond length? (Free C−O bond length in CO is 1.128 A.)
Ni(CO)4
[Co(CO)4]Θ
[Fe(CO)4]2−
[Mn(CO)6]+
The correct order of the stoichiometries of AgCl formed when AgNO3 in excess is treated with the complexes:
CoCl3·6NH3, CoCl3·5NH3, CoCl3·4NH3 respectively is:
1 AgCl, 3AgCl, 2AgCl
3 AgCl, 1 AgCl, 2 AgCl
3 AgCl, 2 AgCl, 1 AgCl
2 AgCl, 3 AgCl, 1 AgCl
Correct increasing order for the wavelengths of absorption in the visible region for the complexes of Co3+ is:
[Co(en)3]3+, [Co(NH3)6]3+, [Co(H2O)6]3+
[Co(H2O)6]3+, [Co(en)3]3+, [Co(NH3)6]3+
[Co(H2O)6]3+, [Co(NH3)6]3+, [Co(en)3]3+
[Co(NH3)6]3+, [Co(en)3]3+, [Co(H2O)6]3+
Pick out the correct statement with respect [Mn(CN)6]3−.
It is sp3d2 hybridised and octahedral.
It is sp3d2 hybridised and tetrahedral.
It is d2sp3 hybridised and octahedral.
It is dsp2 hybridised and square planar.
On treatment of 100 ml of 0.1 M solution of CoCl3·6H2O with excess AgNO3; 1.2 × 1022 ions are precipitated. The complex is ______.
[Co(H2O)6]Cl3
[Co(H2O)5Cl]Cl2.H2O
[Co(H2O)4Cl2]Cl.2H2O
[Co(H2O)3Cl3].3H2O
Consider the following reaction and statements:
\[\ce{[Co(NH3)4Br2]^+ + Br^- -> [Co(NH3)3Br3] + NH3}\]
- Two isomers are produced if the reactant complex ion is a cis-isomer.
- Two isomers are produced if the reactant complex ion is a trans-isomer.
- Only one isomer is produced if the reactant complex ion is a trans-isomer.
- Only one isomer is produced if the reactant complex ions is a cis-isomer.
The correct statements are:
(I) and (II)
(I) and (III)
(III) and (IV)
(II) and (IV)
The type of isomerism shown by the complex [CoCl2(en)2] is ______.
ionization isomerism
coordination isomerism
geometrical isomerism
linkage isomerism
The geometry and magnetic behaviour of the complex [Ni(CO)4] are ______.
square planar geometry and paramagnetic
tetrahedral geometry and diamagnetic
square planar geometry and diamagnetic
tetrahedral geometry and paramagnetic
The species that can have a trans-isomer is ______.
(en = ethane-1, 2-diamine, ox = oxalate)
[Cr(en)2(ox)]+
[Pt(en)Cl2]
[Pt(en)2Cl2]2+
[Zn(en)Cl2]
Three complexes,
[CoCl(NH3)5]2+ (I),
[Co(NH3)5H2O]3+ (II) and
[Co(NH3)6]3+ (III)
absorb light in the visible region. The correct order of the wavelength of light absorbed by them is:
(III) > (I) > (II)
(III) > (II) > (I)
(I) > (II) > (III)
(II) > (I) > (III)
The incorrect statement is:
the colour of [CoCl(NH3)5]2+ is violet as it absorbs the yellow light.
the gemstone, ruby, has Cr3+ ions occupying the octahedral sites of beryl.
the spin-only magnetic moment of [Ni(NH3)4(H2O)2]2+ is 2.83 BM.
the spin-only magnetic moments of [Fe(H2O)6]2+ and [Cr(H2O)6]2+ are nearly similar.
The crystal field stabilization energy (CFSE) of [Fe(H2O)6]Cl2 and K2[NiCl4] respectively, are ______.
