Advertisements
Chapters
1: Solid State
2: Solutions
3: Electrochemistry
4: Chemical Kinetics
▶ 5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 5 - Surface Chemistry Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 5 - Surface Chemistry - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 5: Surface Chemistry
Below listed, you can find solutions for Chapter 5 of CISCE Nootan for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry REVIEW EXERCISES [Pages 292 - 315]
Explain the term physisorption.
Explain the term chemisorption.
How does the adsorption of a gas on solid surface vary with pressure?
What is meant by chemical adsorption?
What is the effect of increase of temperature on the adsorption of a gas on a solid surface?
Give a mathematical expression showing the relation between the amount of a gas adsorbed on a solid surface and the pressure of the gas.
By giving suitable examples distinguish between the terms absorption and adsorption.
Assuming adsorption to be a spontaneous process, show thermodynamically that it is always an exothermic process.
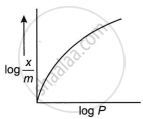
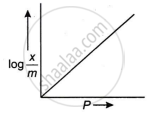
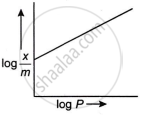
The adsorption of gases on solids can be described by Freundlich’s equation, `x/m = kP^(1//n)`.
- How are the values of k and n determined experimentally?
- Explain, why should the value of n be equal to or greater than one?
Compare physical adsorption and chemical adsorption in terms of rate and prevailing temperature.
Show graphically how the amount of a gas adsorbed on a solid in physical adsorption varies with pressure.
Show graphically how the amount of a gas adsorbed on a solid in physical adsorption varies with temperature.
Explain that adsorption is a surface phenomenon.
State two features of chemical adsorption which are not found with physical adsorption.
What do x and m represent in the following expression?
`(x/m) = k P^(1//n)`
Derive a mathematical expression showing the relationship between the extent of adsorption of a gas on a surface with pressure (within lower and higher ranges). Calculate the extent of adsorption at one atmosphere.
Mention some important applications of adsorption.
How is the phenomenon of adsorption used in the softening of hard water?
Show by a graphic diagram how at a constant pressure a rise in temperature will influence adsorption of a gas on a solid when (i) no compound formation occurs and (ii) chemisorption takes place.
How does chemical adsorption of a gas on a solid vary with temperature?
What is adsorption?
Show graphically how the amount of a gas adsorbed on a solid in physical adsorption varies with temperature.
How does the adsorption of a gas on solid surface vary with pressure?
Define adsorption.
Write three differences between physisorption and chemisorption.
Which will be adsorbed more readily on the surface of charcoal and why?
NH3 or CO2
How do size of particles of adsorbent, pressure of gas and prevailing temperature influence the extent of adsorption of a gas on a solid?
Define adsorption.
Write three differences between physisorption and chemisorption.
How does a catalyst work in homogeneous catalysis?
How does a catalyst work in heterogeneous catalysis?
Give reasons for the catalytic activity of finely divided iron in Haber’s process.
Give four examples of heterogeneous catalysis.
What is a shape selective catalyst?
What is the difference between homogeneous and heterogeneous catalysis?
Differentiate between homogeneous and heterogeneous catalysts with one example each.
Why is a catalyst, in general, used in the form of finely divided powder?
Explain two aspects of choosing heterogeneous catalyst.
Indicate a chemical reaction involving a homogeneous catalyst.
Name two industrial processes in which heterogeneous catalysts are employed.
What is shape selective catalysis?
Name a shape selective catalyst which converts alcohols directly to gasoline.
What is enzyme catalysis? Give some examples.
Write a brief note on the mechanism of enzyme catalysis.
Taking two examples of heterogeneous catalytic reactions, explain how a heterogeneous catalyst helps in the reaction.
What do you understand by activity of catalyst?
What do you mean by selectivity of catalyst?
Describe some features of catalysis by zeolites.
Give one example of homogeneous catalysis.
Give four examples of heterogeneous catalysis.
Explain shape selective catalysis with a suitable example.
Which of the following are not colloidal systems?
Milk, Sugar solution, Butter, Brass, Smoke, Cloud, 50% alcohol, Soap solution.
Explain the term electrodialysis.
Explain the term Tyndall effect.
What are lyophilic sols?
What are lyophobic sols?
Compare lyophilic and lyophobic sols in terms of stability and reversibility.
Explain the term emulsification by giving one example.
Explain the term demulsification by giving one example.
What are micelles?
Explain the cleansing action of soaps.
Explain what is observed when an electric current is passed through a colloidal sol?
What are associated colloids?
How associated colloids are different from multimolecular colloids?
What is the difference between a colloidal solution and an emulsion?
What is the difference between multimolecular and macromolecular colloids? Give one example of each. How are associated colloids different from these two types of colloids?
