Advertisements
Chapters
▶ 1: Solid State
2: Solutions
3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 1 - Solid State Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 1 - Solid State - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 1: Solid State
Below listed, you can find solutions for Chapter 1 of CISCE Nootan for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State REVIEW EXERCISES [Pages 21 - 37]
What do you understand by unit cell?
What parameters are used to describe a unit cell?
Which crystal system can possess all the four types of unit cell?
What is the number of atoms in a body-centred cubic (bcc) unit cell of an element?
Calculate the number of atoms in a cubic based unit cell having one atom at each corner and two atoms on each diagonal.
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
How many atoms of an element are there in one unit cell of simple cubic?
How many atoms of an element are there in one unit cell of body-centred cubic?
How many atoms constitute one unit cell of a face-centered cubic crystal?
The density of a face-centred cubic element (atomic mass = 60.2) is 6.25 g cm−1. Calculate the length of the edge of the unit cell.
Crystalline CsBr has a bcc structure. Calculate the unit cell edge length if the density of CsBr crystal is 4.24 g cm−3. (Atomic masses: Cs = 133; Br = 80)
An element has bcc structure with a cell edge of 288 pm. The density of the element is 7.2 g cm−3. How many atoms are present in 208 g of the element?
The density of KBr is 2.75 g cm−3. The length of the edge of the unit cell is 654 pm. Show that KBr has a face-centred cubic structure (NA = 6.022 × 1023 mol−1, Atomic masses : K = 39, Br = 80).
A solid is made up of two elements P and Q. Atoms Q are in hcp arrangement while atoms P occupy all the tetrahedral sites. What is the formula of the compound?
In an oxide of aluminium, oxide ions are arranged in ccp arranged and the aluminium ions occupy two third of the octahedral voids. What is the formula of the compound?
In a solid, oxide ions are arranged in hcp. One third of octahedral voids are occupied by the cations A and one sixth of the tetrahedral voids are occupied by the cations B. What is the formula of the compound?
In a closed packed structure of mixed oxides, the oxide ions are arranged in hcp array. One eighth of tetrahedral voids are occupied by divalent cations (A) while one half of octahedral voids are occupied by trivalent cations (B). What is the formula of the compound?
Why are molecular solids soft and possess low melting and boiling points?
What are the constituent particles in ionic solids and how are they held together?
Sort out the molecular, ionic and covalent solids from the following:
Naphthalene, ZnS, graphite, ice, Agl, silicon carbide.
What is the significance of radius ratio in deciding the structural arrangement in an ionic solid?
What is the coordination number of NaCl crystal structure?
Why are the properties of diamond very different from those of graphite although both are the different forms of the same substance?
Explain the lubricating property of graphite on the basis of its structure.
Describe the structure of graphite with the help of a labelled diagram.
Discuss the crystal structure of copper metal.
Why do metals possess lustre and conduct electric current?
Write a short note on isomorphism.
Write a short note on isopolymorphism.
Define point defects in solids.
What is Schottky defect in a solid?
What is the effect of the presence of Schottky defects on the density of a crystal?
What are F-centres?
Pure silicon does not conduct electricity. How can it be made a conductor of electricity? Explain with examples.
AgCl is doped with 10−2 mol% of CdCl2. Find the concentration of cation vacancies.
What other element may be added to silicon to make electrons available for conduction of an electric current?
What is the non-stoichiometry defect in crystals?
Why does Frenkel defect not change the density of AgCl crystals?
State the difference between Schottky and Frenkel defects. Which of these two changes the density of the solid?
Name a salt that can be added to AgCl so as to produce cation vacancies.
Which point defect lowers the density of a crystal?
Name a salt that can be added to AgCl so as to produce cation vacancies.
What makes alkali metal halides sometimes coloured, which are otherwise colourless?
Why does table salt, NaCl, sometimes appear yellow in colour?
What is a semiconductor?
Describe the two main types of semiconductors and contrast their conduction mechanism.
What type of crystal defect is produced when sodium chloride is doped with MgCl2?
Name the electrical property possessed by the following oxide:
TiO
Name the electrical property possessed by the following oxide:
Ti2O3
Name the electrical property possessed by the following oxide:
TiO2
Name the electrical property possessed by the following oxide:
CrO2
Name the electrical property possessed by the following oxide:
Cr2O3
Sort out the diamagnetic, paramagnetic and ferromagnetic substances among the following:
O2, Fe, TiO2, Cu2+, Fe3+, Ni, CrO2, C6H6, NaCl.
What are paramagnetic substances?
What are ferromagnetic substances?
Account for the paramagnetic character of transition metal compounds.
How does the paramagnetic character of the bivalent ions of first transition metal series vary from titanium (Z = 22) to copper (Z = 29)?
How does the electrical resistivity of the following class of material vary with temperature?
Semiconductor
How does the electrical resistivity of the following class of material vary with temperature?
Metallic conductor
How does the electrical resistivity of the following class of material vary with temperature?
Superconductor
Name any two superconducting materials which show superconductivity above 100 K.
Compare dia, para and ferromagnetism.
Explain the dielectric properties of solids.
Distinguish between crystalline solid and amorphous solid.
How would you convert a crystalline solid into an amorphous solid?
What is the difference between ferromagnetic and ferrimagnetic substances?
What is the utility of piezoelectric crystals?
What happens when ferrimagnetic Fe3O4 is heated at 850 K and why?
What are amorphous solids?
Discuss the properties of amorphous solids.
Discuss the uses of amorphous solids.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State VERY SHORT ANSWER TYPE QUESTIONS [Pages 42 - 43]
What type of solids do possess a long range order?
Name different types of crystalline solids.
Define the term space lattice.
Define lattice points.
What do you understand by unit cell?
How many atoms of an element are there in one unit cell of simple cubic?
What do you understand by a face-centred unit cell?
What is a body-centred cubic system?
How many atoms constitute one unit cell of a face-centered cubic crystal?
What is the percentage of the occupied space in a simple cubic unit cell?