−0.4 Δo and −0.8 Δt
−0.6 Δo and −0.8 Δt
−2.4 Δo and −1.2 Δt
−0.4 Δo and −1.2 Δt
The Crystal Field Stabilisation Energy (CFSE) for [CoCl6]4− is 18000 cm−1. The CFSE for [CoCl4]2− will be ______.
8000 cm−1
6000 cm−1
16000 cm−1
18000 cm−1
What is the correct electronic configuration of the central atom in K4[Fe(CN)6] based on crystal field theory?
\[\ce{t^4_{2g}e^2_{g}}\]
\[\ce{t^6_{2g}e^0_{g}}\]
\[\ce{e^3 t^3_{2}}\]
\[\ce{e^4 t^2_{2}}\]
Which of the following is the correct order of increasing field strength of ligands to form coordination compounds?
\[\ce{SCN^- < F^- < C_2O^{2-}_4 < CN^-}\]
\[\ce{SCN^- < F^- < CN^- < C_2O^{2-}_4}\]
\[\ce{F^- < SCN^- < C_2O^{2-}_4 < CN^-}\]
\[\ce{CN^- < C_2O^{2-}_4 < SCN^- < F^- }\]
For octahedral Mn(II) and tetrahedral Ni(II) complexes, consider the following statements:
- Both the complexes can be high spin.
- Ni(II) complex can very rarely below spin.
- With strong field Ligands, Mn(II) complexes can be low spin.
- Aqueous solution of Mn (II) ions is yellow in colour.
The correct statements are:
(i) and (ii) only
(i), (ii) and (iii) only
(i), (iii) and (iv) only
(ii), (iii) and (iv) only
Consider that d6 metal ion (M2+) forms a complex with aqua ligands and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilization energy of the complex are ______.
Tetrahedral and −1.6 Δt + 1P
Octahedral and −2.4 Δo + 2P
Tetrahedral and −0.6 Δt
Octahedral and −1.6 Δo
The electronic spectrum of [Ti(H2O)6]3+ shows a single broad peak with a maximum at 20,300 cm−1. The crystal field stabilisation energy (CFSE) of the complex ion, in kJ mol−1, is ______. (1 kJ mol−1 = 83.7 cm−1)
242.5
145.5
83.7
97
The complex that can show optical activity is ______.
trans-[Cr(Cl2)(ox)2]3−
trans-[Fe(NH3)2(CN)4]−
cis-[Fe(NH3)2(CN)4]−
cis-[Cr(Cl2)(ox)2]3− (ox = oxalate)
The molecule in which hybrid MOs involve only one d-orbital of the central atom is ______.
[Ni(CN4)]2−
XeF4
[CrF6]3−
BrF5
The one that can exhibit highest paramagnetic behaviour among the following is:
[Fe(en)(bpy)(NH3)2]2+
[Pd(gly)2]
[Ti(NH3)6]3+
[Co(ox)2(OH)2]− (Δo > P)
The Crystal Field Stabilisation Energy (CFSE) of [CoF3(H2O)3] (Δo > P) is ______.
−0.4 Δo
−0.8 Δo
−0.4 Δo + P
−0.8 Δo + 2P
The value of the crystal field stabilisation energies for a high spin d6 metal ion in octahedral and tetrahedral fields, respectively, are ______.
−2.4 Δo and −0.6 Δt
−1.6 Δo and −0.4 Δt
−0.4 Δo and −0.27 Δt
−0.4 Δo and −0.6 Δt
Ethylene diamine tetraacetate (EDTA) ion is ______.
Tridentate ligand with three “N” donor atoms.
Hexadentate ligand with four “O” and two “N” donor atoms.
Unidentate ligand
Bidentate ligand with two “N” donor atoms.
Match List - I with List - II.
| List - I | List - II | ||
| (A) | [Fe(CN)6]3− | (i) | 5.92 BM |
| (B) | [Fe(H2O)6]3+ | (ii) | 0 BM |
| (C) | [Fe(CN)6]4− | (iii) | 4.90 BM |
| (D) | [Fe(H2O)6]2+ | (iv) | 1.73 BM |
Choose the correct answer from the options given below.