Describe briefly the following:
Dialysis
Describe briefly the following:
Electrophoresis
Describe the following term while stating the property of colloid:
Brownian movement
Describe the following term while stating the property of colloid:
Tyndall effect
Describe briefly the following:
Electrophoresis
Describe a chemical method for the preparation of sol of sulphur in water.
Describe a chemical method for the preparation of sol of platinum in water.
Explain what is observed when an electrolyte NaCl is added to hydrated ferric oxide sol.
Write whether Fe(OH)3 sol is a multimolecular or macromolecular colloid.
Explain why are lyophilic sols relatively more stable than lyophobic sols.
What are micelles?
How do micelles differ from normal colloidal solutions?
Differentiate micelles from multimolecular colloids and give example of each.
What are micelles? Give an example of a micellers system.
Why do colloidal solutions exhibit Tyndall effect?
Distinguish between true solution and colloidal solution of same colour.
Describe a chemical method for the preparation of sol of sulphur in water.
How is the following sol (in water) prepared?
Ferric hydroxide
Explain why are deltas formed where river and sea water meet.
Explain what is observed when a beam of light is passed through a colloidal sol.
Explain the observation likely to be made when an electric current are allowed to pass through a colloidal solution.
Distinguish between true solution and colloidal solution of same colour.
When are gas masks used and how do they work?
What is shape selective catalysis?
What is the difference between multimolecular and macromolecular colloids? Give one example of each. How are associated colloids different from these two types of colloids?
Account for the following:
Ferric hydroxide sol is positively charged.
Account for the following:
The extent of physical adsorption decreases with rise in temperature.
State the principle of electrodialysis of colloidal solutions.
Differentiate between emulsions and micelles taking a suitable example of each.
Explain the term electrodialysis.
Explain the following term:
Phases of a colloidal solution.
What is meant by the term peptization?
What are emulsions?
Write two applications of emulsification.
What happens when an electric field is applied to a colloidal dispersion?
Colloidal solutions can be prepared in water and in air as media. What distinctive names are given to these two types of sols?
Describe a chemical method for the preparation of sol of sulphur in water.
How is the following sol prepared?
Collodion
How does the addition of an alum purify water?
Why do lyophilic sols not require any stabilising agent for their preservation?
How is colloidal sulphur in water prepared?
In what way is a sol different from a gel?
How are the colloids classified on the basis of the nature of interaction between dispersed phase and dispersion medium? Describe an important characteristic of each class. Which of these sols need stabilising agents for preservation?
Explain the following term:
Peptization
Describe briefly the following:
Dialysis
Explain the following term:
Hardy-Schulze rule
Explain the term emulsification by giving one example.
Explain the following term, giving an example.
Coagulation
Explain the following term, giving an example.
Electrophoresis
Explain what is observed when an electrolyte is added to ferric hydroxide sol.
Explain what is observed when an emulsion is subjected to centrifugation.
Explain what is observed when direct current is passed through a colloidal sol.
What are the two types of emulsions and how do they differ from one another? Give one example of each.
Which one of the following electrolytes is most effective for the coagulation of Fe(OH)3 sol and why?
NaCl, Na2SO4, Na3PO4
What are lyophilic colloids? Give some examples.
What are lyophobic sols? Give two examples.
Illustrate with examples:
Multimolecular colloids
Illustrate with examples:
Macromolecular colloids
Write two examples of homogeneous catalysis.
Give four examples of heterogeneous catalysis.
Explain the following observation:
A beam of light passing through a colloidal solution has a visible path.
Explain the following observation:
Passing an electric current through a colloidal solution removes colloidal particles from it.
Explain the following observation:
Ferric hydroxide sol coagulates on addition of a solution of potassium sulphate.
What are the two classes of emulsions? Give one example of each class. State one activity to test the type of an emulsion.
What happens when gelatin is mixed with gold sol?
Explain shape selective catalysis with a suitable example.
Explain the following term with a suitable example.
Dialysis
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry VERY SHORT ANSWER TYPE QUESTIONS [Page 318]
Define adsorption.
What do you understand by absorption?
Define adsorbent.
Define adsorbate.
Out of adsorption and absorption, which is a surface phenomenon?
What do you understand by negative adsorption?
Which is a better adsorbent-a piece of coal or coal powder?
What type of gases do possess a greater tendency to get physically adsorbed on a solid?
Out of physisorption and chemisorption, which decreases with an increase in temperature?
The heat liberated during adsorption of a gas on a solid is 300 kJ mol−1. Predict the nature of adsorption.
What type of substances are used in gas masks?
How would you protect a delicate instrument against moisture?
Define colloidal solutions.
What is the size of particle in a true solution?
What is the size of particle in a colloidal solution?
Does a colloidal system represent a heterogeneous system?
Name the two phases present in a colloidal system.
What is a sol? Give two examples.
What is a gel? Give two examples.
What is an aerosol? Give an example.