What is the name of the unit cell in which the atoms are present at the corners of all 12 edges of a cube in a unit cell?
What is the number of atoms in a body-centred cubic (bcc) unit cell of an element?
What is the edge length of a face-centred cubic unit cell if the radius of the atom is r?
What is the coordination number in a two dimensional hexagonal close packing?
What is a void?
What is the coordination number of atoms in a cubic close-packed structure?
What is the coordination number in a close-packed structure (hcp)?
What is the coordination number of atoms in a bcc structure?
Define a tetrahedral void.
If a close-packed structure contains n particles, what would be the number of tetrahedral voids in the structure?
If a close-packed structure contains n particles, what would be the number of octahedral voids in the structure?
If the radius of a particle is R, what would be the radius of a tetrahedral void present in the close-packed structure of the given particles?
What is the ratio of the radius of an octahedral void to that of the constituent particle?
In an ionic crystal, the radius ratio is greater than 0.732. What is the coordination number of the crystal?
In the crystal of zinc sulphide, zinc occupies tetrahedral voids. What is the coordination number of zinc?
What type of close-packing is possessed by a crystal of NaCl?
Name the ions forming the close-packed structure in NaCl crystals.
Name the ions present in voids in NaCl crystals.
What is Schottky defect in a solid?
What do you understand by the fact that CaF2 has 8 : 4 coordination?
What are electronic imperfections in crystals?
What type of ionic compounds show Schottky defects?
Write the effect on density of a substance in the Frenkel defect?
What are F-centres?
What do you understand by doping of a crystal?
What is a semiconductor?
How do paramagnetic substances behave in the absence of a magnetic field?
What happens when ferrimagnetic Fe3O4 is heated at 850 K and why?
What is pyroelectricity?
What is piezoelectricity?
What happens when an electric field is applied to a piezoelectric crystal?
What do you understand by superconductivity?
What happens when amorphous solids are subjected to annealing?
What type of order is present in amorphous solids?
Give reason for the following:
Ionic compounds do not conduct electricity in solid state.
What is the effect of increasing temperature on the conductivity of a semiconductor?
How does amorphous silica differ from quartz?
What is a void?
Compare dia, para and ferromagnetism.
In a close packing of spheres, what is the value of radius ratio (radius of an octahedral void/radius of sphere)?
How can you convert NaCl structure into CsCl structure?
How can you convert CsCl structure into NaCl structure?
What causes the conduction of electricity by semiconductors?
What type of crystals exhibit piezoelectricity?
Agl crystallises in cubic close-packed ZnS structure. What fraction of tetrahedral sites are occupied by Ag+ ions?
Classify each of the following solids as ionic, metallic, molecular, covalent or amorphous.
- P4O10
- Graphite
- Brass
- (NH4)3PO4
- SiC
- Rb
- I2
- LiBr
- P4
- Si
- Plastic
Classify each of the following as being either a p-type or a n-type semiconductor:
- Ge doped with In
- B doped with Si
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State SHORT ANSWER TYPE QUESTIONS [Pages 43 - 44]
What are the main characteristics of crystalline solids?
What do you understand by space lattice?
What do you understand by unit cell?
What do you understand by unit cell?
What parameters are used to describe a unit cell?
What is a primitive unit cell?
Name the different types of primitive unit cells.
How many types of unit cells are present in different types of crystals? Explain with appropriate figures.
Describe the various types of cubic systems.
Explain why uncharged atoms or molecules never crystallise in a simple cubic structure.
In a cubic unit cell, what is the contribution to the unit cell of the particle present at the corner?
In a cubic unit cell, what is the contribution to the unit cell of the particle present at the centre of a face?
In a cubic unit cell, what is the contribution to the unit cell of the particle present at the body centre?
How many atoms of an element are there in one unit cell of simple cubic?
What is the number of atoms in a body-centred cubic (bcc) unit cell of an element?
Find the number of atoms in the fcc unit cell.
What is the percentage of the occupied space in a simple cubic unit cell?
Show that the face diagonal of a cube is equal to `sqrt2 a`, where a is the edge length.
Show that the packing fraction in a face-centred cubic unit cell is 0.74.
In a face-centred cubic arrangement of atoms A and B, A are present at the corners and B at the face-centres. If A atoms are missing from 4 corners in each unit cell, what is the simplest formula of the compound?
An element crystallises separately both in hcp and ccp structures. Will the two structures have the same density? If yes, why?
Explain why the hexagonal close packing is more efficient than square close packing when considered in a plane?
Distinguish between hexagonal close packing and cubic close packing.
What is meant by the term ‘coordination number’?
Mention the coordination number of each sphere in a hexagonal close-packed structure.
Mention the coordination number of each sphere in a body-centred close-packed structure.
What do you understand by voids in close-packed structures?
Define a tetrahedral void.
Define octahedral void.
Show that the radius of a tetrahedral void is 0.225 times the radius of the sphere forming a close-packed structure.
Show that a particle of radius less than 0.441 times the radius of the constituent particle can only be placed in an octahedral void without disturbing the crystal lattice.
What do you understand by radius ratio?
What is the ideal radius ratio for octahedral arrangement?
An ionic solid possesses a body-centred close-packed structure. What is the ideal radius ratio and coordination number for the structure?
How would you decide that a given hard and rigid solid possessing high melting and boiling points is ionic or covalent?
What types of structures are possessed by ionic solids of the type AB? Explain with diagrams.
Explain the structure of NaCl.
Find the number of ion pairs per unit cell in NaCl.
What is the effect of pressure on the structures of the ionic crystals of the type AB?
What is the effect of temperature on the structures of the ionic crystals of the type AB?
What do you understand by imperfections in ionic crystals?
Name the type of imperfections which generally occur in ionic crystals.
Explain why does the presence of Frenkel defects not affect the density of a crystal.
How does the Schottky defect increase the conductivity of a crystal?
How does the Frenkel defect increase the conductivity of a crystal?
How does a crystal having metal excess defect maintain its electrical neutrality?
Why does a crystal of KCl appear violet in colour in spite of the fact that both K+ and Cl− ions are colourless?