(A)-(iv), (B)-(i), (C)-(ii), (D)-(iii)
(A)-(iv), (B)-(ii), (C)-(i), (D)-(iii)
(A)-(ii), (B)-(iv), (C)-(iii), (D)-(i)
(A)-(i), (B)-(iii), (C)-(iv), (D)-(ii)
The IUPAC name of the complex [Ag(H2O)2] [Ag(CN)2] is ______.
diaquasilver (II) dicyanidoargeltate (II)
dicyanidosilver (I) diaquaargentate (I)
diaquasilver (I) dicyanidoargentate (I)
dicyanidosilver (II) diaquaargentate (II)
The order of energy absorbed which is responsible for the order of complexes:
- [Ni(H2O)2(en)]2+
- [Ni(H2O)4(en)]2+ and
- [Ni(en)3]2+
(C) > (B) > (A)
(C) > (A) > (B)
(B) > (A) > (C)
(A) > (B) > (C)
Match List-I with List-II.
| List-I | List-II | ||
| (A) | [Co(NH3)6][Cr(CN)6] | (i) | Linkage isomerism |
| (B) | [Co(NH3)3(NO2)3] | (ii) | Solvate isomerism |
| (C) | [Cr(H2O)6]Cl3 | (iii) | Co-ordination isomerism |
| (D) | cis-[CrCl2(OX)2]3− | (iv) | Optical isomerism |
Choose the correct answer from the options given below.
A-(iii), B-(i), C-(ii), D-(iv)
A-(iv), B-(ii), C-(iii), D-(i)
A-(ii), B-(i), C-(iii), D-(iv)
A-(i), B-(ii), C-(iii), D-(iv)
The correct structures of trans-[NiBr2(PPh3)2] and meridonial- [Co(NH3)3(NO2)3] respectively are:
The calculated spin only magnetic moments of [Cr(NH3)6]3+ and [CuF6]3− in B.M., respectively are ______.
[Atomic numbers of Cr and Cu are 24 and 29 respectively]
3.87 and 2.84
4.90 and 1.73
3.87 and 1.73
4.90 and 2.84
The pair of complexes wherein both exhibit tetrahedral geometry is (are): (Note: Py = Pyridine)
(Given atomic numbers of Fe, CO, Ni and Cu are 26, 27, 28 and 29 respectively.)
[FeCl4]− and [Fe(CO)4]2−
[Co(CO)4]− and [CoCl4]2−
[Ni(CO)4] and [Ni(CN)4]2−
[Cu(Py)4]+ and [Cu(CN)4]3−
The reaction of K3[Fe(CN)6] with freshly prepared FeSO4 solution produces a dark blue precipitate called Turnbull’s blue. Reaction of K4[Fe(CN)6] with the FeSO4. Solution in complete absence of air produces a white precipitate X, which turns blue in air. Mixing the FeSO4 solution with NaNO3, followed by a slow addition of concentrated H2SO4 through the sides of the test tube produces a brown ring.
The precipitate ‘X’ is:
Fe4[Fe(CN)6]3
Fe[Fe(CN)6]
K2Fe[Fe(CN)6]
KFe[Fe(CN)6]
The reaction of K3[Fe(CN)6] with freshly prepared FeSO4 solution produces a dark blue precipitate called Turnbull’s blue. Reaction of K4[Fe(CN)6] with the FeSO4. Solution in complete absence of air produces a white precipitate X, which turns blue in air. Mixing the FeSO4 solution with NaNO3, followed by a slow addition of concentrated H2SO4 through the sides of the test tube produces a brown ring.
Among the following the brown ring is due to the formation of:
[Fe(NO)2(SO4)2]2−
[Fe(NO)2(H2O)4]3+
[Fe(NO)4(SO4)2]
[Fe(NO)(H2O)5]2+
Transition metal complex with highest value of crystal field splitting (Δo) will be ______.