What are intrinsic colloids? Give an example.
Is starch a hydrophilic colloid?
Define lyophobic colloids.
Out of lyophilic and lyophobic sols, which are more stable?
Name the instrument that is used to detect the presence of colloidal particles in a colloidal system.
Classify the following as multimolecular, macromolecular and associated colloids:
Starch sol, sulphur sol, gold sol, rubber sol, protein sol, soap solution.
Define the following term:
Multimolecular colloids
Define macromolecular colloids.
What is collodion?
Name the process used for the preparation of metal sols.
Can you purify a sol by filtering it through an ordinary filter paper?
What are ultrafilters?
Which colligative property is most significant for colloidal solutions?
Do colloidal solutions exhibit colligative properties?
What is Brownian movement?
Why do colloidal solutions exhibit Tyndall effect?
Why does the sky appear blue?
Which property does indicate the presence of electrical charge on colloidal particles?
Name two positively charged sols.
Name two negatively charged sols.
What happens when NaCl solution is added to a gold sol?
Arrange Fe3+, Na+, Ba2+, Sn4+ ions in the increasing order of their coagulating power to coagulate a negative sol.
What happens when a sol is subjected to persistent dialysis?
What happens when two oppositely charged sols are mixed together?
What type of colloids are used as protective colloids?
Why is gelatin regarded as an efficient protective colloid?
Why are colloidal medicines more effective?
What is the basis of artificial rain?
Which phenomenon is responsible for the formation of deltas in nature?
Define the emulsion. Give one example.
Name the emulsifying agent in milk.
What is demulsification?
What happens when a few drops of water are added to an oil-in-water type emulsion?
Define catalyst.
What do you understand by negative catalysis? Give an example.
Write two examples of homogeneous catalysis.
Give four examples of heterogeneous catalysis.
Out of homogeneous and heterogeneous catalysis, which is referred to as surface catalysis?
What do you mean by selectivity of catalyst?
What is the most important feature of zeolite catalysis?
Give an example of a selective adsorbent.
What do you understand by activity of catalyst?
Name the catalysts used in the following process:
Haber’s process
Name the catalysts used in the following process:
Contact process
Give an example of shape-selective catalysis.
State the Hardy-Schulze rule.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry SHORT ANSWER TYPE QUESTIONS [Pages 319 - 320]
Define adsorption.
Define absorption.
Define sorption.
Explain the term adsorption with one example.
Explain the term absorption with one example.
Explain the term sorption with one example.
By giving suitable examples distinguish between the terms absorption and adsorption.
What do you understand by positive adsorption?
What do you understand by negative adsorption?
Explain that adsorption is a surface phenomenon.
Explain the following:
A substance in the colloidal state is a much better adsorbent as compared to that in suspension form.
Explain the following:
Easily liquefiable gases are absorbed on solid surfaces more readily as compared to difficultly liquefiable gases.
Explain the following:
The physical adsorption decreases with an increase in temperature.
How does the adsorption of a gas on solid surface vary with pressure?
Why is physical adsorption multilayered, whereas chemical adsorption monolayered?
Write three differences between physisorption and chemisorption.
What type of forces are operative in (a) physical adsorption and (b) chemical adsorption? Justify the specificity of the two types of adsorption in the light of these forces.
What is the Freundlich adsorption isotherm? Illustrate it with a graph.
Can the adsorption of a solute from a solution be explained on the basis of the Freundlich adsorption isotherm? Explain.
How is the phenomenon of adsorption used in the softening of hard water?
What do you understand by the colloidal state of matter?
Define colloidal solutions.
How are colloidal solutions different from true solutions and suspensions?
Give an example of the following:
A sol
Give an example of the following:
A gel
Give an example of the following:
An emulsion
Give an example of the following:
An aerosol
What are lyophilic colloids? Give some examples.
What are lyophobic sols? Give two examples.
Distinguish between lyophobic and lyophilic sols.
What are multimolecular colloids? Give some examples.
What are macromolecular colloids and why are they called so? Give two examples.
What are associated colloids?
What are micelles and how are they formed? Explain with an example.
Explain the cleansing action of soaps.
Describe Bredig’s arc method used for the preparation of metal sols.
What is meant by the term peptization?
Describe some chemical reactions which lead to the formation of colloidal solutions.
How is the exchange of solvent useful in the preparation of a colloidal solution? Explain with an example.
What is dialysis and how is it carried out?
What is electrodialysis? Explain with a suitable diagram.
What is ultrafiltration and how is the technique useful in the purification of a colloidal solution?
What are ultrafilters, and how are they prepared?
Why is the magnitude of a colligative property for a colloidal solution much smaller than that obtained for a true solution?
What is Brownian movement and why is it exhibited by colloidal solutions?
What is the Tyndall effect?
Why do colloidal solutions exhibit Tyndall effect?