What are F-centres?
What do you mean by doping?
Why does the impurity-doped silicon act as a semiconductor?
What is a semiconductor?
What factors are responsible for the conduction of current by a semiconductor?
Mention important applications of semiconductors.
What is ferromagnetism?
How does ferromagnetism arise?
Why do ferrimagnetic substances possess small net magnetic moments in spite of the presence of a large number of unpaired electrons?
What happens when a piezoelectric crystal is subjected to a mechanical stress?
What happens when an electric field is applied to a piezoelectric crystal?
Why are the amorphous solids said to possess short range order?
Discuss the properties of amorphous solids.
What type of crystal defect is produced when sodium chloride is doped with MgCl2?
Explain the Schottky defect.
What are the consequences of the Schottky defect in crystals?
What are the consequences of Frenkel defects in crystals?
If the radius of the octahedral void is r and the radius of the atoms in close packing is R, derive the relation between r and R.
Stability of a crystal is reflected in the magnitude of its melting point. Comment.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State LONG ANSWER TYPE QUESTIONS [Pages 44 - 45]
How are the solids classified?
Compare the properties of crystalline and amorphous solids.
Explain the characteristics of different types of crystalline solids.
Discuss the characteristics of various types of unit cells.
How would you calculate the number of particles in a unit cell?
How many atoms of an element are there in one unit cell of simple cubic?
What is the number of atoms in a body-centred cubic (bcc) unit cell of an element?
Find the number of atoms in the fcc unit cell.
Calculate the percentage efficiency of packing in the case of a body-centred cubic crystal.
Calculate the percentage of the occupied space in a face-centred cubic unit cell.
Show that the packing fraction in a simple cubic unit cell is 0.524 and the radius of the atom is equal to half of the edge length.
The unit cell of the crystal of an element has Z atoms and the edge length equal to a. If the molecular mass of the element is M, calculate the density of the crystal.
Explain with a diagram the hcp mode of packing in crystals.
Explain with diagrams the ccp mode of packing in crystals.
Distinguish between hexagonal close packing and cubic close packing.
What are the voids commonly found in close-packed structures?
Show that the radius of a tetrahedral void is 0.225 times the radius of the sphere forming a close-packed structure.
Calculate the radii of octahedral voids in terms of the radius of the spheres forming close-packed structures.
What do you understand by radius ratio?
What is the significance of the radius ratio in determining the structural arrangement and coordination number of an ionic solid?
Explain the structure of NaCl.
Show that a unit cell of sodium chloride contains four Na+Cl− units.
Explain the Schottky defect.
Explain with a diagram the Frenkel defect.
How do Schottky defects affect the properties of crystals?
How do Frenkel defects affect the properties of crystals?
Describe the various types of non-stoichiometric defects found in ionic crystals.
What do you understand by doping of a crystal?
How are the impurity defects introduced into covalent solids?
How are the impurity defects introduced into ionic solids?
In what way is the electrical conductivity of a crystal affected by doping?
Classify the solids on the basis of their electrical properties.
Describe the various types of magnetic substances with the help of electron spin and explain their characteristics.
What is piezoelectricity?
Why are the piezoelectric crystals used in record players?
Write a short note on pyroelectricity.
Write a short note on ferroelectricity.
Write a short note on antiferroelectricity.
Explain why all ferroelectric crystals behave as piezoelectric crystals but the reverse is not true.
Explain why there is no current in a metal although there are free electrons in motion.
Explain why doping produces cation vacancies and not the anion vacancies.
What are amorphous solids?
Discuss the properties of amorphous solids.
Discuss the uses of amorphous solids.
How would you convert silica into a glassy substance? Explain the structure of the substance thus obtained.
Why do the glass objects from ancient civilizations look milky instead of being transparent?
Explain the following with suitable examples:
Ferromagnetism
Explain the following with suitable examples:
Paramagnetism
Explain the following with suitable examples:
Ferrimagnetism
Explain the following with suitable examples:
Piezoelectric effect
Explain the following with suitable examples:
12-16 group compounds
Explain the following with suitable examples:
13-15 group compounds
Explain the Schottky defect.
Explain with a diagram the Frenkel defect.
Explain the following term with a suitable example:
Interstitials
Explain the following term with a suitable example:
F-centres
What makes a glass different from a solid such as quartz? Under what conditions could quartz be converted into glass?
What is a semiconductor?
Describe the two main types of semiconductors and mention their important applications.
How are the crystalline solids classified?
Describe the important characteristics of crystalline solids.
What are molecular solids?
Explain the characteristics of different types of crystalline solids.
Discuss the crystal structure of dry ice.
Give any three characteristics of ionic crystals.
Explain the structure of NaCl.
What is meant by covalent solids?
Discuss the structures of diamond. How would you explain the properties of these substances on the basis of their structures?
Describe the structure of graphite with the help of a labelled diagram.
What are metallic solids?
What are the characteristics of metallic solids?
Describe the important features of metallic bonding.
Discuss the crystal structure of copper metal.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State INTEGER TYPE QUESTIONS [Page 45]
The distance between Na+ and Cl− ions in solid NaCl of density 43.1 g cm−3 is ______ × 10 −10 m (Nearest integer).
(Given: NA = 6.02 × 1023 mol−1)
Atoms of element X form hcp lattice and those of element Y occupy `2/3` of its tetrahedral voids. The percentage of element X in the lattice is ______ (Nearest integer).
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 46 - 47]
Choose the correct option in the following questions.
Which one is not the property of a crystalline solid?
Isotropic
Sharp melting point
A definite and regular geometry
High intermolecular forces
Tetragonal crystal system has the following unit cell dimensions:
a = b = c, α = β = γ = 90°
a = b ≠ c, α = β = γ = 90°
a ≠ b ≠ c, α = β = γ = 90°
a = b ≠ c, α = β = γ = 90°, γ = 120°
The coordination number of each sphere in hcp arrangement is ______.
8
12
6
4
The number of atoms contained in one face-centred cubic unit cell of a monoatomic substance is ______.
