Advertisements
Chapters
1: Solid State
2: Solutions
3: Electrochemistry
▶ 4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 4 - Chemical Kinetics Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 4 - Chemical Kinetics - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 4: Chemical Kinetics
Below listed, you can find solutions for Chapter 4 of CISCE Nootan for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics REVIEW EXERCISES [Pages 223 - 254]
What do you understand by the rate of a reaction?
Define average rate of reaction.
Define instantaneous rate of reaction.
Express the average rate of following reaction in terms of concentrations of reactants and products:
\[\ce{2NO2 + F2 -> 2NO2F}\]
Express the average rate of following reaction in terms of concentrations of reactant and product:
\[\ce{H2_{(g)} + I2_{(g)} -> 2HI_{(g)}}\]
Express the average rate of following reaction in terms of concentration of reactant and product:
\[\ce{2ICl_{(g)} + H2_{(g)} -> I2_{(g)} + 2HCl_{(g)}}\]
For a chemical reaction \[\ce{2A + B -> 2C + 3D}\], the rate of disappearance of A is 0.10 mol L−1 s−1. Calculate the rate of reaction and the rate of appearance of D.
The reaction, \[\ce{2N2O5_{(g)} -> 4NO2_{(g)} + O2_{(g)}}\] is carried out in a closed vessel. The concentration of N2O5(g) is found to decrease by 2 × 10−2 mol L−1 in 10 seconds. Calculate the rate of reaction and the rate of appearance of NO2(g).
For the reaction, \[\ce{N2_{(g)} + 3H2_{(g)} -> 2NH3_{(g)}}\], the rate of reaction is found to be 2 × 10−4 mol L−1 s−1. Calculate the rate of disappearance of Ni(g) and that of H2(g).
For the reaction, \[\ce{N2O4_{(g)} <=> 2NO2_{(g)}}\], it has been found that the pressure of N2O4 falls from 0.64 atm to 0.38 atm in 28 minutes. Calculate the rate of reaction and the rate of appearance of NO2(g).
Express the relationship between the rate of production of water and the rate of disappearance of oxygen in the following reaction:
\[\ce{2H2 + O2 -> 2H2O}\]
What aspect of a reaction is influenced by presence of catalyst which increases the rate or possibility of the reaction?
Express the rate of the following reaction in terms of disappearance of hydrogen in the reaction:
\[\ce{3H2_{(g)} + N2_{(g)} -> 2NH3_{(g)}}\]
What do you understand by rate law of a reaction?
What is the rate constant?
What are the units of rate constant for a reaction of order n?
Define the following term:
Order of a reaction
What is specific reaction rate?
How is specific reaction rate different from the reaction rate?
How is rate constant related to the concentration of reactants?
Name one zero order reaction.
Identify the reaction order if the units of rate constant are s−1.
The rate constant of a reaction is 2 × 10−2 L mol−1 s−1. What is the order of reaction?
The reaction, A + B → C, has zero order. Write the rate equation.
The form of rate law for a reaction is expressed as
Rate = k[Cl2] [NO]2
Find out the order of reaction with respect to Cl2, with respect to NO and also the overall order of the reaction.
For the reaction \[\ce{2A + B + C -> A2B + C}\], rate law has been determined to be, Rate = k [A] [B]2. If the rate constant k = 2.0 × 10−6 mol−2Ls−1, what will be the initial rate of the reaction with [A] = 0.1 mol L−1, [B] = 0.2 mol L−1 and [C = 0.8 mol L−1?
The reaction \[\ce{2A + B -> C + D}\] obeys the rate equation, Rate = k [A]2 [B]1/2. Find the order of this reaction when A is present in large excess.
Determine the order of reaction which has the rate expression, Rate = k [A]1/2[B]1/2.
Express the relationship between the rate of production of oxygen and the rate of disappearance of hydrogen peroxide (H2O2) in the following reaction.
\[\ce{2H2O2 -> 2H2O + O2}\]
The rate of formation of a dimer in a second order dimerisation reaction is 6.5 × 10−6 mol L−1s−1 at 0.01 mol L−1 monomer concentration. Calculate the rate constant.
The rate of a gaseous reaction is halved when the volume of the vessel is doubled. What is the order of reaction?
What is meant by relative rates of reaction?
Write the relative rate expressions for the following chemical reaction:
\[\ce{4NH3_{(g)} + 5O2_{(g)} -> 4NO_{(g)} + 6H2O_{(g)}}\]
For the reaction, A + B → Products, the rate law is Rate = k [A]2/3 [B]2. What are the units of rate constant?
For the reaction, \[\ce{2A + B + C -> A2B + C}\], the rate law has been determined to be
Rate = k[A][B]2.
If the value of k is 2.0 × 10−6 mol−2 L2 s−1 for the reaction, determine the initial rate of the reaction with [A] = 0.2 mol L−1, [B] = 0.1 mol L−1 and [C] = 0.5 mol L−1.
The rate of decomposition of a substance A becomes eight times when its concentration is doubled. What is the order of this reaction?
The rate law for the decomposition of N2O5 is Rate = k [N2O5].
What is the significance of k in this equation?
A reaction is first order in A and second order in B.
- Write differential rate equation.
- How is the rate affected if the concentration of B is tripled?
- How is the rate affected if the concentrations of both A and B are doubled?
What is the significance of rate constant in the rate expression?
Following reaction takes places in one step:
\[\ce{2NO_{(g)} + O2_{(g)} -> 2NO2_{(g)}}\]
How will the rate of the above reaction change if the volume of the reaction vessel is diminished to one third of its original volume? Will there be any change in the order of the reaction with the reduced volume?
Give one example of a pseudo first-order reaction.
For the reaction \[\ce{2X -> X2}\], rate of reaction becomes three times when the concentration of X is increased by 27 times. What is the order of reaction?
For the reaction \[\ce{Cl2_{(g)} + 2NO_{(g)} -> 2NOCl_{(g)}}\] the rate law is expressed as rate = k [Cl2][NO]2. What is the overall order of this reaction?
The decomposition of NH3 on platinum surface,
\[\ce{2NH3_{(g)} ->[Pt] N2_{(g)} + 3H2_{(g)}}\]
is a zero order reaction with k = 2.5 × 10−4 Ms−4. What are the rates of production of N2 and H2?
What are the main characteristics of a first order reaction?
What is the rate constant?
Define half-life period.
Show that for a first order reaction half life is independent of initial concentration.
Write the rate law for a first order reaction.
Justify the statement that half-life of such a reaction is independent of the initial concentration of the reactant.
How is the progress of reaction followed in the following first order reaction:
Decomposition of N2O5;
How is the progress of reaction followed in the following first order reaction:
Hydrolysis of ethyl acetate in the presence of dil. HCl;
How is the progress of reaction followed in the following first order reaction:
Inversion of cane sugar?
What will be the shape of the curve if the concentration of the reactant is plotted against time for a first order reaction?
A reaction is 50% complete in 2 h and 75% complete in 4 h. What is the order of reaction?
A first order reaction is 20% complete in 10 minutes. Calculate the time taken for the reaction to go to 80% completion.
Time required to decompose SO2Cl2 to half of its initial amount is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
Find the three-fourths life, t3/4 of a first order reaction when for it k = 7.4 × 10−5 s−1.
A first order reaction 'has a specific reaction rate of 10−2 s−1. How much time will it take for 10 g of its reactant to be reduced to 2.5 g?
A reaction that is first order with respect to reactant A has a rate constant of 6 min−1. If we start with [A] = 50 mol L−1, when would [Al reach the value of 0.05 mol L−1?
A first order reaction takes 100 minutes for completion of 60 per cent of reaction. Find the time when 90% of the reaction will be completed.
Calculate the half-life of a first order reaction having a value of 2 × 10−3 s−1 for its rate constant.
A radioactive element is half disintegrated in 40 minutes. What is the time required for the decay of 75% of the element?
In a reaction, 5 g ethyl acetate is hydrolysed per litre in the presence of dil. HCl in 300 min. If the reaction is of first order, and the initial concentration of ethyl acetate is 22 g/L, then calculate the rate constant of the reaction.
The decomposition of N2O5 in CCl4 solution follows the first order rate law. The concentration of N2O5 measured at different time intervals are given below.
| Time in seconds (t) | 0 | 80 | 160 | 410 | 600 | 1130 | 1720 |
| [N2O5] (mol/L) | 5.5 | 5.0 | 4.8 | 4.0 | 3.4 | 2.4 | 1.6 |
Calculate its rate constant at t = 410 s and t = 1130 s. What do these results show?
The decomposition of [N2O5] in a carbon tetrachloride solution has been found to be of first order with rate constant k = 6.2 × 10−4 s−l.
\[\ce{N2O5 (Solution) -> 2NO2 (Solution) + \frac{1}{2} O2_{(g)}}\]
Calculate the rate of reaction when [N2O5] = 1.75 mol L−l. At what concentration of N2O5 the reaction would have a rate of 3.8 × 10−3 mol L−1 s−1?