Why is the Tyndall effect exhibited by colloidal solutions but not by true solutions?
Describe an experiment to show that colloidal particles carry a particular type of electrical charge.
How do colloidal particles develop electrical charges on them?
What is electrophoresis? How is the phenomenon useful in sewage disposal?
Define electro-osmosis and illustrate it diagrammatically.
How does the presence of electrical charge on colloidal particles keep a hydrophobic sol stable?
What do you understand by coagulation?
How can coagulation be achieved?
What do you understand by the flocculation value of an electrolyte?
What is the significance of the flocculation value?
What are protective colloids and how do they protect a hydrophobic sol?
Define gold number.
Discuss the significance of the gold number.
Name the property of colloidal solution involved in the following application:
Smoke precipitation
Name the property of colloidal solution involved in the following application:
Purification of water
Name the property of colloidal solution involved in the following application:
Cleansing action of soaps
Name the property of colloidal solution involved in the following application:
Sewage disposal
Explain the following:
Muddy water can be purified by adding potash alum to it.
Explain the following:
Colloidal dust is sprayed on clouds to compel them to rain.
Explain why are deltas formed where river and sea water meet.
What are emulsions?
How are emulsions prepared?
What do you understand by o/w type emulsions? Give one example.
What do you understand by w/o type emulsions? Give one example.
How would you ascertain that a given emulsion is of o/w or w/o type?
What is the role of an emulsifier in the preparation of an emulsion?
What is the role of an emulsifier in the stability of an emulsion?
Describe the role of emulsification during the digestion of fats in the intestine.
What do you understand by negative catalysis? Give an example.
What is homogeneous catalysis?
Explain the theory of homogeneous catalysis with the help of a suitable example.
What is heterogeneous catalysis?
Give four examples of heterogeneous catalysis.
How does a heterogeneous catalyst catalyse a chemical reaction? Explain in the light of adsorption theory.
Discuss the two important aspects of solid catalysts.
What are zeolites?
What is the most important feature of zeolite catalysis?
What is shape selective catalysis?
What structural features of zeolites are responsible for the shape selective catalysis exhibited by them?
Give reason why a finely divided substance is more effective as an adsorbent.
What do you understand by activation of adsorbent?
How is activation of adsorbent achieved?
Action of soap is due to emulsification and micelle formation. Comment.
What role does adsorption play in heterogeneous catalysis?
Comment on the statement that “colloid is not a substance but a state of substance”.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry LONG ANSWER TYPE QUESTIONS [Page 320]
Differentiate between the following:
Adsorption and Absorption
What are the factors which influence the adsorption of a gas on a solid?
Give two important characteristics of physisorption.
Write any two characteristics of chemisorption.
Write three differences between physisorption and chemisorption.
What is the Freundlich adsorption isotherm? Suggest a method to evaluate the constants k and n present in the equation involved. What form does this isotherm assume when applied to adsorption of solute from solutions?
What is adsorption?
Mention some important applications of adsorption.
What are colloidal solutions?
Give examples of different types of colloidal solutions.
Describe the important methods used for the preparation of colloidal solutions.
Why is it necessary to purify colloidal solutions?
Describe the important methods employed for the purification of a sol.
What are lyophilic sols?
What are lyophobic sols?
Why are lyophilic sols called reversible sols?
Why are lyophobic sols called irreversible sols?
Distinguish between lyophobic and lyophilic sols.
What are multimolecular colloids? Give some examples.
Illustrate with examples:
Macromolecular colloids
What are associated colloids?
What are micelles?
Explain micelle formation in a soap solution.
Explain the cleansing action of soaps.
How do colloidal particles develop electrical charges on them?
Describe the important electrical properties of colloidal solutions.
How can a lyophobic sol be protected against coagulation caused by an electrolyte?
What are protective colloids and how do they work?
Define gold number.
Describe some of the important applications of colloids.
What are emulsions?
How are emulsions classified?
Describe the preparation of emulsions.
Describe the properties of emulsions.
Write two applications of emulsification.
What do you understand by catalyst?
What do you understand by catalysis?
How is catalysis classified on the basis of physical states of reactants and the catalyst? Give two examples of each type.
What is homogeneous catalysis?
What is heterogeneous catalysis?
Explain the mechanism of homogeneous catalysis?
Explain the mechanism of heterogeneous catalysis?
What do you mean by selectivity of catalyst?
Write a note on the shape selective catalysis exhibited by zeolites.
What is an adsorption isotherm?
Distinguish between freundlich adsorption isotherm and langmuir adsorption isotherm.
How are the colloidal solutions classified on the basis of physical states of the dispersed phase and dispersion medium?
How are colloids classified on the basis of the nature of the dispersion medium?
How are colloids classified on the basis of interaction between the dispersed phase and the dispersion medium?
Explain what is observed when a beam of light is passed through a colloidal sol.