1
2
4
3
How many chloride ions are surrounding sodium ions in sodium chloride crystal?
4
8
6
12
3
For an ionic crystal of general formula AX and coordination number 6, the value of radius ratio will be ______.
greater than 0.73
in between 0.73 and 0.41
in between 0.41 and 0.22
less than 0.22
Potassium crystallises in bcc lattice, the coordination number of potassium in potassium metal is ______.
12
4
6
8
Coordination number of Cu is ______.
4
6
8
12
An ionic compound is made up of P cations and Q anions. If P are present at alternate corners and Q is present on the body of the diagonal, then the formula of the ionic compound will be ______.
PQ
PQ2
P2Q
P4Q
In a crystal, the atoms are located at the position of ______.
maximum P.E.
minimum P.E.
zero P.E.
infinite P.E.
An element A (atomic mass 60) has simple cubic lattice of edge 100 pm. The density of crystal (N0 = 6 × 1023) is ______.
600 g cm−3
1 × 104 g cm−3
6 × 10−2 g cm−3
1 × 102 g cm−3
The intermetallic compound LiAg crystallises in cubic lattice in which both lithium and silver have coordination number of eight. The crystal class is ______.
simple cubic
body-centred cubic
face-centred cubic
none of these
A solid has a structure in which W atoms are located at the corners of a cubic lattice, O atoms at the centre of edges and Na atoms at the centre of the cube. The formula of the compound is ______.
NaWO2
NaWO3
Na2WO3
NaWO4
For tetrahedral coordination, the radius ratio (r+/r−) should be ______.
0.155−0.225
0.225−0.414
0.444−0.732
0.732−1
If edge of a bcc crystal of an element is a cm, M is the atomic mass and N0 the Avogadro’s number, the density of the crystal is ______.
`(4M)/(N_0a^3)`
`(2N_0)/(Ma^3)`
`(2M)/(N_0a^3)`
`(Ma^3)/(2N_0)`
In a compound AB2O4, oxide ions are arranged in ccp and cations A are present in octahedral voids. Cations B are equally distributed between octahedral and tetrahedral voids. The fraction of the octahedral voids occupied is ______.
`1/2`
`1/4`
`1/8`
`1/6`
Potassium has a bcc structure with nearest neighbour distance of 4.52 Å. Its atomic weight is 39. Its density will be ______.
454 kg m−3
804 kg m−3
852 kg m−3
908 kg m−3
A compound formed by atoms X and Y crystallises in the cubic structure. The atoms X are present at the corners of a cube while atoms Y are at the face-centres. The formula of the compound is ______.
XY
X2Y
X3Y
XY3
In a face-centred cubic system, the distance d between the nearest neighbours is given by ______.
d = a
`d = sqrt 2a`
`d = (sqrt 3a)/2`
`d = a/sqrt2`
An element crystallises in body-centred cubic structure. If the edge length of the cubic unit cell is 400 pm, the interatomic distance in the crystal is ______.
346.4 pm
282.8 pm
400 pm
200 pm
A face-centred cubic element (atomic mass = 60) has a cell edge of 400 pm. What is its density?
0.623 g cm−3
6.23 g cm−3
62.3 g cm−3
0.0623 g cm−3
An element occurs in bcc structure. Its density is 8.0 g cm−3. If the cell edge is 250 pm, the atomic mass of the element is ______.
26.4 g mol−1
37.6 g mol−1
54.5 g mol−1
86.1 g mol−1
Which of the following statements is not true?
The radius of a tetrahedral void is 0.225 times the radius of the particle.
The radius of an octahedral void is 0.732 times the radius of the particle.
The space occupied in bcc arrangement is 68%.
The number of tetrahedral voids in a close-packed structure is double that of the number of constituent particles.
Gold crystallises in a ccp lattice. How many nearest neighbours does a gold atom possess?
6
8
10
12
Which of the following statements is not true for rock salt (NaCl) structure?
The structure is of fcc type.
Cl− ions are present at the corners and face-centres of a cube.
Na+ ions are present in alternate tetrahedral voids.
The structure has 6 : 6 coordination.
Which of the following substances is ferromagnetic?
Fe
Ni
MnO
Fe3O4
The point defect which lowers the density of a crystal is ______.
Schottky defect
Frenkel defect
Schottky defect and Frenkel defect
None of these
The presence of F-centres in a crystal makes it ______.
conducting
colourless
non-conducting
coloured
When a group 13 element is added in small amounts to Ge, the material obtained is ______.
an insulator
an n-type semiconductor
a p-type semiconductor
a super conductor
Which of the following sets of magnetic moments represents an antiferromagnetic substance?
↑↑↑↑↑↑
↑↓↑↓↑↓
↑↑↓↑↑↓
↑↑↓↓↑
Which of the following statements is not true?
Piezoelectric crystals produce electricity when subjected to a mechanical stress.
The electric current produced on heating certain polar crystals is called ferroelectricity.
PbZrO3 is an antiferroelectric substance.
Piezoelectric crystals are used as pickups in record players.
In a solid lattice, the cation has left a lattice site and is located at an interstitial position. The lattice defect is ______.
Frenkel defect
Schottky defect
Non-stoichiometric defect
Valence defect
CsBr contains bcc structure. The length of its one side is 4.3 Å. The minimum distance between Cs+ and Br− ions will be ______.
0.897 Å
3.72 Å
1.794 Å
none of these
Germanium is an example of ______.
an intrinsic semiconductor
an extrinsic semiconductor
an insulator
an n-type semiconductor
Which type of semiconductor is obtained on mixing arsenic into silicon?
n-type
p-type
p-n type
n-p type
How many unit cells are present in a cube-shaped ideal crystal of NaCl of mass 1.00 g? [Atomic masses: Na = 23, Cl = 35.5]
5.14 × 1021 unit cells
1.28 × 1021 unit cells
1.71 × 1021 unit cells
2.57 × 1021 unit cells
What type of crystal defect is indicated in the diagram below?