The following data were obtained at 300 K for the reaction 2A + B → C + D:
| Experiment number | [A] (mol L−1) | [B] (mol L−1) | Rate of formation of D (mol L−1 min−1) |
| 1 | 0.1 | 0.1 | 6.0 × 10−3 |
| 2 | 0.3 | 0.2 | 7.4 × 10−2 |
| 3 | 0.3 | 0.4 | 2.88 × 10−1 |
| 4 | 0.4 | 0.1 | 2.4 × 10−2 |
Calculate the rate of formation of D when [A] = 0.5 mol L−1 and [B] = 0.2 mol L−1.
Following data were obtained for the decomposition of N2O5 which is a first order reaction.
| t (min) | [N2O5] (mol L−1) |
| 0 | 1.000 |
| 10 | 0.705 |
| 20 | 0.497 |
| 30 | 0.349 |
From the data given above, determine the value of
- specific reaction rate and
- half-life of the reaction
Consider the following data for the reaction:
\[\ce{A + B -> Product}\]
| Experiment Number | [A] mol L−1 | [B] mol L−1 | Rate mol sec−1 |
| 1. | 0.10 M | 1.0 | 2.1 × 10−3 |
| 2. | 0.20 M | 2.0 | 8.4 × 10−3 |
| 3. | 0.30 M | 3.0 | 8.4 × 10−3 |
Determine the order of reaction with respect to A and with respect to B and the overall order of a reaction.
For the reaction, \[\ce{2A + B + C -> A2B + C}\], the rate law has been determined to be
Rate = k[A][B]2.
If the value of k is 2.0 × 10−6 mol−2 L2 s−1 for the reaction, determine the initial rate of the reaction with [A] = 0.2 mol L−1, [B] = 0.1 mol L−1 and [C] = 0.5 mol L−1.
For the reaction \[\ce{2NO_{(g)} + C2_{(g)} -> 2NOCl_{(g)}}\], following data were obtained.
| Experiment | Initial conc. of NO (mol L−1) | Initial conc. of Cl2 (mol L−1) | Initial rate (mol L−1 s−1) |
| 1 | 0.010 | 0.020 | 2.40 × 10−4 |
| 2 | 0.030 | 0.020 | 2.16 × 10−3 |
| 3 | 0.030 | 0.040 | 4.32 × 10−3 |
Determine the orders with respect to NO and Cl2, the rate law and the rate constant.
Find the two-third life, t2/3, of a first order reaction in which k = 5.4 × 10−14 s.
The following rate data were obtained at 300 K for the reaction, \[\ce{2A + B -> C + D}\]
| Experiment number | [A] (mol L−1) | [B] (mol L−1) | Rate of formation of D (mol L−1 min−1) |
| 1 | 0.1 | 0.1 | 4.5 × 10−3 |
| 2 | 0.3 | 0.2 | 5.4 × 10−2 |
| 3 | 0.3 | 0.4 | 2.16 × 10−1 |
| 4 | 0.4 | 0.1 | 1.8 × 10−2 |
Calculate the rate of formation of D when [A] = 0.5 mol L−1 and [B] = 0.3 mol L−1.
For the reaction \[\ce{A -> B + C}\], the data given below were obtained. Prove that the reaction is of first order.
| t | 0 | 90 | 180 |
| [A] | 50.8 | 19.7 | 7.62 |
The following data were obtained for the decomposition of phosphine.
| Initial pressure (mm of Hg) | 707 | 500 | 79 |
| Half-life | 84 | 83.8 | 84 |
Find the order of reaction.
State a condition under which a bimolecular reaction is kinetically first order reaction.
How is Half-life related to initial concentration for a second order reaction?
Express the relation between the half-life period of a reactant and its initial concentration for a reaction of nth order.
The reaction \[\ce{A + 2B -> C}\] obeys the equation,
Rate = k[A]1/2 [B]3/2
What is the order of the reaction?
Define temperature coefficient of a reaction.
What is threshold energy?
How is threshold energy related to the activation energy of the reaction?
Define activation energy.
Explain the temperature dependence of the reaction rate on activation energy basis.
Define activation energy.
How does activation of a reaction vary with rise in temperature?
How is the rate constant of a reaction related to its activation energy? Name the equation which gives the relationship.
The following values for the first order reaction rate constants were obtained for a reaction:
| Temp. (K) | k |
| 298 | 3.46 × 10−5 s−1 |
| 308 | 13.50 × 10−5 s−1 |
Calculate the activation energy for the reaction.
The rate of a reaction triples when temperature changes from 20 to 50°C. Calculate the energy of activation for such a reaction.
The rate constant of a first order reaction becomes 6 times when the temperature is raised from 350 K to 410 K. Calculate the energy of activation. (Gas constant, R = 8.314 J K−1 mol−1)
The slope of the line in the graph of log k (k = rate constant) versus `1/T` for a reaction is −5400 K. Calculate the energy of activation for this reaction. (R = 8.314 J K−1 mol−1).
The slope of the line in the graph of log k versus `1/T` for the reaction
\[\ce{N2O -> 2NO2 + \frac{1}{2} O2_{(g)}}\]
is −5000 K. Calculate the energy of activation for the reaction. (R = 8.314 J K−1 mol−1).
The rate constant of a reaction is 0.01439 min−1 at 25°C and its activation energy is 70,000 J mol−1. What is the value of rate constant at 40°C? (Given; R = 8.314 J K−1 mol−1)
The rate constant of a reaction is 1.2 × 10−3 s−1 at 303 K and 2.1 × 10−3 s−1 at 313 K. Calculate the energy of activation of the reaction. (R = 8.314 J K−1 mol−1)
The decomposition of phosphine
\[\ce{4PH3_{(g)} -> P4_{(g)} + 6H2_{(g)}}\]
has the rate law, Rate = k [PH3].
The rate constant is 6.0 × 10−4 s−1 at 300 K and activation energy is 3.05 × 105 J mol−1. What is the value of rate constant at 310 K? (R = 8.314 J K−1 mol−1)
The specific rate constant for a particular reaction is 2.34 × 10−3 mol L−1 s−1 at 370 K and 7.50 × 10−2 mol L−1 s−1 at 400 K. Calculate the activation energy for the reaction. (R = 8.314 JK−1 mol−1).
In some cases it is found that a large number of colliding molecules have energy more than the threshold value, yet the reaction is slow, why?
Define activation energy.
The activation energy of a reaction is 75.2 kJ mol−1 in the absence of a catalyst and 50.14 kJ mol−1 with a catalyst. How many times will the rate of reaction grow in the presence of the catalyst if the reaction proceeds at 25°C? (R = 8.314 JK−1 mol−1).
The first order rate constant for the decomposition of ethyl iodide by the reaction
\[\ce{C2H5I_{(g)} -> C2H4_{(g)} + HI_{(g)}}\]
at 600 K is 1.60 × 10−5 s−l. Its energy of activation is 209 kJ/mol. Calculate the rate constant of the reaction at 700 K.
What do you understand by molecularity of a reaction?
Explain why the reactions having molecularity greater than 2 are rare.
What is the difference between the order of a reaction and the molecularity of a reaction?
What are pseudo-unimolecular reactions and why are they called so? Explain with an example.
What do you understand by the mechanism of a reaction?
The experimentally determined rate law for the reaction
\[\ce{H2O2 + 2H+ + 2I- -> I2 + 2H2O}\]
is found to be
Rate = k [H2O2][I−]
Postulate a mechanism for the reaction if OI− ions have been detected as intermediate during the progress of the reaction.
What is the rate of reaction and the order of reaction if the mechanism of the reaction is
\[\ce{2NO + H2 -> N2 + H2O2}\] (slow)
\[\ce{H2O2 + H2 -> 2H2O}\] (fast)
For the reaction \[\ce{NO2_{(g)} + CO_{(g)} -> CO2_{(g)} + NO_{(g)}}\], the experimentally determined rate expression below 440 K is
Rate = k [NO2]2
What mechanism can be proposed for the above reaction?
Differentiate between rate of reaction and rate constant of a reaction.
Distinguish between Order and Molecularity of reaction.
Nitric oxide reacts with hydrogen to give nitrogen and water.
\[\ce{2NO + 2H2 -> N2 + 2H2O}\]
The kinetics of this reaction is explained by the following steps:
- \[\ce{2NO + H2 -> N2 + H2O2}\] (slow)
- \[\ce{H2O2 + H2 -> 2H2O}\]
What is the predicted rate law?
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics VERY SHORT ANSWER TYPE QUESTIONS [Pages 262 - 263]
Define the rate of a reaction.
What are the units of rate of a reaction?
What are the units of the rate of a reaction involving gaseous reactants and products?
Define average rate of reaction.
Express the average rate of the reaction, \[\ce{H2_{(g)} + I2_{(g)} -> 2HI_{(g)}}\], in terms of all reactants and products.
Define instantaneous rate of reaction.
Which type of rate is given by the slope of conc. of reactants vs time curve at a particular point?
Why does the rate of a reaction usually increase on increasing the concentration of reactants?
Why do finely divided solid reactants react faster as compared to massive reactants?
What do you understand by rate law of a reaction?
Define rate constant.
What will be the effect of temperature on rate constant?
What are the units of rate constant for a reaction of order n?
Define the following term:
Order of a reaction
Can the order of a reaction be negative?
What is the order of reaction if the rate law is
Rate = k [A]1/2 [B]2 [C]0?
The reaction, \[\ce{A + B -> Products}\], follows the rate law Rate = k[A][B]2.
What will be the effect on the rate if the concentration of A is doubled and that of B is halved?
What are the units of rate constant for a second order reaction?
The rate constant of a reaction possesses the units of time−1. What is the order of reaction?
For the reaction \[\ce{2X -> X2}\], rate of reaction becomes three times when the concentration of X is increased by 27 times. What is the order of reaction?
For the reaction, \[\ce{A + B -> Products}\], the rate becomes doubled on doubling the concentration of A but remains unchanged on doubling the concentration of B. What is the rate law?
What are the units of rate constant for a gaseous second order reaction?
The reaction, \[\ce{A -> Products}\], is a zero order reaction. To what extent will the rate of the reaction change if the concentration of A is trebled?
What are the units of rate constant for a third order reaction?
The units of rate constant for a reaction are mol L−1 s−1. What is the order of the reaction?
The value of rate constant for the reaction, \[\ce{A -> Products}\], is 5.6 × 10−3 mol−1 L s−1. How will the rate change when the concentration of A is halved?
How is the half-life of a first order reaction related to the concentration of the reactant?
What is the shape of the curve obtained on plotting log10[A] against t for a first order reaction?
What will be the order of reaction if the plot of log10[A] vs time is a straight line with a negative slope?
Write two factors that affect the rate of reaction.
How is specific reaction rate different from the reaction rate?
How is the slope of the straight line in the plot of log10C vs time for a first order reaction related to the rate constant of the reaction?
How is the rate constant of a first order reaction related to the half-life of the reaction?
If [A]0 is the initial concentration of a reactant taking part in a first order reaction, how much of the reactant will be left after n half-lives?
How is the progress of a reaction monitored for its kinetic study?
What is the order of reaction if the plot of rate vs [A]2 is a straight line?
Write the equation for integrated rate law for a first order reaction.
How is the half-life of a reaction of order n related to the initial concentration of the reactant?
Define temperature coefficient of a reaction.
Define threshold energy of a reaction.
Define activation energy.
The plot of log k vs X is linear with a slope = `(-E_a)/(2.303 R)`. What is X?
Define molecularity of a reaction.
Can the molecularity of a complex reaction be derived from the stoichiometry of the reaction?
Which step in a complex reaction does determine the overall rate of the reaction?
Define pseudo-unimolecular reactions.
Give an example of pseudo-unimolecular reactions.
Answer the following in one or two sentences.
What is the rate-determining step?
A reaction proceeds in three steps, one is slow while the other two are fast. Which step will determine the rate of the reaction?
The rate law of a reaction is given by
Rate = k[A][B]2
Which of the following will react fastest?
1 mol of A and 2 mol of B in 1 L vessel.
2 mol of A and 1 mol of B in 500 ml vessel.
3 mol of A and 2 mol of B in 2 L vessel.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics SHORT ANSWER TYPE QUESTIONS [Pages 263 - 264]
Define chemical kinetics.
What is the importance of kinetic study of a chemical reaction?
Define the rate of a reaction.
What do you understand by average rate?
What do you understand by instantaneous rate?
Express the average rate of the following reaction in terms of all reactants and products.
\[\ce{2A + B -> 3C + 4D}\]
Express the instantaneous rate of the following reaction in terms of all reactants and products.
\[\ce{2A + B -> 3C + 4D}\]
How is the instantaneous rate of a reaction determined at a particular instant? Explain graphically.
Write two factors that affect the rate of reaction.
How does a catalyst increase the rate of a reaction?
Why do finely divided solid reactants react faster as compared to massive reactants?
Why does the rate of a reaction usually increase on increasing the concentration of reactants?
What do you understand by rate law of a reaction?
What is rate law significance?
Explain the rate of reaction
\[\ce{2N2O5 -> 4NO2 + O2}\]
using
- concentration change of N2O5,
- concentration change of O2.
What is specific reaction rate?
Derive specific reaction rate units for a reaction of order n.
What is the rate constant?
Write the unit of rate constant [k] for the first order reaction.
What are the units of rate constant for a second order reaction?
Derive rate constant units for third order reactions.
What are the characteristics of rate constant?
Differentiate between rate of reaction and rate constant of a reaction.
What do you understand by the order of a reaction?
How is order of a reaction obtained from the rate law of a reaction?
For the reaction \[\ce{aA + bB + cC -> Products}\],
following conclusions have been derived on the basis of experimental studies:
- The rate of reaction remains unchanged on doubling the concentration of A.
- The rate of reaction becomes half on doubling the concentration of B.
- The rate of reaction increases 8 times on doubling the concentration of C.
Derive the rate law of the reaction and determine its order.
What is the difference between the order of a reaction and the molecularity of a reaction?
For the following reaction, state the order with respect to the reactant and the overall order.
\[\ce{2NO2_{(g)} -> 2NO_{(g)} + O2_{(g)}}\] Rate = k[NO2]2
For the following reaction, state the order with respect to the reactant and the overall order.
\[\ce{H2O2 + 3I- + 2H+ -> 2H2O + I_3^-}\] Rate = k[H2O2][I−]
For the following reactions, state the order with respect to each reactant and the overall order.
\[\ce{CHCl3_{(g)} + Cl2_{(g)} -> CCl_{(g)} + HCl_{(g)}}\] Rate = k [CHCl3][Cl2]1/2
Write the unit of rate constant [k] for the first order reaction.
Give three examples of first order reactions and state their rate expressions.
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
State the order of reaction whose rate constant possess the following unit:
atm−2 s−1
State the order of reaction whose rate constant possess the following unit:
hour−1
The units of rate constant for a reaction are mol L−1 s−1. What is the order of the reaction?
Answer the following in brief.
Derive the integrated rate law for the first-order reaction.
How is integrated rate equation for a first order reaction related to the rate constant?
Derive the rate equation for the rate constant of a first order reaction and show that the time required for the completion of half of the first order reaction is independent of initial concentration.
Derive the rate equation for the rate constant of a first order reaction and show that the time required for the completion of half of the first order reaction is independent of initial concentration.
Derive the relation Ct = C0 e−kt for a first order reaction.
What is the graphical behaviour of a first order reaction on plotting rate of reaction against time
What is the graphical behaviour of a first order reaction on plotting [A] against t?
What is the shape of the curve obtained on plotting log10[A] against t for a first order reaction?
Prove that `[A] = [A]_0 (1/2)^n`, where [A]0 = initial concentration, [A] = concentration after time t, and n = number of half-lives.
What are the main characteristics of a first order reaction?
Which property is used to study the kinetics of the inversion of cane sugar? How is the property helpful in determining the order of the reaction?
Describe the graphical representation of first order reaction.
By taking a suitable example, describe the initial rate method for the determination of the order of a reaction.
Describe the integrated rate law method for determining the order of a first order reaction.
How is the half-life of a reaction of order n related to the initial concentration of the reactant?
Describe the half-life method for determining the order of a first order reaction.
What is half-life of a reaction?
Answer the following in brief.
Obtain the relationship between the rate constant and half-life of a first-order reaction.
Explain the following:
One gram of pulverised wood burns faster than a one gram piece of wood.
Explain the following:
An increase of 10 K in temperature rarely doubles the kinetic energy of particles but this increase in temperature may be enough to double the rate of reaction.
What is the effect of temperature on the rate of a chemical reaction?
Define temperature coefficient of a reaction.
Explain the following term:
Energy barrier
Explain the following term:
Orientation barrier.
What is threshold energy?
Answer the following in one or two sentences
What is the activation energy of a reaction?
Why do different reactions occur at different rates?
How would you account for the increase in the reaction rate on increasing the temperature on the basis of collision theory?
State Arrhenius equation and explain the terms involved in it.
How would you determine the activation energy of a reaction with the help of Arrhenius equation?
What do you understand by molecularity of a reaction?
How is the molecularity of a reaction determined for a complex reaction?
Distinguish between Order and Molecularity of reaction.
Explain with suitable example, how the molecularity of a reaction is different from the order of the reaction.
What do you understand by molecularity of a reaction?
What do you understand by the order of a reaction?
Explain why molecularity and order of a reaction values are different for the following reaction:
\[\ce{CH3COOC2H5 + H2O -> CH3COOH + C2H5OH}\]
What are pseudo-unimolecular reactions and why are they called so? Explain with an example.
Give two examples of pseudo-unimolecular reactions.
How would you define the molecularity of a complex reaction? Explain with an example.
Answer the following in one or two sentences.
What is the rate-determining step?
How is the rate determining step helpful in postulating the mechanism of a reaction? Explain with an example.
The rate law of the reaction,
\[\ce{2NO2_{(g)} + F2_{(g)} -> 2NO2F_{(g)}}\], has been found to be Rate = k[NO2] [F2]. During the progress of the reaction, the transient existence of F atom has been confirmed. Postulate a mechanism for the reaction.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics LONG ANSWER TYPE QUESTIONS [Page 265]
Describe the method used to determine the rate of a reaction.
Write two factors that affect the rate of reaction.
What do you understand by rate law of a reaction?
What is the rate constant?
Derive the units of rate constant for the reactions of various orders.
What do you understand by the order of a reaction?
Distinguish between Order and Molecularity of reaction.
Describe a method which can be used to determine the order of a first order reaction.
Define first-order reaction.
Answer the following in brief.
Derive the integrated rate law for the first-order reaction.
Describe the graphical representation of first order reaction.
Define half-life period.
Derive an expression for the relation between half-life and rate constant for first-order reaction.
Taking the example of decomposition of N2O5, describe how a first order reaction is studied kinetically.
Which observable property is used in the kinetic study of the following first order reaction and why?
\[\ce{H2O2_{(aq)} ->[Pt] H2O_{(l)} + \frac{1}{2} O2_{(g)}}\]
Which observable property is used in the kinetic study of the following first order reaction and why?
\[\ce{CH3COOC2H5 + H2O ->[H+] CH3COOH + C2H5OH}\]
Which observable property is used in the kinetic study of the following first order reaction and why?
\[\ce{C12H22O11 + H2O ->[H+] C6H12O6 + C6H12O6}\]
Describe the integrated rate law method for determining the order of a reaction.
Describe the half-life method for determining the order of a reaction.
Describe the collision theory of reaction rate.
How would you account for the increase in the reaction rate on increasing the temperature on the basis of collision theory?
Answer the following in one or two sentences
What is the activation energy of a reaction?
How would you determine the activation energy of a reaction with the help of Arrhenius equation?
State Arrhenius equation and explain the terms involved in it.
On the basis of Arrhenius equation, how would you determine the energy of activation of a reaction?
On the basis of Arrhenius equation, how would you determine the temperature coefficient of a reaction?
Define molecularity of a reaction.
Show that the molecularity of a complex reaction cannot be determined by the stoichiometry of the reaction.
What do you understand by the mechanism of a reaction?
How is the rate determining step helpful in postulating the mechanism of a reaction? Explain with an example.
Write two factors that affect the rate of reaction.
Write Arrhenius equation and explains the terms involved.
The rate constant of any reaction increases linearly with increase in temperature. Explain.
The rate constant of any reaction is proportional to the concentration of the reactants. Comment.
The initial rate of a reaction \[\ce{A + B -> products}\] is doubled when the initial concentration of A is doubled and increases eight fold when the initial concentration of both A and B are doubled. State the order of the reaction with respect to A and with respect to B. Write the rate equation.
Draw a graph which is used to calculate the activation energy of a reaction. Give the appropriate expressions used to calculate the activation energy graphically.
Give one example of homogeneous catalysis.
Give four examples of heterogeneous catalysis.
Define molecularity of a reaction.
Distinguish between Order and Molecularity of reaction.
Write two factors that affect the rate of reaction.
Complete the following statement by selecting the correct alternative from the choices given.
The reaction between X and Y is first order with respect to X and second order with respect to Y. If the concentration of X is halved and the concentration of Y is doubled the rate of reaction will be ______.
same as the initial value.
three times the initial value.
double the initial value.
half the initial value.
Fill in the blanks:
The half-life period of a ______ order reaction is ______ on the concentration of the reactant.
The rate constant (k) of a first-order reaction is 4.5 × 10−2 sec−1. What will be the time required for the initial concentration of 0.4 M of the reactant to be reduced to 0.2 M?
An alkyl bromide undergoes reaction in the alkaline medium to form the corresponding alcohol. When the concentration of the alkyl bromide is doubled keeping the concentration of the alkali constant, the rate of the reaction is doubled. When the concentration of the alkali is doubled keeping the concentration of the alkyl bromide constant, the rate of the reaction remains the same.
Write the mechanistic steps for the reaction and state the type of the reaction and the nature of the reagent.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics INTEGER TYPE QUESTIONS [Page 265]
The reaction \[\ce{2A + B2 -> 2AB}\] is an elementary reaction. For a certain quantity of reactants, if the volume of the reaction vessel is reduced by a factor of 3, the rate of the reaction increases by a factor of ______ (Round off to the nearest integer).
A reaction has a half-life of 1 min. The time required for 99.9% completion of the reaction is ______ min. (Round off to the nearest integer).
[Use In 2 = 0.69, In 10 = 2.3]
\[\ce{2NO_{(g)} + Cl2_{(g)} <=> 2NOCl_{(s)}}\]
This reaction was studied at −10°C and the following data was obtained
| Run | [NO]0 | [Cl2]0 | r0 |
| 1 | 0.10 | 0.10 | 0.18 |
| 2 | 0.10 | 0.20 | 0.35 |
| 3 | 0.20 | 0.20 | 1.40 |
[NO]0 and [Cl2]0 are the initial concentrations and r0 is the initial reaction rate. The overall order of the reaction is ______ (Round off to the nearest integer).
The decomposition of formic acid on gold surface follows first order kinetics. If the rate constant at 300 K is 1.0 × 10−3 s−1 and the activation energy Ea = 11.488 kJ mol−1, the rate constant at 200 K is ______ × 10−5 s−1 (Round off to the nearest integer) (Given R = 8.314 JK−1 mol−1).
The half-life for the decomposition of gaseous compound A is 240 s when the gaseous pressure was 500 torr initially. When the pressure was 250 torr, the half-life was found to be 4 min. The order of the reaction is ______ (Nearest integer).
For the decomposition of azomethane
\[\ce{CH3N2CH3_{(g)} -> CH3CH3_{(g)} + N2_{(g)}}\] is a first order reaction, the variation in partial pressure with time at 600 K is given by
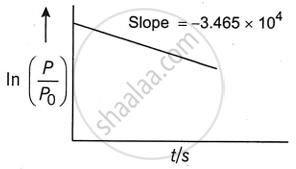
The half-life of the reaction is ______ × 10−5 s (Nearest integer).
For a reaction, given below is the graph of ln K vs `1/T`. The activation energy for the reaction is equal to ______ Cal mol−1 (Nearest integer).
(Given R = 2 Cal K−1 mol−1).
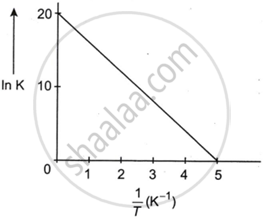
For the given first order reaction, \[\ce{A -> B}\]. the half-life of the reaction is 0.3010 min. The ratio of the initial concentration of reactant to the concentration of reactant at time 2.0 min will be equal to ______ (Nearest integer).
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 266 - 270]
Choose the correct option in the following questions.
For the hypothetical reaction, \[\ce{2A -> 3C}\], the reaction rate r is given by ______.
`r = -(d[A])/(dt)`
`r = -1/2 (d[A])/(dt)`
`r = -1/3 (d[A])/(dt)`
`r = (d[A])/(dt)`
The rate constant of a reaction depends on ______.
initial concentration of the reactants
time of reaction
temperature
extent of reaction
The rate of the reaction, \[\ce{A + B + C -> Products}\], is given by \[\ce{- \frac{d[A]}{dt} = k [A]^{1/2} [B]^{1/3} [C]^{1/4}}\].
The order of reaction is ______.
`1/2`
`13/12`
1
2
The rate of reaction, \[\ce{A + B -> Products}\], is given by the equation, r = k [A] [B]. If B is taken in large excess, the order of reaction would be ______.
2
1
0
unpredictable
The inversion of cane sugar is represented as \[\ce{C12H22O11 + H2O -> C6H12O6 + C6H12O6}\].
It is a ______.
second order reaction
unimolecular reaction
pseudo-unimolecular reaction
zero order reaction
The rate of the reaction, \[\ce{2NO + O2 -> 2NO2}\], at 25°C is 0.028 mol L−1 s−1. The experimental rate is given by r = k [NO]2 [O2].
If the initial concentration of the reactants are [O2] = 0.040 mol L−1 and [NO] = 0.010 mol L−1, the rate constant of the reaction is ______.
7.0 × 10−2 mol−1 L s−1
7.0 × 10−4 mol−2 L2 s−1
7.0 × 102 mol−2 L2 s−1
7.0 × 103 mol−2 L2 s−1
Select the rate law that corresponds to the data shown for the following reaction:
\[\ce{A + B -> C}\]
| Expt. no | [A]0 | [B]0 | Initial Rate |
| 1. | 0.012 | 0.035 | 0.10 |
| 2. | 0.024 | 0.070 | 0.80 |
| 3. | 0.024 | 0.035 | 0.10 |
| 4. | 0.012 | 0.070 | 0.80 |
Rate = k [B]3
Rate = k [B]4
Rate = k [A] [B]3
Rate = k [A]2 [B]2
The rate law for the reaction
\[\ce{RCl + NaOH_{(aq)} -> ROH + NaCl}\]
is given by, Rate = k [RCl]. The rate of reaction will be ______.
unaffected by increasing the temperature of the reaction.
doubled on doubling the concentration of NaOH.
halved on reducing the concentration of NaOH to one half.
halved on reducing the concentration of RCl to one half.
The half-life period of a first order reaction is 10 minutes. The time required for the concentration of the reactant to change from 0.08 M to 0.02 M is ______.
10 min
20 min
30 min
40 min
The first order rate constant for the decomposition of N2O5 is 6 × 10−4 s−1. The half-life period for this decomposition is ______.
1155 s
1117 s
223.4 s
160.9 s
For a zero order reaction ______.
t1/2 ∝ a
`t_(1//2) prop 1/a`
t1/2 ∝ a2
`t_(1//2) prop 1/a^2`
The rate of a gaseous reaction is given by the expression, r = k [A] [B]. If the volume of the reaction vessel is suddenly reduced to one fourth of the initial volume, the reaction rate relative to the original rate will be ______.
`1/10`
`1/8`
8
16
The ratio of the time required for `3/4`th of the reaction of first order to complete to that required for half of the reaction is ______.
4 : 3
3 : 2
2 : 1
1 : 2
The slope of the line obtained on plotting log10 k against `1/T` is equal to ______.
`E_a/R`
`- E_a/R`
`E_a/(2.303 R)`
`- E_a/(2.303 R)`
If the plot of log10 [A] against t is a straight line with a negative slope, the reaction is of ______.
zero order
first order
second order
third order
Time required to decompose half of the substance for nth order reaction is inversely proportional to ______.
an + 1
an − 1
an − 2
an
If the initial concentration of the reactant is doubled, time for half reaction is also doubled; the order of reaction is ______.
zero
first
second
third
The rate constant, the activation energy and the Arrhenius parameter of a chemical reaction at 25°C are 3.0 × 10−4 s−1, 104.4 kJ mol−1 and 6.0 × 1014 s−1 respectively. The value of rate constant at \[\ce{T -> \infty}\] is ______.
2.0 × 1018 s−1
6.0 × 1014 s−1
infinity
3.6 × 1030 s−1
Which of the following statements is not correct?
A catalyst can not be recovered unchanged chemically at the end of reaction.
A catalyst usually does not initiate a chemical reaction.
A very small amount of catalyst is sufficient to catalyse the reaction.
The action of a catalyst is specific.
Diazonium salt decomposes as
\[\ce{C6H5N{^+_2}Cl- -> C6H5Cl + N2}\]
At 0°C, the evolution of N2 becomes two times faster when the initial concentration of the salt is doubled. Therefore it is ______.
a first order reaction
a second order reaction
independent of the initial cone, of the salt
a zero order reaction
In a reaction, \[\ce{A -> B}\], the rate of reaction increases two times on increasing the concentration of reactants four times. The order of reaction is ______.
0
2
`1/2`
4
Inversion of sugar follows first order rate equation which can be followed by noting the change in rotation of the plane of polarisation of light in the polarimeter. If r∞, rt and r0 are the rotations at t = ∞, t = t and t = 0, then first order reaction can be written as ______.
`k =1/t log_e (r_t - r_infty)/(r_0 - r_infty)`
`k =1/t log_e (r_0 - r_infty)/(r_t - r_infty)`
`k =1/t log_e (r_infty - r_0)/(r_infty - r_t)`
`k =1/t log_e (r_infty - r_t)/(r_infty - r_0)`
The minimum energy required for molecules to enter into chemical reaction is called ______.
kinetic energy
potential energy
threshold energy
activation energy
The number of molecules of the reactants taking part in a single step of the reaction tells about ______.
molecularity of the reaction
mechanism of the reaction
order of reaction
all of the above
The elementary step of the reaction,
\[\ce{2Na + Cl2 -> 2NaCl}\], is found to follow a third order kinetics. The molecularity of the reaction is ______.
1
2
3
4
In many reactions, the reaction proceeds in a sequence of steps. The overall rate of such reactions is determined by ______.
slowest step
fastest step
molecularity of the steps
order of different steps
According to Arrhenius equation, the rate constant of a chemical reaction is equal to ______.
`A e^(-E_a//RT)`
`A e^(E_a//RT)`
`A e^(-RT//E_a)`
`A e^(RT//E_a)`
75% of a first order reaction was completed in 32 minutes; when was 50% of the reaction completed?
4 min
8 min
24 min
16 min
The rate of the reaction,
\[\ce{CCl3CHO + NO -> CHCl3 + NO + CO}\]
is given by the equation, rate = k [CCl3CHO)[NO]. If concentration is expressed in mol/litre, the units of k are ______.
mol−2 L2 s−1
mol L−1 s−1
mol−1 L s−1
s−1
The rate law for the chemical reaction,
\[\ce{2NO2Cl -> 2NO2 + Cl2}\], is Rate = k [NO2Cl].
The rate determining step is:
\[\ce{2NO2Cl -> 2NO2 + Cl2}\]
\[\ce{NO2 + Cl2 -> NO2Cl + Cl}\]
\[\ce{NO2Cl + Cl -> NO2 + Cl2}\]
\[\ce{NO2Cl -> NO2Cl}\]
For the reaction,
\[\ce{N2O5_{(g)} -> 2NO2_{(g)} + \frac{1}{2} O2_{(g)}}\]
the value of rate of disappearance of N2O5 is given as 6.25 × 10−3 mol L−1 s−1. The rate of formation of NO2 and O2 is given respectively as:
6.25 × 10−3 mol L−1 s−1 and 6.25 × 10−3 mol L−1 s−1
1.25 × 10−2 mol L−1 s−1 and 3.125 × 10−3 mol L−1 s−1
6.25 × 10−3 mol L−1 s−1 and 3.125 × 10−3 mol L−1 s−1
1.25 × 10−2 mol L−1 s−1 and 6.25 × 10−3 mol L−1 s−1
For an endothermic reaction, energy of activation is Ea and enthalpy of reaction is ΔH (both of these in kJ/mol). Minimum value of Ea will be ______.
less than ΔH
equal to ΔH
more than ΔH
equal to zero
Which one of the following statements for the order of a reaction is incorrect?
Order is not influenced by stoichiometric coefficient of the reactants.
Order of reaction is sum of power to the concentration terms of reactants to express the rate of reaction.
Order of reaction is always whole number.
Order can be determined only experimentally.
In a reaction, \[\ce{A + B -> Product}\], rate is doubled when the concentration of B is doubled, and rate increases by a factor of 8 when the concentrations of both the reactants (A and B) are doubled. Rate law for the reaction can be written as ______.
Rate = k[A][B]2
Rate = k[A]2[B]2
Rate = k[A][B]
Rate = k[A]2[B]
In a zero order reaction.for every 10° rise of temperatue, the rate is doubled. If the temperature is increased from 10°C to 100°C, the rate of the reaction will become ______.
256 times
512 times
64 times
128 times
The rate of a reaction doubles when its temperature changes from 300 K to 310 K. Activation energy of such a reaction will be ______. (R = 8.314 JK−1 mol−1 and log 2 = 0.301)
53.6 kJ mol−1
48.6 kJ mol−1
58.5 kJ mol−1
60.5 kJ mol−1
For the non-stoichiometric reaction, \[\ce{2A + B -> C + D}\], the following kinetic data were obtained in three separate experiments, all at 298 K.
| Initial concentration (A) |
Initial concentration (B) |
Initial rate of formation of C (mol L−1 s−1) |
| 0.1 M | 0.1 M | 1.2 × 10−3 |
| 0.1 M | 0.2 M | 1.2 × 10−3 |
| 0.2 M | 0.1 M | 2.4 × 10−3 |
The rate law for the formation of C is:
`(dc)/(dt) = k[A][B]`
`(dc)/(dt) = k[A]^2[B]`
`(dc)/(dt) = k[A][B]^2`
`(dc)/(dt) = k[A]`
Higher order (> 3) reactions are rare due to ______.
low probability of simultaneous collision of all the reacting species.
increase in entropy and activation energy as more molecules are involved.
shifting of equilibrium towards reactants due to elastic collisions.
loss of active species on collision.