Explain what is observed when an electrolyte NaCl is added to hydrated ferric oxide sol.
Explain what is observed when an electric current is passed through a colloidal sol?
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 320 - 324]
Choose the correct option in the following questions.
Chemisorption is ______.
multimolecular in nature
reversible
often highly specific and directional
not very specific
The function of negative catalyst is ______.
to remove the active intermediate from the reaction
to terminate the chain reaction
both to remove the active intermediate from the reaction and to terminate the chain reaction
none of the above
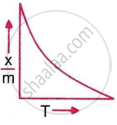
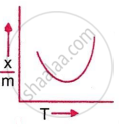
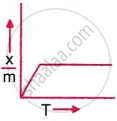
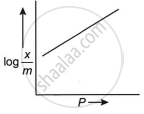
Which plot is the adsorption isobar for chemisorption, where x is the amount of gas adsorbed on mass m (at constant pressure) at temperature T?
Gas masks containing activated charcoal to remove poisonous gases from the atmosphere make use of the phenomenon of ______.
adsorption
absorption
sorption
desorption
Which statement is not correct?
Physical adsorption is due to van der Waals’ forces.
Physical adsorption decreases at high temperature and low pressure.
Physical adsorption is reversible.
Adsorption energy for a chemical adsorption is generally lesser than that of physical adsorption.
For adsorption of gas on solid surface, the plot of log `x/m` vs log P is linear with a slope equal to ______.
k
log k
ln k
`1/n` (n being an integer)
Which does form a colloidal solution in water?
NaCl
Glucose
Starch
Barium nitrate
Colloidal solutions are not purified by ______.
dialysis
electrodialysis
electrophoresis
ultrafiltration
The Brownian movement is due to ______.
temperature fluctuations within the liquid phase
attraction and repulsion between charges on the colloidal particles
impact of the molecules of the dispersion medium on the colloidal particles
convective currents
Peptization is a process of ______.
precipitating colloidal particles
purifying colloidal particles
dispersing the precipitate into colloidal state
none of the above
Which reaction does yield a colloidal solution?
\[\ce{Cu + HgCl2 -> CuCl2 + Hg}\]
\[\ce{2HNO3 + 3H2S -> 3S + 4H2O + 2NO}\]
\[\ce{2Mg + CO2 -> 2MgO + C}\]
\[\ce{Cu + CuCl2 -> CuCl2}\]
The charge on As2S3 sol is due to the adsorption of ______.
H+
OH−
O2−
S2−
The method employed for sol destruction is ______.
condensation
peptization
addition of electrolyte
none of these
Fog is a colloidal solution of ______.
liquid particles dispersed in gas
gaseous particles dispersed in a liquid
solid particles dispersed in a liquid
solid particles dispersed in gas
Brownian motion of sol particle is the ______ property of sol.
electrical
optical
kinetic
colligative
Surface tension of lyophilic sols is ______.
lower than H2O
more than H2O
equal to H2O
none of these
In emulsion, the dispersed phase and dispersion medium are ______.
both solids
both liquids
a solid and liquid
a liquid and solid
A colloidal system in which gas is dispersed in a liquid to form bubbles is known as ______.
foam
sol
aerosol
emulsion
Cod liver oil is ______.
fat dispersed in water
water dispersed in fat
water dispersed in oil
fat dispersed in fat
Which does not show the Tyndall effect?
Emulsion
Blood
Milk
Sugar solution
At CMC, the surfactant molecules undergo:
association
aggregation
micelle formation
all of these
Lyophilic sols are more stable than lyophobic sols because ______.
the colloidal particles have positive charge.
the colloidal particles have no charge.
the colloidal particles are solvated.
there are strong electrostatic repulsions between the negatively charged colloidal particles.
Which of the following electrolytes is least effective in causing flocculation of ferric hydroxide sol?
K3Fe(CN)6
K2CrO4
KBr
K2SO4
The colour of the colloidal particles of gold obtained by different methods differs because of ______.
variable valency of gold
different concentrations of gold particles
different types of impurities
different diameters of colloidal particles
The activity and selectivity of zeolites as catalysts are based on ______.
their pore size
size of their cavities on the surface
both their pore size and size of their cavities on the surface
none of the above
Which is not true in the case of a catalyst?
The catalyst is unchanged chemically at the end of a reaction.
The catalyst accelerates a reaction.
In a reversible reaction, the catalyst alters the equilibrium position.
A small amount of catalyst is often sufficient to bring about a large change in reaction.
Which is used in the Haber’s process for the manufacture of NH3?
Al2O3
Fe + Mo
CuO
Pt
Which process does not involve a catalyst?