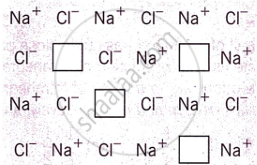
Frenkel defect
Schottky defect
Interstitial defect
Frenkel and Schottky defects
An ionic compound has a unit cell consisting of A ions at the corners of a cube and B ions on the centres of the faces of the cube. The empirical formula for this compound would be ______.
A3B
AB3
A2B
AB
Total volume of atoms present in a face-centred cubic unit cell of a metal (r is atomic radius) is ______.
`20/3 pi r^3`
`24/3 pi r^3`
`12/3 pi r^3`
`16/3 pi r^3`
In a compound, atoms of element Y form ccp lattice and those of element X occupy `2/3`rd of tetrahedral voids. The formula of the compound will be ______.
X4Y3
X2Y3
X2Y
X3Y4
AB crystallises in a body-centred cubic lattice with edge length ‘a’ equal to 387 pm. The distance between two oppositively charged ions in the lattice is ______.
335 pm
250 pm
200 pm
300 pm
A metal crystallises with a face-centred cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal atom is ______.
288 pm
408 pm
144 pm
204 pm
The number of octahedral void(s) per atom present in a cubic close-packed structure is ______.
1
3
2
4
If ‘a’ is the length of the side of the cube, the distance between the body-centered atom and one corner atom in the cube will be ______.
`(2/sqrt 3) a`
`(4/sqrt 3) a`
`(sqrt 3/4) a`
`(sqrt 3/2) a`
Sodium metal crystallises in a body-centred cubic lattice with a unit cell edge of 4.29 Å. The radius of the sodium atom is approximately ______.
5.72 Å
0.93 Å
1.86 Å
3.22 Å
If the unit cell of a mineral has cubic close packed (ccp) array of oxygen atoms with m fraction of octahedral holes occupied by aluminium ions and n fraction of tetrahedral holes occupied by magnesium ions, m and n, respectively, are ______.
`1/2, 1/8`
`1, 1/4`
`1/2, 1/2`
`1/4, 1/8`
Lithium has a bcc structure. Its density is 530 kg m−3 and its atomic mass is 6.94 g mol−1. Calculate the edge length of a unit cell of lithium metal. (NA = 6.02 × 1023 mol−1)
154 pm
352 pm
527 pm
264 pm
The ionic radii of A+ and B− are 0.98 × 10−10 m and 1.81 × 10−10 m. The coordination number of each ion in AB is ______.
8
2
6
4
Which is the incorrect statement?
FeO0.98 has non stoichiometric metal deficiency defect.
Density decreases in case of crystals with Schottky’s defect.
NaCl (s) is insulator, silicon is semiconductor, silver is conductor, quartz is piezoelectric crystal.
Frenkel defect is favoured in those ionic compounds in which sizes of cations and anions are almost equal.
A metal crystallises in a face-centred cubic structure. If the edge length of its unit cell is ‘a’, the closest approach between two atoms in metallic crystal will be ______.
`sqrt 2 a`
`a/sqrt 2`
2a
`2 sqrt 2 a`
Which type of ‘defect’ has the presence of cations in the interstitial sites?
Schottky defect
Vacancy defect
Frenkel defect
Metal deficiency defect
Iron exhibits bcc structure at room temperature. Above 900°C, it transforms to fcc structure. The ratio of density of iron at room temperature to that at 900°C (assuming molar mass and atomic radii of iron remains constant with temperature) is ______.
`(3 sqrt 3)/(4 sqrt 2)`
`(4 sqrt 3)/(3 sqrt 2)`
`sqrt 3/sqrt 2`
`1/2`
An element has a face-centred cubic (fcc) structure with a cell edge of a. The distance between the centres of two nearest tetrahedral voids in the lattice is ______.
a
`3/2 a`
`a/2`
`sqrt 2 a`
A compound is formed by cation C and anion A. The anions form hexagonal close-packed (hcp) lattice and the cations occupy 75% of octahedral voids. The formula of the compound is ______.
C2A3
C3A2
C3A4
C4A3
An element has a body centered cubic (bcc) structure with a cell edge of 288 pm. The atomic radius is ______.
`sqrt 3/4 xx 288 "pm"`
`sqrt 2/4 xx 288 "pm"`
`4/sqrt3 xx 288 "pm"`
`4/sqrt 2 xx 288 "pm"`
A diatomic molecule X2 has a body-centered cubic (bcc) structure with a cell edge of 300 pm. The density of the molecule is 6.47 g cm−3. The number of molecules present in 200 g of X2 is ______.
4 NA
2 NA
40 NA
8 NA
The right option for the number of tetrahedral and octahedral voids in the hexagonal primitive unit cell is ______.
6, 12
2, 1
12, 6
8, 4
The correct option for the number of body-centered unit cells in all 14 types of Bravais lattice unit cells is ______.
3
7
5
2
In a binary compound, atoms of element A form a hcp structure and those of element M occupy `2/3` of the tetrahedral voids of 3 the hcp structure. The formula of the binary compound is ______.
M2A3
M4A3
M4A
MA4
For the given close packed structure of a salt made of cation X and anion Y shown below (ions of only one face are shown for clarity), the packing fraction is approximately:
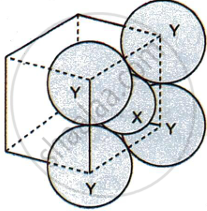
0.74
0.63
0.52
0.48
Copper crystallises in fcc unit cell with cell edge length of 3.608 × 10−8 cm. The density of copper is 8.92 g cm−3. Calculate the atomic mass of copper.
31.55 u
60 u
65 u
63.1 u
Choose the correct statement:
Diamond is covalent and graphite is ionic.
Diamond is sp3 hybridised and graphite is sp2 hybridised.
Both diamond and graphite are used as dry lubricants.
Diamond and graphite have two dimensional network.
The incorrect statement about the imperfections in solids is ______.
Schottky defect decreases the density of the substance.
Interstitial defect increases the density of the substance.
Frenkel defect does not alter the density of the substance.