Decomposition of H2O2 follows a first order reaction. In fifty minutes the concentration of H2O2 decreases from 0.5 to 0.125 M in one such decomposition. When the concentration of H2O2 formation of O2 will be ______.
6.93 × 10−2 mol min−1
6.93 × 10−4 mol min−1
2.66 L min−1 at STP
1.34 × 10−2 mol min−1
According to the Arrhenius equation ______.
a high activation energy usually implies a fast reaction.
rate constant increases with increase in temperature. This is due to a greater number of collisions whose energy exceeds the activation energy.
higher the magnitude of activation energy, stronger is the temperature dependence of the rate constant.
the pre-exponential factor is a measure of the rate at which collisions occur, irrespective of their energy.
The rate of a first order reaction is 0.04 mol l−1 s−1 at 10 seconds and 0.03 mol l−1 s−1 at 20 seconds after initiation of the reaction. The half-life period of the reaction is ______.
24.1 s
34.1 s
44.1 s
54.1 s
A first order reaction has a specific reaction rate of 10−2 s−1. How much time will it take for 20 g of the reactant to reduce to 5 g?
238.6 second
138.6 second
346.5 second
693.0.second
Two reactions R1 and R2 have identical pre-exponential factors. Activation energy of R1 exceeds that of R2 by 10 kJ mol−1. If k1 and k2 are rate constants for reactions R1 and R2 respectively at 300 K then ln (k2/k1) is equal to ______. (R = 8.314 J mol−1 K−1)
6
4
8
12
Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic reaction?
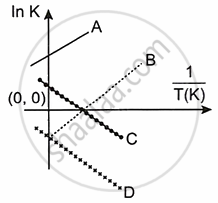
A and B
B and C
C and D
A and D
At 518°C, the rate of decomposition of a sample of gaseous acetaldehyde, initially at a pressure of 363 Torr, was 1.00 Torr s−1 when 5% had reacted and 0.5 Torr s−1 when 33% had reacted. The order of the reaction is:
2
3
1
0
The correct difference between first and second order reactions is that
A first-order reaction can be catalysed; a second-order reaction cannot be catalysed.
The half-life of a first-order reaction does not depend on [A0]; the half-life of a second-order reaction does depend on [A0].
The rate of a first-order reaction does not depend on reactant concentrations; the rate of a second-order reaction does depend on reactant concentrations.
The rate of a first-order reaction does depend on reactant concentrations; the rate of a second-order reaction does not depend on reactant concentrations.
When initial concentration of the reactant is doubled, the half-life period of a zero-order reaction ______.
is tripled
is doubled
is halved
remains unchanged
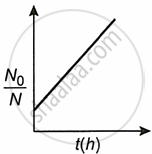
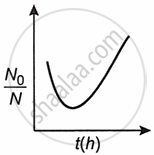
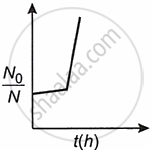
A bacterial infection in an internal wound grows as N'(t) = N0 exp(t), where the time t is in hours. A dose of antibiotic, taken orally, needs 1 hour to reach the wound. Once it reaches there, the bacterial population goes down-as `(dN)/(dt)` = −5 N2. What will be the plot of `N_0/N` vs t after 1 hour?
For the reaction of H2 with I2, the rate constant is 2.5 × 10−4 dm3 mol−1 s−1 at 327°C and 1.0 dm3 mol−1 at 527°C. The activation energy for the reaction, in kJ mol−1 is:
(R = 8.314 JK−1 mol−1)
59
166
72
150
A first order reaction has a rate constant of 2.303 × 10−3 s−1. The time required for 40 g of this reactant to reduce to 10 g will be ______.
602 s
230.3 s
301 s
2000 s
For a reaction, activation energy Ea = 0 and the rate constant at 200 K is 1.6 × 106 s−1. The rate constant at 400 K will be ______.
[Given that gas constant, R = 8.314 J K−1 mol−1]
3.2 × 106 s−1
3.2 × 104 s−1
1.6 × 106 s−1
3.2 × 103 s−1
If the rate constant for a first-order reaction is k, the time (t) required for the completion of 99% of the reaction is given by:
t = 0.693/k
t = 6.909/k
t = 4.606/k
t = 2.303/k
For the chemical reaction,
\[\ce{N2_{(g)} + 3H2_{(g)} <=> 2NH3_{(g)}}\]
The correct option is:
\[\ce{-\frac{1}{3} \frac{d[H2]}{dt} = -\frac{1}{2} \frac{d[NH3]}{dt}}\]
\[\ce{-\frac{d[N2]}{dt} = 2 \frac{d[NH3]}{dt}}\]
\[\ce{-\frac{d[N2]}{dt} = \frac{1}{2} \frac{d[NH3]}{dt}}\]
\[\ce{3 \frac{d[H2]}{dt} = 2 \frac{d[NH3]}{dt}}\]
An increase in the concentration of the reactants of a reaction leads to change in ______.
activation energy
heat of reaction
threshold energy
collision frequency
The rate constant for a first order reaction is 4.606 × 10−3 s−1. The time required to reduce 2.0 g of the reactant to 0.2 g is ______.
100 s
200 s
500 s
1000 s
It is true that ______.
A first order reaction is always a single step reaction.
A second order reaction is always a multistep reaction.
A zero order reaction is a single step reaction.
A zero order reaction is a multistep reaction.
A flask contains a mixture of compounds A and B. Both compounds decompose by first-order kinetics. The half-lives for A and B are 300 s and 180 s, respectively. If the concentrations of A and B are equal initially, the time required for the concentration of A to be four times that of B (in s) is ______. (Use ln 2 = 0.693)
120
300
180
900
For the following reaction
\[\ce{2X +Y ->[K] P}\]
The rate of reaction is \[\ce{\frac{d[P]}{dt} = K[X]}\]. Two moles of X are mixed with one mole of Y to make 1.0 L of solution. At 50 s, 0.5 mole of Y is left in the reaction mixture. The correct statement (s) about the reaction is (are)
(Use In 2 = 0.693).
The rate constant K of the reaction is 13.86 × 10−4 s−1.
Half-life of ‘X’ is 50 s.
At 50 s, \[\ce{- \frac{d[X]}{dt}}\] = 13.86 × 10−3 mol L−1 s−1.
At 100 s, \[\ce{- \frac{d[Y]}{dt}}\] = 3.46 × 10−3 mol L−1 s−1.
The given graph is a representation of kinetics of a reaction.

The y and x axes for zero and first order reactions respectively are
zero order (y = concentration and x = time)
first order (y = rate constant and x = concentration)zero order (y = rate and x = concentration)
first order (y = t1/2 and x = concentration)zero order (y = rate and x = concentration)
first order (y = rate and x = t1/2)zero order (y = concentration and x = time)
first order (y = t1/2 and x = concentration)
For a first order reaction,
\[\ce{A -> products}\], initial concentration of A is 0.1 M, which becomes 0.001 M after 5 minutes. Rate constant for the reaction in min−1 is ______.
0.9212
0.4606
0.2303
1.3818
At 30°C, the half-life for the decomposition of AB2 is 200 s and is independent of the initial concentration of AB2. The time required for 80% of the AB2 to decompose is ______.
(Given log 2 = 0.30, log 3 = 0.48)
200 s
323 s
467 s
532 s
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics FILL IN THE BLANKS TYPE QUESTIONS [Page 271]
For the reaction, \[\ce{2N2O5_{(g)} -> 4NO2_{(g)} + O2_{(g)}}\]; ______ × Rate of disappearance of N2O5 = ______ × Rate of formation of NO2.
The rate of change of concentration of any of the reactants or any of the products at a particular instant is called ______ rate of the reaction.
A chemical reaction involving covalent reactants involves a ______ of bonds and is ______.
A catalyst forms an ______ at ______ potential energy and thus ______ the activation energy of the reaction.
The experimentally observed dependence of the reaction rate on the concentration of reactants is expressed in the form of ______.
The rate constant of a reaction is equal to the rate of reaction when the concentration of each ______ is ______.
The units of rate constant for a first order reaction are ______.
The units of rate constant for a reaction of order 3 are ______.
For a first order reaction, k = `2.303/t log_10` ______.
The half-life of a first order reaction is ______ of the initial conc. of the reactant.
The slope of the line obtained on plotting log10[A] vs t for a first order reaction is ______.