Haber’s process
Thermite process
Ostwald’s process
Contact process
Shape selective catalysis is exhibited by ______.
finely divided nickel
ZSM-5
Lindlar’s catalyst
platinised asbestos
Zeolites:
are microporous aluminosilicates.
have general formula Mx/n [(AlO2)x (SiO2)y] · mH2O.
have a pore size between 260-740 pm.
all of the above
On the addition of 1 mL of 10% NaCl to 10 mL of gold sol in the presence of 0.0250 g of starch, the coagulation is just prevented. Starch has the gold number:
0.025
0.25
2.5
25
The ability of an ion to bring about the coagulation of a given colloid depends upon ______.
its charge
the sign of the charge alone
the magnitude of charge
both magnitude and charge
Gelatin is mostly used in making ice creams in order to ______.
prevent forming the colloidal sol
enrich the fragrance
prevent crystallisation and stabilise the mix
modify the taste
Which of the following is a lyophilic colloid?
Milk
Gum
Fog
Blood
The process of separation of a colloid from crystalloid through semipermeable membrane is known as ______.
dialysis
coagulation
ultrafiltration
titration
For a linear plot of `log (x/m)` versus log p in a Freundlich adsorption isotherm, which of the following statement is correct? (k and n are constants.)
Both k and `1/n` appear in the slope term.
`1/n` appears as the intercept.
Only `1/n` appears as the slope.
`log (1/n)` appears as the intercept.
Which one of the following characteristics is associated with adsorption?
ΔG and ΔH are negative but ΔS is positive.
ΔG and ∆S are negative but ∆H is positive.
∆G is negative but ∆H and ∆S are positive.
∆G, ∆H and ∆S all are negative.
The addition of a catalyst during a chemical reaction alters which of the following quantities?
Enthalpy
Activation energy
Entropy
Internal energy
Fog is a colloidal solution of ______.
Solid in gas
Gas in gas
Liquid in gas
Gas in liquid
Which one of the following statements is not correct?
Catalyst does not initiate any reaction.
The value of the equilibrium constant is changed in the presence of a catalyst in the reaction at equilibrium.
Enzymes catalyse mainly biochemical reactions.
Coenzymes increase the catalytic activity of enzymes.
On which of the following properties does the coagulating power of an ion depend?
Both magnitude and sign of the charge on the ion.
Size of the ion alone.
The magnitude of the charge on the ion alone.
The sign of charge on the ion alone.
A gas undergoes physical adsorption on a surface and follows the given Freundlich adsorption isotherm equation `x/m = kp^0.5`·
Adsorption of the gas increases with:
decrease in p and increase in T.
increase in p and decrease in T.
decrease in p and decrease in T.
increase in p and increase in T.
The correct option among the following is:
Addition of alum to water makes it unfit for drinking.
Colloidal medicines are more effective because they have small surface area.
Colloidal particles in lyophobic sols can be precipitated by electrophoresis.
Brownian motion in colloidal solution is faster if the viscosity of the solution is very high.
Peptization is a ______.
process of converting soluble particles to form colloidal solution
process of converting precipitate into colloidal solution
process of bringing colloidal molecule into solution
process of converting a colloidal solution into precipitate
The correct option representing a Freundlich adsorption isotherm is:
`x/m = kp^-1`
`x/m = kp^0.3`
`x/m = kp^2.5`
`x/m = kp^-0.5`
Which mixture of the solutions will lead to the formation of negatively charged colloidal [AgI]I− sol?
50 mL of 1 M AgNO3 + 50 mL of 1.5 M KI
50 mL of 1 M AgNO3 + 50 mL of 2 M KI
50 mL of 2 M AgNO3 + 50 mL of 1.5 M KI
50 mL of 0.1 M AgNO3 + 50 mL of 0.1 M KI
Measuring Zeta potential is useful in determining which property of colloidal solution?
Viscosity
Solubility
Stability of the colloidal particles
Size of the colloidal particles
Which of the following is used for the preparation of colloids?
Ostwald process
Van Arkel method
Mand process
Bredig’s Arc method
Tyndall effect is observed when ______.
The diameter of dispersed particles is similar to the wavelength of light used.
The refractive index of dispersed phase is greater than that of the dispersion medium.
The diameter of dispersed particles is much smaller than the wavelength of light used.
The diameter of dispersed particles is much larger than the wavelength of light used.
A sample of red ink (a colloidal suspension) is prepared by mixing eosin dye, egg white, HCHO and water. The component which ensures stability of the ink sample is ______.
HCHO
Water
Eosin dye
Egg white
The right option for the statement “Tyndall effect is exhibited by”, is:
Urea solution
NaCl solution
Glucose solution
Starch solution
The incorrect statement below regarding colloidal solutions is:
A colloidal solution shows colligative properties.
An ordinary filter paper can stop the flow of colloidal particles.
The flocculating power of Al+3 is more than that of Na+.
A colloidal solution shows Brownian motion of colloidal particles.