Vacancy defect increases the density of the substance.
Atom X occupies the fcc lattice sites as well as alternate tetrahedral voids of the same lattice. The packing efficiency (in %) of the resultant solid is closest to ______.
25
35
55
75
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State FILL IN THE BLANKS TYPE QUESTIONS [Pages 49 - 50]
If a molten crystalline solid is cooled, the original ______ reappears again.
Amorphous solids are regarded as ______ liquids.
The smallest three dimensional group of lattice points, which when repeated in three dimensions in space gives the whole lattice of the crystal, is called ______.
The unit cell in which one constituent particle is present at the centre in addition to the particles at its corners is called ______ unit cell.
The unit cell of a body-centred cubic system contains ______ atoms.
Both ccp and hcp are ______ packings and occupy about ______% of the available space.
The number of nearest neighbours with which a given particle is in direct contact in a crystal is called the ______.
In a close-packed structure, the number of tetrahedral voids is ______ the number of constituent particles because every void has ______ particles and there are ______ voids around each particle.
A cation would exactly fit into an octahedral void in the lattice formed by anions if `r_+/r_-` is exactly equal to ______.
In the sodium chloride structure, each Na+ ion is surrounded by six Cl− ions nearest neighbours and ______ Na+ ions next nearest neighbours.
If the radius ratio `(r_+/r_-)` is in the range 0.414 − 0.732, the compound AB is likely to have ______ structure.
The percentages of unoccupied spaces in bcc and fcc arrangements are ______ and ______ respectively.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State ASSERTION-REASON TYPE QUESTIONS [Page 50]
The questions given below consist of an Assertion and a Reason. You have to choose the correct answer (a), (b), (c) or (d) according to the following clue:
Assertion: The presence of Frenkel defect in a crystal lowers its density.
Reason: Frenkel defect involves the creation of a hole in the lattice of a crystal due to migration of a cation from its lattice site to an interstitial site.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Silicon forms covalent crystals.
Reason: Its lattice consists of silicon atoms bonded together by covalent bonds in a three dimensional network.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The unit cell of a face-centred cubic system contains 4 atoms.
Reason: The unit cell of a face-centred cubic system consists of atoms at face centres in addition to atoms at the corners.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: In rock salt structure, Cl− ions form a close-packed lattice and Na+ ions occupy the octahedral voids.
Reason: Sodium chloride possesses a face-centred cubic arrangement.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: KCl crystals appear violet in colour.
Reason: The violet colour is due to electrons trapped in anion vacancies.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: In a particular point defect, an ionic solid is electrically neutral, even if few of its cations are missing from its unit cells.
Reason: In an ionic solid, Frenkel defect arises due to dislocation of cation from its lattice site to interstitial site maintaining overall electrical neutrality.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State NUMERICAL PROBLEMS [Pages 50 - 51]
A unit cell of sodium chloride has four formula units. The edge length of the unit cell is 0.564 nm. What is the density of sodium chloride?
Chromium metal crystallises with a body-centred cubic lattice. The length of the unit cell edge is found to be 287 pm. Calculate the atomic radius. What would be the density of chromium in g cm−3?
Iron occurs as body-centred as well as face-centred cubic systems. If the effective radius of an atom of iron is 124 pm, calculate the density of iron in both the structures.
Molybdenum forms body-centred cubic crystals and at 20°C the density is 10.3 g/cm3. Calculate the distance between the centres of the nearest molybdenum atoms.
In the cubic crystal of CsCl (d = 3.97 g/cm3), the eight corners are occupied by Cl− with a Cs+ at the centre and vice-versa. Calculate the distance between the neighbouring Cs+ and Cl− ions. What is the radius ratio of the two ions? (At. masses: Cs = 132.91, Cl = 35.45)
A compound formed by atoms A and B crystallises in the cubic structures. The atoms A occupy the corners while atoms Y the face-centres of the cube. Find the formula of the compound. If the side length is 5 Å, calculate the density of the solid. (Atomic masses: A = 60, B = 90.)
A solid AB crystallises as a body-centred cubic lattice with the unit cell edge length equal to 384 pm. Calculate
- the distance between the oppositely charged ions in the lattice, and
- the radius of A+ ion if that of B− is 180 pm.
The density of crystalline sodium chloride is 2.165 g cm−3. What is the edge length of the cubic unit cell? What would be the dimension of a cube containing one mole of NaCl?
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State 'NCERT TEXT-BOOK' Exercises [Pages 52 - 55]
Define the term ‘amorphous’.
Give a few examples of amorphous solids.
What makes a glass different from a solid such as quartz? Under what conditions could quartz be converted into glass?
Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous.
- Tetra phosphorus decoxide (P4O10)
- Ammonium phosphate (NH4)3PO4
- SiC
- I2
- P4
- Plastic
- Graphite
- Brass
- Rb
- LiBr
- Si
What is meant by the term ‘coordination number’?
What is the coordination number of atoms in a cubic close-packed structure?
What is the coordination number of atoms in a bcc structure?
How can you determine the atomic mass of an unknown metal if you know its density and the dimension of its unit cell? Explain.
‘Stability of a crystal is reflected in the magnitude of its melting points.’ Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules?
Distinguish between hexagonal close packing and cubic close packing.
How will you distinguish between the following pair of terms?
Crystal lattice and unit cell
How will you distinguish between the following pair of terms?
Tetrahedral and octahedral voids
How many lattice points are there in one unit cell of the following lattice?
Face-centred cubic
How many lattice points are there in one unit cell of the following lattice?
Face-centred tetragonal
How many lattice points are there in one unit cell of the following lattice?
Body-centred
Explain the basis of similarities between metallic and ionic crystals.
Explain the basis of differences between metallic and ionic crystals.
Explain ionic solids are hard and brittle.
What is the percentage of the occupied space in a simple cubic unit cell?
Calculate the percentage efficiency of packing in the case of a body-centred cubic crystal.
Calculate the percentage of the occupied space in a face-centred cubic unit cell.