[Cr(H2O)6]3+
[Mo(H2O)6]3+
[Fe(H2O)6]3+
[Os(H2O)6]3+
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
[Ag(NH3)4]Cl2
[Ag(Cl)2(NH3)2]
[Ag(NH3)2]Cl
[Ag(NH3)Cl]Cl
Which statement is not true with respect to nitrate ion test?
A dark brown ring is formed at the junction of two solutions.
Ring is formed due to nitroferrous sulphate complex.
The brown complex is [Fe(H2O)5NO]SO4.
Heating the nitrate salt with conc. H2SO4 light brown fumes are evolved.
Which of the following will have maximum stabilization due to crystal field?
[Ti(H2O)6]3+
[Co(H2O)6]2+
[Co(CN)6]3−
[Cu(NH3)4]2+
Arrange the following coordination compounds in the increasing order of magnetic moments. (Atomic numbers: Mn = 25, Fe = 26)
- [FeF6]3−
- [Fe(CN)6]3−
- [MnCl6]3− (High spin)
- [Mn(CN)6]3−
A < B < D < C
B < D < C < A
A < C < D < B
B < D < A < C
Given below are two statements:
Statement I: [Ni(CN)4]2− is square planar and diamagnetic complex with dsp2 hybridization for Ni but [Ni(Co)4] is tetrahedral, paramagnetic and with sp3 hybridization for Ni.
Statement II: [NiCl4]2− and [Ni(CO)4] both have same d-electron configuration, have same geometry and are paramagnetic.
In light of the above statements, choose the correct answer from the options given below:
Both statement I and statement II are true.
Both statement I and statement II are false.
Statement I is correct but statement II is false.
Statement I is incorrect but statement II is true.
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
Both Statement I and Statement II are correct.
Both Statement I and Statement II are incorrect.
Statement I is correct but Statement II is incorrect.
Statement I is incorrect but Statement II is correct.
List-I contains metal species and List-II contains their properties:
| List-I | List-II | ||
| (A) | [Cr(CN)6]4− | (i) | t2g orbitals contain 4 electrons |
| (B) | [RuCl6]2− | (ii) | µ(spin only) = 4.9 BM |
| (C) | [Cr(H2O)6]2+ | (iii) | low spin complex ion |
| (D) | [Fe(H2O)6]2+ | (iv) | metal ion in +4 oxidation state |
| (v) | d4 species | ||
[Given: Atomic number of Cr = 24, Ru = 44, Fe = 26]
Match each metal species in List-I with their properties in List-II and choose the correct option.
(A)-(iii), (v) B-(i), (iv) C-(ii), (v) D-(i), (ii)
(A)-(iii), (iv) B-(i), (v) C-(i), (ii) D-(ii), (v)
(A)-(i), (iii) B-(iii), (iv) C-(iii), (v) D-(i), (v)
(A)-(ii), (v) B-(iv), (v) C-(i), (v) D-(ii), (iii)
The reaction of Pb(NO3)2 and NaCl in water produces a precipitate that dissolves upon the addition of HCl of appropriate concentration. The dissolution of the precipitate is due to the formation of ______.
PbCl2
PbCl4
[PbCl4]2−
[PbCl6]2−
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds FILL IN THE BLANKS TYPE QUESTIONS [Page 556]
The total number of ions furnished by K4[Fe(CN)6] in solution is ______.
The electrically charged species formed by the union of a central metal atom or ion with one or more ligands is called ______.
The coordination number of platinum in [PtCl4]2− is ______.
Ethylenediamine is a ______ dentate ligand.
The IUPAC name of the complex K3[AgF4] is ______.
The isomerism arising due to the interchange of ligands between the coordination spheres of positive and negative parts is called ______ isomerism.
When all the electrons in a complex are paired, it is ______ in nature.
Outer orbital complexes involve ______ hybridisation and are ______ spin complexes.
The complexes involving ______ hybridisation are called inner orbital complexes. They are ______ spin complexes.
The oxidation state of Fe in [Fe(CN)6]3− is ______.
The complex [Co(NH3)6]3+ has a ______ geometry and is ______ magnetic in nature.
The number of unpaired electrons present in [Cr(NH3)6]3+ is ______.
The complex [Ni(CO)4]0 is ______ in shape and is ______ magnetic.
Tetracyanocuprate(II) ion involves ______ hybridisation and is ______ in shape.
Greater the basic strength of the ligand, ______ is the stability of the complex.
The ligand used in the estimation of the hardness of water is ______.
Vitamin-B12 is a complex of ______.
Cisplatin found useful in the treatment of cancer has the formula ______.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds ASSERTION-REASON TYPE QUESTIONS [Page 557]
The questions given below consist of an Assertion and a Reason. You have to choose the correct answer (a), (b), (c) or (d) according to the following clue:
Assertion: Mohr’s salt, FeSO4·(NH4)2SO4·6H2O is not a coordination compound.
Reason: The aqueous solution of this salt gives the test of Fe2+, NH4 and \[\ce{SO^{4-}_2}\] ions.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The complexes, [Co(pn)2Cl2]+ and [Co(tn)2Cl2]+ are ligand isomers.
Reason: The ligand 1,2-diaminopropane can exist both as 1,2-diaminopropane (pn) and 1,3-diaminopropane (tn).
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The tetrahedral complexes can show geometrical isomerism.
Reason: This is because all the four ligands lie at the same distance from central metal atom in a tetrahedral geometry.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The trans-[CoCl2(en)2]+ is optically inactive.
Reason: It is symmetrical because it contains a plane of symmetry.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The complex [Fe(H2O)6]2+ is paramagnetic in nature.
Reason: It consists of two unpaired electrons.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds 'NCERT TEXT-BOOK' Exercises [Pages 557 - 560]
Explain the bonding in coordination compounds in terms of Werner’s postulates.
FeSO4 solution mixed with (NH4)2SO4 solution in 1 : 1 molar ratio gives the test of Fe2+ ion but CuSO4 solution mixed with aqueous ammonia in 1 : 4 molar ratio does not give the test of Cu2+ ion. Explain why?
Explain the following, giving two examples:
Coordination entity
Explain with two examples of the following:
ligand
Explain the following, giving two examples:
Coordination number
Explain the following, giving two examples:
Coordination polyhedron
Explain the following, giving two examples:
Homoleptic
Explain the following, giving two examples:
Heteroleptic
What is meant by unidentate ligand?
What is meant by didentate ligand?
What is meant by ambidentate ligand?
Give two examples of unidentate ligands.
Give two examples of didentate ligands.
Give two examples of ambidentate ligands.