For the coagulation of negative sol, the species below that has the highest flocculating power is:
\[\ce{SO^{2-}4}\]
Ba+2
Na+
\[\ce{PO^{3-}4}\]
The charges on the colloidal CdS sol and TiO2 sol are respectively:
positive and positive
positive and negative
negative and negative
negative and positive
The correct statement(s) related to colloids is (are):
The process of precipitating colloidal sol by an electrolyte is called peptization.
Colloidal solution freezes at high temperature than the true solution at the same concentration.
Surfactants form micelle; critical micelle concentration (CMC) depends on temperature.
Micelles are macromolecular colloids.
The incorrect statement regarding enzymes is:
Like chemical catalysts enzymes reduce the activation energy of bio processes.
Enzymes are polysaccharides.
Enzymes are very specific for a particular reaction and substrate.
Enzymes are biocatalysts.
Statement I: In the coagulation of a negative sol, the flocculating power of the three given ions is in the order:
Al+3 > Ba+2 > Na+
Statement II: In the coagulation of a positive sol, the flocculating power of three given salts is in the order:
NaCl > Na2SO4 > Na3PO4
In the light of above statements, choose the most appropriate answer from the options given below:
Both statement I and statement II are incorrect.
Statement I is correct but statement II is incorrect.
Statement I is incorrect but statement II is correct.
Both statements I and II are correct.
For micelle formation, which of the following statements are correct?
- Micelle formation is an exothermic process.
- Micelle formation is an endothermic process.
- The entropy change is positive.
- The entropy change is negative.
A and D only
A and C only
B and C only
B and D only
Among the following, the number of curves not in accordance with Freundlich adsorption isotherm is (nearest integer):
100 ml of 5% (W.V.) solution of NaCl in water was prepared in 250 ml beaker. Albumin from the egg was poured into NaCl solution and stirred well. This resulted in a/an ______.
Lyophilic sol
Lyopholic sol
Emulsion
Precipitate
Given below are the critical temperatures of some of the gases:
| Gas | Critical temperature (K) |
| He | 5.2 |
| CH4 | 190 |
| CO2 | 304.2 |
| NH3 | 405.5 |
The gas showing the least adsorption on a definite amount of charcoal is:
He
CH4
CO2
NH3
The correct option(s) related to adsorption processes is (are):
Chemisorption results in a unimolecular layer.
The enthalpy change during physisorption is in the range of 100 to 140 kJ mol−1.
Chemisorption is an endothermic process.
Lowering the temperature favours physisorption processes.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry FILL IN THE BLANKS TYPE QUESTIONS [Page 324]
The change of concentration at a surface brought about by the action of residual surface forces is known as ______.
Absorption is a ______ phenomenon.
In chemical adsorption, the adsorption first ______ and then ______ with an increase in temperature.
Physical adsorption is ______ layered, whereas chemical adsorption is ______ layered.
The slope of the line obtained on plotting \[\text{log}_{10}\frac{x}{m}\] against log10 P is equal to ______.
Colloidal particles can be made to settle under ______.
The colloidal dispersion of a liquid into a solid is called a ______.
Lyophilic sols are ______ in nature, whereas lyophobic sols are ______ in nature.
Among lyophilic and lyophobic sols, ______ sols are more stable.
When the dispersed particles themselves are large molecules, the colloidal dispersions are termed as ______ colloids.
Colloidal solution of cellulose nitrate in ethyl alcohol is called ______.
The process of dispersing a freshly prepared precipitate into colloidal form is called ______.
The most significant colligative property for colloidal solution is ______.
The movement of colloidal particles under the influence of an electric field is called ______.
The precipitation of a colloidal solution on addition of an electrolyte is called ______ or ______.
Colloidal dispersion of two ______ liquids is called an ______.
In a w/o type emulsion, water acts as the ______ while the oil as ______.
When a small amount of electrolyte is added to an oil-in-water type emulsion, the conductance ______.
The hydrolysis of an ester in the presence of dilute sulphuric acid is an example of ______ catalysis.
Heterogeneous catalysis can be explained on the basis of ______ theory.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry ASSERTION-REASON TYPE QUESTIONS [Page 325]
Assertion: Chemisorption is multilayered in nature.
Reason: In chemisorption, adsorbate molecules are attached to the adsorbent surface by strong valence bond forces.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: In homogeneous catalysis, the catalyst enhances the rate of reaction by providing an alternative path of lower activation energy.
Reason: The catalyst combines with a reactant to form an unstable intermediate compound.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: Zeolites are shape selective catalysts.
Reason: The catalysing action of a zeolite depends upon the size of pores and cavities present in it.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: The soap micelle is a negatively charged colloidal particle.
Reason: The negative charge on the micelle is due to the presence of OH− ions of water.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: Tyndall effect is not exhibited by true solutions.
Reason: The particles present in a true solution are too small to scatter light.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: Colloidal solutions are stable but colloidal particles do not settle down.