Silver crystallises in fcc lattice. If edge length of the cell is 4.07 × 10−8 cm and density is 10.5 g cm−3, calculate the atomic mass of silver.
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
Niobium crystallises in body-centred cubic structure. If density is 8.55 g cm−3, calculate atomic radius of niobium using its atomic mass 93 u.
If the radius of the octahedral void is r and the radius of the atoms in close packing is R, derive the relation between r and R.
Copper crystallises into a fcc lattice with edge length 3.61 × 10−8 cm. Show that the calculated density is in agreement with its measured value of 8.92 g cm−3.
Analysis shows that nickel oxide has the formula Ni0.98O1.00. What fractions of nickel exist as Ni2+ and Ni3+ ions?
What is a semiconductor?
Describe the two main types of semiconductors and contrast their conduction mechanism.
Non-stoichiometric cuprous oxide, Cu2O can be prepared in laboratory. In this oxide, copper to oxygen ratio is slightly less than 2 : 1. Can you account for the fact that this substance is a p-type semiconductor?
Ferric oxide crystallises in a hexagonal close-packed array of oxide ions with two out of every three octahedral holes occupied by ferric ions. Derive the formula of the ferric oxide.
Classify each of the following as being either a p-type or a n-type semiconductor:
- Ge doped with In
- B doped with Si
Gold (atomic radius = 0.144 nm) crystallises in a face-centred unit cell. What is the length of a side of the cell?
In terms of band theory, what is the difference between a conductor and an insulator?
In terms of band theory, what is the difference between a conductor and a semiconductor?
Explain the Schottky defect.
Explain the following term with suitable examples:
Frenkel defect
Explain the following term with a suitable example:
Interstitials
Explain the following term with a suitable example:
F-centres
Aluminium crystallises in a cubic close-packed structure. Its metallic radius is 125 pm.
- What is the length of the side of the unit cell?
- How many unit cells are there in 1.00 cm3 of aluminium?
If NaCl is doped with 10−3 mol % of SrCl2, what is the concentration of cation vacancies?
Explain the following with suitable examples:
Ferromagnetism
Explain the following with suitable examples:
Paramagnetism
Explain the following with suitable examples:
Ferrimagnetism
Explain the following with suitable examples:
Antiferromagnetism
Explain the following with suitable examples:
12-16 group compounds
Explain the following with suitable examples:
13-15 group compounds
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 1 Solid State QUESTIONS FROM ISC EXAMINATION PAPERS [Pages 55 - 57]
Distinguish between crystalline solid and amorphous solid.
State the element present at the lattice sites in diamond.
The number of nearest neighbours for each atom in diamond.
The type of unit cell of diamond.
For sodium chloride crystal, state the type of unit cell.
For sodium chloride crystal, state the nature of the forces holding the particles together.
For sodium chloride crystal, state the number of nearest neighbours around each sodium ion.
For sodium chloride crystal, state the geometry of the sodium ions which are arranged around a chloride ion.
Diamond is hard and a bad conductor of electricity, while graphite is soft and a good conductor of electricity. Explain.
What is the state of hybridisation of carbon in diamond?
What is the state of hybridisation of carbon in graphite?
Describe the unit cell of sodium chloride with a neat diagram stating:
- Type of bonding
- Type of unit cell
- Number of nearest neighbours around sodium and chloride.
Fill in the blanks.
Crystals of ______ and ______ have face-centred cubic lattices.
Give any three characteristics of ionic crystals.
State the main characteristics of a network type of crystal citing a suitable example.
Name the crystal structure of the copper metal.
What is the coordination number of copper in its crystalline state?
How many sodium ions and chloride ions are present in a unit cell of sodium chloride crystal?
What is the coordination number of sodium and chloride ions in sodium chloride crystals?
State the element present at the lattice sites in diamond.
The number of nearest neighbours for each atom in diamond.
The type of unit cell of diamond.
What is the state of hybridisation of carbon in diamond?
What is the number of atoms in a body-centred cubic (bcc) unit cell of an element?
Give reasons:
Graphite is used as a lubricant.
Define piezoelectricity.
Give one use of piezoelectric crystals.
What is a semiconductor?
What is the effect of increasing temperature on the conductivity of a semiconductor?
A compound AB has a simple cubic structure and has a molecular mass of 99. Its density is 3.4 g cm−3. What will be the edge length of the unit cell?
What are F-centres?
Why are crystals having F-centres paramagnetic?
What is the state of hybridisation of carbon in diamond?
For a crystal of diamond, state the coordination number of each carbon atom.
State the element present at the lattice sites in diamond.
For a crystal of diamond, state the number of carbon atoms present per unit cell.
An ionic compound is made up of A cations and B anions. If A cations are present at the alternate corners and B anion is present on the body of the diagonal, what is the formula of the ionic compound?
Define the Frenkel defect in a solid crystal.
Explain giving reasons, why ionic solids conduct electricity in the molten state, but not in solid-state?
In a crystal of diamond:
- How many carbon atoms are present per unit cell?
- What type of lattice does diamond crystallise in?
- How many carbon atoms surround each carbon atom?
- How are they arranged?
Name the crystal structure of the copper metal.
Chromium metal crystallises with a body centred cubic lattice. The edge length of the unit cell is found to be 287 pm. Calculate the atomic radius. What would be the density of chromium in g/cm3? (atomic mass of Cr = 52.99)
Why does sodium chloride on heating with sodium vapours acquire yellow colour?
For a crystal of sodium chloride, state the type of lattice in which it crystallises.
What is the coordination number of sodium and chloride ions in sodium chloride crystals?
How many sodium ions and chloride ions are present in a unit cell of sodium chloride crystal?
For a crystal of sodium chloride, state the structural arrangement of the sodium chloride crystal.
The edge length of the unit cell of a body-centred cubic (bcc) crystal is 352 pm. Calculate the radius of the atom.
Graphite is anisotropic with respect to the conduction of electric current. Explain.
How many lattice points are there in one unit cell of the following lattice?
Body-centred
How many atoms constitute one unit cell of a face-centered cubic crystal?