Specify the oxidation number of the metal in the following coordination entity:
[Co(H2O)(CN)(en)2]2+
Specify the oxidation number of the metal in the following coordination entity:
[CoBr2(en)2]+
Specify the oxidation number of the metal in the following coordination entity:
[PtCl4]2−
Specify the oxidation number of the metal in the following coordination entity:
K3[Fe(CN)6]
Specify the oxidation number of the metal in the following coordination entity:
[Cr(NH3)3Cl3]
Using IUPAC norms, write the formula for the following:
Tetrahydroxidozincate(II)
Using IUPAC norms, write the formula for the following:
Potassium tetrachloridopalladate(II)
Using IUPAC norms write the formula for the following:
Diamminedichloridoplatinum(II)
Using IUPAC norms, write the formula for the following:
Potassium tetracyanidonickelate(II)
Write the formula for the following complex:
Pentaamminenitrito-O-Cobalt(III)
Using IUPAC norms, write the formula for the following:
Hexaamminecobalt (III) sulphate
Using IUPAC norms, write the formula for the following:
Potassium tri(oxalato)chromate(III)
Using IUPAC norms, write the formula for the following:
Hexaammineplatinum(IV)
Using IUPAC norms, write the formula for the following:
Tetrabromidocuprate(II)
Using IUPAC norms, write the formula for the following:
Pentaamminenitrito-N-Cobalt (III)
Write the IUPAC name of the following coordination compound:
[Co(NH3)6]Cl3
Write the IUPAC name of the following coordination compound:
[Pt(NH3)2Cl(NH2CH3)]Cl
Using IUPAC norms, write the systematic name of the following:
[Ti(H2O)6]3+
Using IUPAC norms, write the systematic name of the following:
[Co(NH3)4Cl(NO2)]Cl
Using IUPAC norms, write the systematic name of the following:
[Mn(H2O)6]2+
Using IUPAC norms, write the systematic name of the following:
[NiCl4]2−
Using IUPAC norms, write the systematic name of the following:
[Ni(NH3)6]Cl2
Using IUPAC norms, write the systematic name of the following:
[Co(en)3]3+
Using IUPAC norms, write the systematic name of the following:
[Ni(CO)4]
List various types of isomerism possible for coordination compounds, giving an example of each.
How many geometrical isomers are possible in the following coordination entity?
[Cr(C2O4)3]3−
How many geometrical isomers are possible in the following coordination entity?
[Co(NH3)3Cl3]
Draw the structure of optical isomers of [Cr(C2O4)3]3−.
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [CoCl2(en)2]+.
Draw all the isomers (geometrical and optical) of [Co(NH3)Cl(en)2]2+.
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
Write all the geometrical isomers of [Pt(NH3)(Br)(Cl)(py)] and how many of these will exhibit optical isomers?
Aqueous copper sulphate solution (blue in colour) gives a green precipitate with aqueous potassium fluoride. Explain this experimental result.
Aqueous copper sulphate solution (blue in colour) gives a bright green solution with aqueous potassium chloride. Explain this experimental result.
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Fe(CN)6]4−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[FeF6]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Co(C2O4)3]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[CoF6]3−
Draw figure to show the splitting of d orbitals in an octahedral crystal field.
What is the spectrochemical series?
What is the difference between a weak field ligand and a strong field ligand?
What is crystal field splitting energy?
How does the magnitude of Δ0 decide the actual configuration of d orbitals in a coordination entity?
[Cr(NH3)6]3+ is paramagnetic while [Ni(CN)4]2− is diamagnetic. Explain why?
A solution of [Ni(H2O)6]2+ is green but a solution of [Ni(CN)4]2− is colourless. Explain.
[Fe(CN)6]4− and [Fe(H2O)6]2+ are of different colours in dilute solutions. Why?
Discuss the nature of bonding in metal carbonyls.
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
cis-[CrCl2(en)2]Cl
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K[Cr(H2O)2(C2O4)2].3H2O
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[Co(NH3)5Cl]Cl2
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[CrCl3(py)3]
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
Cs[FeCl4]
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K4[Mn(CN)6]
What is meant by stability of a coordination compound in solution?
State the factors which govern stability of complexes.
What is meant by the chelate effect?
Give an example of chelate effect.
Discuss briefly, giving an example of the role of coordination compounds in biological systems.
Discuss briefly, giving an example of the role of coordination compounds in medicinal chemistry.
Discuss briefly, giving an example of the role of coordination compounds in analytical chemistry.
Discuss briefly, giving an example of the role of coordination compounds in extraction/metallurgy of metals.
How many ions are produced from the complex Co(NH3)6Cl2 in solution?
6
4
3
2
Amongst the following ions, which one has the highest magnetic moment value?
[Cr(H2O)6]3+
[Fe(H2O)6]2+
[Zn(H2O)6]2+
The oxidation number of cobalt in K[Co(CO)4] is ______.
