Advertisements
Advertisements
प्रश्न
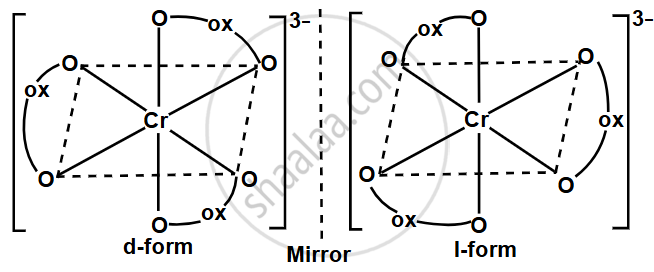
Write the optical isomers of [Cr(C2O4)3]3−.
दीर्घउत्तर
Advertisements
उत्तर
- The complex [Cr(C2O4)3]3− exhibits optical isomerism because it contains three bidentate oxalate ligands that coordinate to the central Cr3+ ion in an octahedral geometry.
- The chelation by the oxalate ligands creates an asymmetrical, chiral arrangement that leads to the formation of two non-superimposable mirror images, known as optical isomers.
- These isomers are designated as the Δ-isomer and the Λ-isomer, depending on the spatial arrangement of the ligands around the chromium ion.
- The Δ-isomer has a right-handed (clockwise) arrangement, while the Λ-isomer has a left-handed (anticlockwise) arrangement. Both isomers rotate plane-polarised light in opposite directions and are mirror images of each other, but they cannot be superimposed.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