A compound AB has a simple cubic structure and has a molecular mass of 99. Its density is 3.4 g cm−3. What will be the edge length of the unit cell?
In a face-centred cubic lattice, atom (A) occupies the corner positions and atom (B) occupies the face centre positions. If one atom of (B) is missing from one of the face-centred points, the formula of the compound is ______.
A2B5
A2B3
AB2
A2B
How many sodium ions and chloride ions are present in a unit cell of sodium chloride crystal?
Frenkel defect does not change the density of the ionic crystal whereas Schottky defect lowers the density of ionic crystal. Give a reason.
Lead sulphide has a face-centred cubic crystal structure. If the edge length of the unit cell of lead sulphide is 495 pm, calculate the density of the crystal.
(Atomic weight of Pb = 207, S = 32)
Examine the defective crystal given below and answer the question that follows:
| A+ | B− | A+ | B− | A+ |
| B− | B− | A+ | B− | |
| A+ | B− | A+ | A+ | |
| B− | A+ | B− | A+ | B− |
State if the above defect is stoichiometric or non-stoichiometric. How does this defect affect the density of the crystal? Also, write the term used for this type of defect.
An element occurs in a body-centred cubic structure. Its density is 8.0 g/cm3. If the cell edge is 250 pm, calculate the atomic mass of an atom of this element. (NA = 6.023 × 1023)
The packing efficiency of simple cubic structure, body centred cubic structure and face-centred cubic structure respectively is ______.
52.4%, 74%, 68%
74%, 68%, 52.4%
52.4%, 68%, 74%
68%, 74%, 52.4%
An element has atomic mass 93 g mol−1 and density 11.5 g cm–3. If the edge length of its unit cell is 300 pm, identify the type of unit cell. (NA = 6.023 × 1023 mol−1)
Calculate the radius of copper atom. The atomic weight of copper is 63.55 g mol−1. It crystallises in face-centred cubic lattice and has density of 8.93 g cm−3 at 298 K.
(NA = 6.023 × 1023 mol−1)
An alloy of gold (Au) and cadmium (Cd) crystallises with a cubic structure in which gold atoms occupy the corners and cadmium atoms fit into the face centres. What is the formula of this alloy?
Select and write the correct alternative from the choices given below
How many chloride ions are surrounding sodium ions in sodium chloride crystal?
4
8
6
12
3
The ratio of number of atoms present in a simple cubic, body-centred cubic and face-centred cubic structure are, respectively ______.
8 : 1 : 6
1 : 2 : 4
4 : 2 : 1
4 : 2 : 3
A solid has a structure in which ‘Y’ atoms are located at the corners of a cubic lattice, ‘O’ atoms at the centre of edges and ‘K’ atoms at the centre of the cube. What is the formula of the compound?
KYO2
KYO3
K2YO3
K4YO3
The appearance of colour in solid alkali metal halides is generally due to ______.
Schottky defect
Frenkel defect
Interstitial positions
F-centre
An element ‘X’ having atomic mass 60 has density 6.23 g cm−3. The edge length of its unit cubic cell is 400 pm. (NA = 6.02 × 1023 mol−1).
What is the type of unit cell known as?
Body centred cubic
Face centred cubic
Simple cubic
Side centred cubic
An element ‘X’ having atomic mass 60 has density 6.23 g cm−3. The edge length of its unit cubic cell is 400 pm. (NA = 6.02 × 1023 mol−1).
What is the radius of an atom of this element?
210.5 pm
344.4 pm
141.4 pm
115.3 pm
The radius of silver atom is 143.5 pm and it crystallises in face centred cubic arrangement. (Atomic mass of Ag = 107.47, NA = 6.023 × 1023)
What is the edge length of the unit cell?
405.8 pm
40.6 pm
331.4 pm
287 pm
The radius of silver atom is 143.5 pm and it crystallises in face centred cubic arrangement. (Atomic mass of Ag = 107.47, NA = 6.023 × 1023)
What is the density of silver metal?
5.36 g/cm3
8.60 g/cm3
10.72 g/cm3
7.07 g/cm3
Niobium crystallises in body centred cubic structure. Its density is 8.55 g cm−3 and its atomic mass is 93 g mol−1. (NA = 6.023 × 1023)
What is the edge length of niobium?
314.50 pm
330.56 pm
340.43 pm
346.30 pm
Niobium crystallises in body centred cubic structure. Its density is 8.55 g cm−3 and its atomic mass is 93 g mol−1. (NA = 6.023 × 1023)
What is the atomic radius of niobium?
136 pm
140 pm
143 pm
149 pm
Solutions for 1: Solid State
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 1 - Solid State Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 1 - Solid State - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 1 - Solid State
Shaalaa.com has the CISCE Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE 1 (Solid State) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 1 Solid State are Introduction to Solid State, Classification of Solids, Classification of Crystalline Solids, Space Lattice, Definition of Unit Cell, Different Types of Cubic Systems, Number of Particles Per Unit Cell in Different Cubic Systems, Calculation of the Space Occupied (Packing Fraction) in the Unit Cells of Different Types of Cubic Systems, Calculation of Density of a Crystal, Close-packed Structures, Packing of Constituent Particles in Crystals, Voids in Close-Packed Structures, Dimensions of Voids, Location of Tetrahedral Voids, Location of Octahedral Voids, Radius Ratio Rules, Number of Voids Filled and the Formula of the Compound, Types of Crystalline Solids: Molecular Solids, Types of Crystalline Solids: Ionic Solids, Types of Crystalline Solids: Covalent Solids {Atomic or Network Solids), Types of Crystalline Solids: Metallic Solids, Imperfections (Defects) in Solids, Imperfections (Defects) in Solids: Electronic Imperfections, Imperfections (Defects) in Solids: Atomic Imperfections, Imperfections (Defects) Caused by Impurities, Properties of Solids: Electrical Properties, Properties of Solids: Magnetic Properties, Properties of Solids: Dielectric Properties, Amorphous Solids.
Using Nootan केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी solutions Solid State exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 1, Solid State केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी additional questions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.