The half-life of a ______ order reaction is inversely proportional to the initial concentration of the reactant.
The collisions between reacting molecules are effective only when they acquire ______ energy.
According to Arrhenius equation, loge k = loge A − ______.
The reactions which occur in two or more steps are called ______ reactions.
The overall rate of a complex reaction is governed by the rate of ______ elementary step which is also called ______ step.
Molecularity is a ______ concept and its value is derived from the ______ of reaction.
The first order reactions having molecularity greater than one are called ______ reactions.
When the concentration of a reactant of first order reaction is doubled, the rate becomes ______ times, but for ______ order reaction, the rate remains same.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics ASSERTION-REASON TYPE QUESTIONS [Page 272]
The question given below consist of an Assertion and a Reason. You have to choose the correct answer according to the following clue:
Assertion: Finely divided solid reactants react much faster as compared to massive reactants.
Reason: Finely divided solid reactants possess a larger surface area.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The rate constant does not depend upon the initial concentration of reactants.
Reason: The rate of reaction in the beginning cannot be measured accurately.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The inversion of cane sugar \[\ce{(C12H22O11 + H2O ->[H+] C6H12O6 + C6H12O6)}\] is a pseudo first order reaction.
Reason: The reaction is catalysed by a dilute mineral acid, e.g., HCl.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: For slow reactions, the energies of activation are quite low.
Reason: Every reaction has an energy barrier.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The overall rate of a complex reaction is governed by the rate of the slowest elementary step.
Reason: All the elementary steps have to wait for the occurrence of the slowest step and the rate of overall reaction cannot exceed the rate of the slowest step.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics NUMERICAL PROBLEMS [Pages 272 - 273]
The· reaction, \[\ce{2A + B + C -> D + E}\], is found to be of first order in A, second order in B and zero order in C.
- Give the rate law for the reaction in the form of a differential equation.
- How is the rate affected on increasing the concentration of A, B and C two times?
Decomposition of N2O5(g) into NO2(g) and O2(g) is a first order reaction. If the initial concentration of N2O5(g), i.e., [N2O5] is 0.030 mol L−1, what will be its concentration after 30 minutes? Rate constant of the reaction is 1.35 × 10−4 s−1.
From the following data for decomposition of N2O5 in CCl4 at 48°C, show that the reaction is of first order.
| Time in minutes | 10 | 15 | 20 | 25 | ∞ |
| Volume in mL | 6.30 | 8.90 | 11.40 | 13.50 | 34.75 |
A first order reaction is 50% complete in 30 minutes at 27°C and in 10 minutes at 47°C. Calculate the reaction rate constant at 27°C and the energy of activation of the reaction in kJ/mol.
Two reactions, (i) \[\ce{A -> Products}\], and (ii) \[\ce{B -> Products}\], follow first order kinetics. The rate of reaction (i) is doubled when the temperature is raised from 300 K to 310 K. The half-life for this reaction at 310 K is 30 minutes. At the same temperature, B decomposes twice as fast as A. If the energy of activation for the reaction (ii) is half that of reaction (i) calculate the rate constant of the reaction (ii) at 300 K.
For the reaction
\[\ce{H2O2_{(aq)} + 2H{^+_{(aq)}} + 2I{^-_{(aq)}} -> I2_{(aq)} + H2O}\],
following data are obtained:
| Expt. | [H2O2] | [I−] | [H+] | d[I2]/dt |
| I | 0.01 | 0.01 | 0.10 | 1.75 × 10−6 |
| II | 0.03 | 0.01 | 0.10 | 5.25 × 10−6 |
| III | 0.03 | 0.02 | 0.10 | 1.05 × 10−5 |
| IV | 0.03 | 0.02 | 0.20 | 1.05 × 10−5 |
Determine the rate law and calculate the value of rate constant.
At 380°C, the half-life period for the first order decomposition of H2O2 is 360 minutes. The energy of activation of the reaction is 200 kJ mol−1. Calculate the time required for 75% decomposition at 450°C.
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 × 1013 sec−1 and 98.6 kJ mol−1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K−1 mol−1]
From the following data for the reaction between A and B:
| [A] | [B] | Initial rate (mol L−1 s-1) at | |
| 300 K | 320 K | ||
| 2.5 × 10−4 | 3.0 × 10−5 | 5.0 × 10−4 | 2.0 × 10−3 |
| 5.0 × 10−4 | 6.0 × 10−5 | 4.0 × 10−3 | - |
| 1.0 × 10−3 | 6.0 × 10−5 | 1.6 × 10−2 | - |
Calculate
- the order of the reaction with respect to A and with respect to B,
- the rate constant at 300K,
- the energy of activation, and
- the pre-exponential factor.
For a first order reaction, show that time required for 99% completion is twice the time required for the completion of 90% of reaction.
The half-life period and initial concentration for a reaction are as follows. What is the order of reaction?
| Initial conc. [mol L−1] | 350 | 540 | 158 |
| t1/2 (sec) | 425 | 275 | 941 |
Determine the order of the reaction,
\[\ce{C12H22O11 + H2O ->[H+] C6H12O6 + C6H12O6}\],
from the following data:
| Time (in minutes) | 0 | 30 | 90 | 230 | ∞ |
| Polarimeter reading (in degree) | +46.75 | +41.0 | +30.75 | +12.75 | −18.75 |
Also calculate the rate constant of the reaction.
The rate constant for the first order decomposition of a certain reaction is given by the equation
ln k (s−1) = `14.34 - (1.25 xx 10^4)/T`
Calculate
- the energy of activation,
- the rate constant at 500 K.
- At what temperature will its half-life period be 256 minutes?
Given the temperature coefficient for the hydrolysis of ethyl acetate by NaOH is 1.75. Calculate the activation energy of the reaction.
What will be the initial rate of a reaction if its rate constant is 10−3 min−1 and concentration of reactant is 0.2 mol dm−3. How much of the reactant will be converted into products in 200 minutes?
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics 'NCERT TEXT-BOOK' Exercises [Pages 277 - 283]
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
For the reaction:
\[\ce{2A + B → A2B}\]
the rate = k[A][B]2 with k = 2.0 × 10−6 mol−2 L2 s−1. Calculate the initial rate of the reaction when [A] = 0.1 mol L−1, [B] = 0.2 mol L−1. Calculate the rate of reaction after [A] is reduced to 0.06 mol L−1.
The decomposition of NH3 on platinum surface is zero order reaction. What are the rates of production of N2 and H2 if k = 2.5 × 10−4 mol−1 L s−1?
The decomposition of dimethyl ether leads to the formation of CH4, H2 and CO and the reaction rate is given by Rate = k [CH3OCH3]3/2. The rate of reaction is followed by increase in pressure in a closed vessel, so the rate can also be expressed in terms of the partial pressure of dimethyl ether, i.e., Rate = \[\ce{k(P_{CH_3OCH_3})^{3/2}}\].
If the pressure is measured in bar and time in minutes, then what are the units of rate and rate constants?
Write two factors that affect the rate of reaction.
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is doubled?
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is reduced to half?
What will be the effect of temperature on rate constant?
In a pseudo first order hydrolysis of ester in water, the following results were obtained:
| t/s | 0 | 30 | 60 | 90 |
| Ester/mol L−1 | 0.55 | 0.31 | 0.17 | 0.085 |
- Calculate the average rate of reaction between the time interval 30 to 60 seconds.
- Calculate the pseudo first order rate constant for the hydrolysis of ester.
A reaction is first order in A and second order in B.
- Write differential rate equation.
- How is the rate affected if the concentration of B is tripled?
- How is the rate affected if the concentrations of both A and B are doubled?
In a reaction between A and B, the initial rate of reaction (r0) was measured for different initial concentrations of A and B as given below:
| A/mol L−1 | 0.20 | 0.20 | 0.40 |
| B/mol L−1 | 0.30 | 0.10 | 0.05 |
| r0/mol L−1 s−1 | 5.07 × 10−5 | 5.07 × 10−5 | 1.43 × 10−4 |
What is the order of the reaction with respect to A and B?
The following results have been obtained during the kinetic studies of the reaction:
\[\ce{2A + B -> C + D}\]
| Experiment | [A]/mol L−1 | [B]/mol L−1 | Initial rate of formation of D/mol L−1 min−1 |
| I | 0.1 | 0.1 | 6.0 × 10−3 |
| II | 0.3 | 0.2 | 7.2 × 10−2 |
| III | 0.3 | 0.4 | 2.88 × 10−1 |
| IV | 0.4 | 0.1 | 2.40 × 10−2 |
Determine the rate law and the rate constant for the reaction.