+1
+3
−1
−3
Amongst the following, the most stable complex is:
[Fe(H2O)6]3+
[Fe(NH3)6]3+
[Fe(C2O4)3]3−
[FeCl6]3−
What will be the correct order for the wavelengths of absorption in the visible region for the following?
[Ni(NO2)6]4−, [Ni(NH3)6]2+, [Ni(H2O)6]2+
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 9 Coordination Compounds QUESTIONS FROM ISC EXAMINATION PAPERS [Pages 561 - 562]
Correct the following,statement by changing the underlined part of the sentence.
In a coordination complex, donation of electron pair takes place from the central metal atom to the ligands.
Name the type of isomerism exhibited by the following pair of compounds:
[Co(NH3)5(ONO)]Cl2 and [Co(NH3)5(NO2)]Cl2
Name the type of isomerism exhibited by the following pair of compounds:
[Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O
Give the IUPAC name for the following:
Na3[AlF6]
Write the IUPAC name of the following coordination compound:
[Co(NH3)6]Cl3
For the complex ion, [Fe(CN)6]3−:
Show the hybridisation diagrammatically.
For the complex ion, [Fe(CN)6]3−:
Is it an inner orbital complex or an outer orbital complex?
For the complex ion, [Fe(CN)6]3−:
State its magnetic property.
Write the formula of the following coordination compound:
potassium tetracyanidonickel (0)
Write the formula of the following coordination compound:
triamminetrinitrocobalt (III)
[CoF6]3− is a coordination complex ion.
What is the oxidation number of cobalt in the complex?
[CoF6]3− is a coordination complex ion.
How many unpaired electrons are there in the complex?
[CoF6]3− is a coordination complex ion.
State the magnetic behaviour of the complex.
[CoF6]3− is a coordination complex ion.
Give the IUPAC name of the complex.
State the geometry and magnetic property of tetracarbonyl nickel according to the valence bond theory.
What type of structural isomers are [Pt(OH)2(NH3)4]SO4 and [PtSO4(NH3)4](OH)2? How will you identify the isomers with a chemical test?
Name the coordination compound used for the following:
Treatment of cancer
Name the coordination compound used for the following:
Treatment of lead poisoning
Write the formula of the following compound:
Triamminetriaquachromium (III) chloride
Write the formula of the following compound:
Potassium hexacyanidoferrate (III)
Name the type of isomerism shown by the following pair of compounds:
[CoCl(H2O)(NH3)4]Cl2 and [CoCl2(NH3)4]Cl.H2O
Name the type of isomerism shown by the following pair of compounds:
[Pt(NH3)4][PtCl6] and [Pt(NH3)4Cl2][PtCl4]
For the coordination complex ion [Co(NH3)6]3+:
State the type of hybridisation of the complex ion.
For the coordination complex ion [Co(NH3)6]3+:
State the magnetic nature of the complex.
Write the IUPAC name of the following coordination compound:
[Cr(NH3)4(H2O)2]Cl3
Write the IUPAC name of the following coordination compound:
[PtCl2(NH3)4][PtCl4]
State the hybridization and magnetic property of [Fe(CN)6]3− ion according to the valence bond theory.
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
Write the structures of optical isomers of the complex ion [Co(en)2Cl2]+.
Write the IUPAC name of the following:
[CO(NH3)4SO4]NO3
Write the IUPAC name of the following complex:
K[Pt(NH3)Cl3]
What type of isomerism is exhibited by the following pair of compounds?
[PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2
What type of isomerism is exhibited by the following pair of compounds?
[Cr(SCN)(H2O)5]2+ and [Cr(NCS)(H2O)5]2+
How does K2[PtCl4] get ionised when dissolved in water? Will it form a precipitate when AgNO3 solution is added to it? Give a reason for your answer.
