Advertisements
Advertisements
प्रश्न
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[CoF6]3−
Advertisements
उत्तर
Co(27) = [Ar] 3d74s2
Co3+ = [Ar] 3d64s0
F− being a weak field ligand cannot pair electrons.
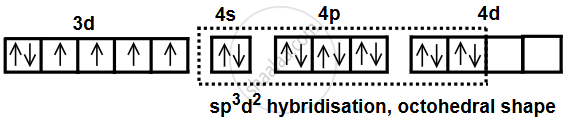
The oxidation state of cobalt in the complex is +3. Its electronic configuration is [Ar] 3d6. F− ion provides a weak ligand field and is unable to pair up the unpaired electrons of the 3d orbitals.
Hence, six equivalent hybrid orbitals are obtained by mixing of one 4s, three 4p and two 4d orbitals. Six empty sp3d2 hybrid orbitals are filled up by six electron pairs provided by F− ion. The highly paramagnetic nature of the complex further confirms the presence of four unpaired electrons.
As d-orbitals of the outermost shell participate in hybridisation, the complexes thus formed are called outer orbital complexes.
Hence, [CoF6]3− is a paramagnetic and octahedral complex.
APPEARS IN
संबंधित प्रश्न
On the basis of valence bond theory explain the nature of bonding in [CoF6]3 ion.
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Fe(CN)6]4−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Co(C2O4)3]3−
Explain the geometry of `[Co(NH_3)_6]^(3+)` on the basis of hybridisation. (Z of Co = 27)
[NiCl4]2- is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why? (Atomic no. Ni = 28)
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[FeCl6]^{4-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Write the hybridization and shape of the following complexes:
[Ni(CN)4]2−
How many radial nodes for 3p orbital?
Which of the statement given below is incorrect about H2O2?
When the hybridization state of carbon changes from sp3 to sp2 and finally to sp, the angle between hybridized orbital will
Using valence bond theory, predict the hybridization and magnetic character of the following:
[CoF6]3– [Atomic number of Co = 27]
According to the valence bond theory, the hybridization of central metal atom is dsp2 for which one of the following compounds?
Write the hybridisation and magnetic behaviour of [CoF6]3−.
[Given: Atomic number of Co = 27]
The magnetic moment of [NiCl4]2− is ______.
[Atomic number: Ni = 28]
[Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.
[Atomic number: Ni = 28]
During chemistry class, a teacher wrote \[\ce{[Ni(CN)4]^2-}\] as a coordination complex ion on the board. The students were asked to find out the magnetic behaviour and shape of the complex. Pari, a student, wrote the answer paramagnetic and tetrahedral whereas another student Suhail wrote diamagnetic and square planer.
Evaluate Pari’s and Suhail’s responses.
Which of the following are paramagnetic?
- [NiCl4]2−
- Ni(CO)4
- [Ni(CN)4]2−
- [Ni(H2O)6]2+
- Ni(PPh3)4
Choose the correct answer from the options given below:
