Advertisements
Advertisements
प्रश्न
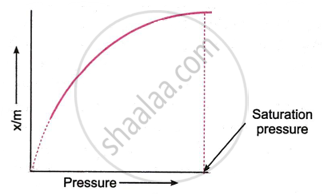
How does the adsorption of a gas on solid surface vary with pressure?
How does adsorption of a gas on a solid surface vary with pressure? Illustrate with the help of appropriate graph.
How does the extent of adsorption vary with pressure? Illustrate your answer with a graph.
आलेख
लघु उत्तरीय
Advertisements
उत्तर
The adsorption of a gas on a solid surface generally increases with pressure. At low pressure, adsorption rises rapidly as more gas molecules attach to the surface. As pressure increases further, adsorption approaches a maximum value because the surface becomes saturated with gas molecules. This relationship is often described by adsorption isotherms. Thus, adsorption increases with pressure but levels off at high pressures due to surface saturation.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