Reason: Brownian movement counters the force of gravity actively on colloidal particles.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: Finest gold is red in colour. As the size of the particles increases, it appears purple then blue and finally gold.
Reason: The colour of the colloidal solution depends upon the wavelength of the light scattered by the dispersed particles.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Assertion: Dissolved substances can be removed from a colloidal solution by diffusion through parchment paper.
Reason: Particles in a true solution cannot pass through parchment paper but the colloidal particles can pass through the parchment paper.
If both Assertion and Reason are correct and Reason is the correct explanation of the Assertion.
If both Assertion and Reason are correct but Reason is not the correct explanation of the Assertion.
If Assertion is correct but Reason is incorrect.
If Assertion is incorrect but Reason is correct.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry 'NCERT TEXT-BOOK' Exercises [Pages 325 - 326]
Distinguish between the meaning of the terms adsorption and absorption. Give one example of.
Write three differences between physisorption and chemisorption.
Give reason why a finely divided substance is more effective as an adsorbent.
What are the factors which influence the adsorption of a gas on a solid?
What is an adsorption isotherm?
What is the Freundlich adsorption isotherm? Illustrate it with a graph.
How is activation of adsorbent achieved?
What do you understand by activation of adsorbent?
How is activation of adsorbent achieved?
What role does adsorption play in heterogeneous catalysis?
Why is adsorption always exothermic?
How are the colloidal solutions classified on the basis of physical states of the dispersed phase and dispersion medium?
Discuss the effect of pressure on the adsorption of gases on solids.
Discuss the effect of temperature on the adsorption of gases on solids.
What are lyophilic colloids? Give some examples.
What are lyophobic sols? Give two examples.
Why are hydrophobic sols easily coagulated?
What is the difference between multimolecular and macromolecular colloids? Give one example of each. How are associated colloids different from these two types of colloids?
What are enzymes?
Write a brief note on the mechanism of enzyme catalysis.
How are the colloidal solutions classified on the basis of physical states of the dispersed phase and dispersion medium?
How are colloids classified on the basis of the nature of the dispersion medium?
How are colloids classified on the basis of interaction between the dispersed phase and the dispersion medium?
Explain what is observed when a beam of light is passed through a colloidal sol.
Explain what is observed when an electrolyte NaCl is added to hydrated ferric oxide sol.
Explain what is observed when an electric current is passed through a colloidal sol?
What are emulsions?
What are the different types of emulsions? Give one example of each type.
What is demulsification?
Name two demulsifiers.
Action of soap is due to emulsification and micelle formation. Comment.
Give four examples of heterogeneous catalysis.
What do you understand by activity of catalyst?
What do you mean by selectivity of catalyst?
Describe some features of catalysis by zeolites.
What is shape selective catalysis?
Explain the following term, giving an example.
Electrophoresis
Explain the following term, giving an example.
Coagulation
Explain the following term with a suitable example.
Dialysis
Explain the term Tyndall effect.
Give four uses of emulsions.
What are micelles? Give an example of a micellers system.
Explain the term with suitable examples:
Alcosol
Explain the term with suitable examples:
Aerosol
Explain the term with suitable examples:
Hydrosol
Comment on the statement that “colloid is not a substance but a state of substance”.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 5 Surface Chemistry QUESTIONS FROM ISC EXAMINATION PAPERS [Page 327]
Give a reason for the following:
For ferric hydroxide sol the coagulating power of phosphate ion is more than chloride ion.
Give a reason for the following:
Medicines are more effective in their colloidal form.
Give a reason for the following:
Gelatin is added to ice creams.
Define the following term with suitable examples:
Peptisation
Define the following term with suitable examples:
Electrophoresis
Define the following term with suitable examples:
Dialysis
Which of the following electrolytes is least effective in causing the flocculation of positively charged ferric hydroxide sol?
K3[Fe(CN)6]
K2CrO4
K4[Fe(CN)6]
KBr
Explain what is observed when a beam of light is passed through a colloidal sol.
Explain what is observed when an electric current is passed through a colloidal sol?
Explain what is observed when an electrolyte (AlCl3) is added to a colloidal solution of arsenious sulphide (As2S3).
Answer the following questions:
Which aqueous solution of concentration 0.01 M salt i.e., KCl, MgCl2 and AlCl3 would be most effective in coagulating a negatively charged colloidal solution of As2S3?
Bleeding caused by a nick from a razor during shaving can be stopped by rubbing alum. Give a reason.
Muddy water can be purified by adding potash alum to it. Give a reason.
Solutions for 5: Surface Chemistry
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 5 - Surface Chemistry Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 5 - Surface Chemistry - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 5 - Surface Chemistry
Shaalaa.com has the CISCE Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE 5 (Surface Chemistry) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 5 Surface Chemistry are .
Using Nootan केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी solutions Surface Chemistry exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 5, Surface Chemistry केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी additional questions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.