Using IUPAC norms write the formulae for Potassium trioxalatoaluminate(III)
Write the formula of the following compound:
Hexaaquairon (II) sulphate
Name the type of isomerism shown by the following compounds:
[Cu(NH3)4][PtCl4] and [Pt(NH3)4][CuCl4]
Name the type of isomerism shown by the following pair of compounds:
[Co(Pn)2Cl2]+ and [Co(tn)2Cl2]+
For the coordination complex ion [Co(NH3)6]3+ :
Give the IUPAC name of the complexion.
For the coordination complex ion [Co(NH3)6]3+:
What is the oxidation number of cobalt in the complexion?
For the coordination complex ion [Co(NH3)6]3+:
State the type of hybridisation of the complex ion.
For the coordination complex ion [Co(NH3)6]3+:
State the magnetic nature of the complex.
What type of isomerism is shown by the following coordination compounds?
[PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2.
Write their IUPAC names.
For the complex ion [Fe(CN)6]3−, state the type of hybridisation.
For the complex ion, [Fe(CN)6]3−:
State its magnetic property.
For the complex ion [Fe(CN)6]3−, state the oxidation number of the central metal atom.
Write the IUPAC name of [Co(en)2Cl2]+ ion.
Draw the structures of [Co(en)2Cl2]+ geometrical isomers.
When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3 moles of AgCl are precipitated per mole of the compound. Write the structural formula of the coordination compound.
When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3moles of AgCl are precipitated per mole of the compound. Write the IUPAC name of the coordination compound.
Write the IUPAC name of the K3[Fe(C2O4)3].
Write the IUPAC name of the following:
Co[(NH3)5Cl]SO4
[Fe(CN)6]4− is a coordination complex ion.
Calculate the oxidation number of iron in the complex.
[Fe(CN)6]4− is a coordination complex ion.
Is the complex ion diamagnetic or paramagnetic?
[Fe(CN)6]4− is a coordination complex ion.
What is the hybridisation state of the central metal atom?
[Fe(CN)6]4− is a coordination complex ion.
Write the IUPAC name of the complex ion.
Give the IUPAC name of the following complex:
[Co(NH3)6][CdCl5]
Give the IUPAC name of the following complex:
[Co(en)2Cl2]
Give the IUPAC name of the following complex:
K3[Al(C2O4)3]
With reference to the coordination complex ion [Fe(H2O)6]2+ answer the following: (at. no. of Fe = 26)
- Give the IUPAC name of the complex ion.
- What is the oxidation number of the central metal atom?
- How many unpaired electrons are there in the complex ion?
- State the type of hybridisation of the complex ion.
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Name the type of isomerism exhibited by the following pair of compounds:
[Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O
Name the type of isomerism exhibited by the following pair of compounds:
[Co(NH3)6][Cr(CN)6] and [Co(CN)6][Cr(NH3)6]
Using the valence bond approach, predict the shape, hybridisation and magnetic behaviour of [Ni(CO)4]. (at. no. of Ni = 28)
Write the IUPAC name of the following compound:
[Co(NH3)4Cl2]Cl
Give the IUPAC name of the following complex:
K3[Al(C2O4)3]
[Fe(CN)6]4− is a coordination complex ion.
Is the complex ion diamagnetic or paramagnetic?
[Fe(CN)6]4− is a coordination complex ion.
What is the hybridisation state of the central metal atom?
Name the type of isomerism shown by the following pair of coordination compounds:
[Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4
Select and write the correct alternative from the choices given below:
The coordination number and the oxidation state of the element ‘E’ in the complex [E(NH3)4Cl2]NO3 are respectively.
6 and 3
6 and 2
4 and 2
4 and 3
Solutions for 9: Coordination Compounds
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 9 - Coordination Compounds Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 9 - Coordination Compounds - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 9 - Coordination Compounds
Shaalaa.com has the CISCE Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE 9 (Coordination Compounds) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 9 Coordination Compounds are Overview of Coordination Compounds.
Using Nootan केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी solutions Coordination Compounds exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 9, Coordination Compounds केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी additional questions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.