The reaction between A and B is first order with respect to A and zero order with respect to B. Fill in the blanks in the following table:
| Experiment | A/mol L−1 | B/mol L−1 | Initial rate/mol L−1 min−1 |
| I | 0.1 | 0.1 | 2.0 × 10−2 |
| II | ______ | 0.2 | 4.0 × 10−2 |
| III | 0.4 | 0.4 | ______ |
| IV | ______ | 0.2 | 2.0 × 10−2 |
Calculate the half-life of a first order reaction from the rate constant given below:
200 s−1
Calculate the half-life of a first order reaction from the rate constant given below:
2 min−1
Calculate the half-life of a first order reaction from the rate constant given below:
4 year−1
The half-life for radioactive decay of 14C is 5730 years. An archaeological artifact containing wood had only 80% of the 14C found in a living tree. Estimate the age of the sample.
The experimental data for decomposition of N2O5.
\[\ce{2N2O5 -> 4NO2 + O2}\] in gas phase at 318 K are given below:
| t/s | 0 | 400 | 800 | 1200 | 1600 | 2000 | 2400 | 2800 | 3200 |
| 102 × [N2O5]/mol L−1 | 1.63 | 1.36 | 1.14 | 0.93 | 0.78 | 0.64 | 0.53 | 0.43 | 0.35 |
- Plot [N2O5] against t.
- Find the half-life period for the reaction.
- Draw a graph between log [N2O5] and t.
- What is the rate law?
- Calculate the rate constant.
- Calculate the half-life period from k and compare it with (ii).
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
During nuclear explosion, one of the products is 90Sr with half-life of 28.1 years. If 1μg of 90Sr was absorbed in the bones of a newly born baby instead of calcium, how much of it will remain after 10 years and 60 years if it is not lost metabolically.
For a first order reaction, show that time required for 99% completion is twice the time required for the completion of 90% of reaction.
A first order decomposition reaction takes 40 minutes for 30% decomposition. Calculate its t1/2 value.
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data are obtained.
| t (sec) | P(mm of Hg) |
| 0 | 35.0 |
| 360 | 54.0 |
| 720 | 63.0 |
Calculate the rate constant.
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume.
\[\ce{SO2Cl2_{(g)} -> SO2_{(g)} + Cl2_{(g)}}\]
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.5 |
| 2 | 100 | 0.6 |
Calculate the rate of the reaction when total pressure is 0.65 atm.
The rate constant for the decomposition of N2O5 at various temperatures is given below:
| T/°C | 0 | 20 | 40 | 60 | 80 |
| 105 × k/s−1 | 0.0787 | 1.70 | 25.7 | 178 | 2140 |
Draw a graph between ln k and `1/T` and calculate the values of A and Ea. Predict the rate constant at 30º and 50ºC.
The rate constant for the decomposition of hydrocarbons is 2.418 × 10−5 s−1 at 546 K. If the energy of activation is 179.9 kJ/mol, what will be the value of pre-exponential factor?
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with `t_(1/2)` = 3 hours. What fraction of the sample of sucrose remains after 8 hours?
The decomposition of hydrocarbon follows the equation k = `(4.5 xx 10^11 s^-1) e^(-28000 K//T)`
Calculate Ea.
The rate constant for the first order decomposition of H2O2 is given by the following equation:
log k = 14.34 − 1.25 × 104 K/T.
Calculate Ea for this reaction and at what temperature will its half-period be 256 minutes?
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K. If the value of A is 4 × 1010 s−1. Calculate k at 318 K and Ea.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 4 Chemical Kinetics QUESTIONS FROM ISC EXAMINATION PAPERS [Pages 283 - 285]
A first order reaction is 50% complete in 30 minutes at 27°C. Calculate the rate constant of the reaction at 27°C.
Draw a graph which is used to calculate the activation energy of a reaction. Give the appropriate expressions used to calculate the activation energy graphically.
A quantitative relationship between the temperature and rate constant of a reaction is given by ______.
Nernst equation
Arrhenius equation
van’t Hoff equation
Henderson equation
Consider the reaction, \[\ce{A + B -> C + D}\]
The initial rate for different initial concentrations of the reactants are given below:
| S. No. | Initial concentration (mol L−1) | Initial rate (mol L−1 s−1) | |
| A | B | ||
| (i) | 1.0 | 1.0 | 2.0 × 10−3 |
| (ii) | 2.0 | 1.0 | 4.0 × 10−3 |
| (iii) | 4.0 | 1.0 | 8.0 × 10−3 |
| (iv) | 1.0 | 2.0 | 2.0 × 10−3 |
| (v) | 1.0 | 4.0 | 2.0 × 10−3 |
- What are the orders with respect to A and B?
- What is the overall order?
- Write the rate law equation.
- Calculate the rate constant.
- Suggest a possible mechanism.
Complete the following statement by selecting the correct alternative from the choices given:
75% of a first order reaction was completed in 32 minutes; when was 50% of the reaction completed?
4 min
8 min
24 min
16 min
What is meant by promoter?
Give an example of a promoter.
Give one example (equation) of a homogeneously catalysed reaction and name the catalyst.
A study of chemical kinetics of the reaction \[\ce{A + B-> Products}\], gave the following data at 25°C:
| Experiment | [A] | [B] | `bb((d["Products"])/(dt))` |
| 1 | 1.0 | 0.15 | 4.20 × 10−6 |
| 2 | 2.0 | 0.15 | 8.40 × 10−6 |
| 3 | 1.0 | 0.20 | 5.60 × 10−6 |
Find:
- The order of reaction with respect to A.
- The order of reaction with respect to B.
- The rate law.
Fill in the blanks:
A catalyst ______ start a reaction but it can increase the ______ of the reaction.
For reaction \[\ce{2N2O5 -> 2NO2 + O2}\], the rate and rate constants are 1.02 × 10−4 mol litre−1 sec−1 and 3 4 × 10−5 sec−1. respectively. The concentration of N2O5 at that time will be:
1.732 mol L−1
3 mol L−1
1.02 × 10−4 mol L−1
3.2 × 105 mol L−1
Explain graphically how the rate of a reaction changes with every 10°C rise in temperature.
How is the rate constant of a reaction related to its activation energy? Name the equation which gives the relationship.
The half-life period for the decomposition of a substance is 2.5 hours. If the initial weight of the substance is 160 g, how much of the substance will be left after 10 hours?
- Write the mathematical expression relating the variation of rate constant of a reaction with temperature.
- How can you graphically find the activation energy of the reaction from the above expression?
The slope of the line in the graph of log k (k = rate constant) versus `1/T` is −5841. Calculate the activation energy of the reaction.
What is the difference between the order of a reaction and the molecularity of a reaction?
A substance decomposes by following first order kinetics. If 50% of the compound is decomposed in 120 minutes, how long will it take for 90% of the compound to decompose?
What is the order of reaction whose rate constant has the same unit as the rate of reaction?
In a first-order reaction, 10% of the reactant is consumed in 25 minutes. Calculate:
- The half-life period of the reaction.
- The time required for completing 87.5% of the reaction.
The half-life period of a first-order reaction is 20 minutes. The time required for the concentration of the reactant to change from 0.16 M to 0.02 M is ______.
80 minutes
60 minutes
40 minutes
20 minutes
Identify the order of reaction from the following unit of rate constant (k):
mol L−1 sec−1
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
Name the order of reaction which proceeds with a uniform rate throughout.
For the reaction: \[\ce{2H2 + 2NO <=> 2H2O + N2}\], the following rate data was obtained:
| S.No. | [NO] mol L−1 | [H2] mol L−1 | Rate: mol L−1 sec−1 |
| 1 | 0.40 | 0.40 | 4.6 × 10−3 |
| 2 | 0.80 | 0.40 | 18.4 × 10−3 |
| 3 | 0.40 | 0.80 | 9.2 × 10−3 |
Calculate the following:
- The overall order of a reaction.
- The rate law.
- The value of rate constant (k).
Distinguish between Order and Molecularity of reaction.
Name the order of reaction which proceeds with a uniform rate throughout.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
- Write the rate law expression for the reaction \[\ce{A + B + C -> D + E}\], if the order of reaction is first, second and zero with respect to A, B and C respectively.
- How many times the rate of reaction will increase if the concentration of A, B and C are doubled in the equation given in (a) above?
A first order reaction is 40% complete in 50 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
For a reaction, \[\ce{A + B -> C + D}\], the initial rate for different reactions and initial concentrations of reactants are given below:
| S. No. | Initial Conc. | Initial rate (mole L–1 sec–1) | |
| [A] mole L–1 | [B] mole L–1 | ||
| 1. | 1.0 | 1.0 | 2 × 10−3 |
| 2. | 2.0 | 1.0 | 4 × 10−3 |
| 3. | 4.0 | 1.0 | 8 × 10−3 |
| 4. | 1.0 | 2.0 | 2 × 10−3 |
| 5. | 1.0 | 4.0 | 2 × 10−3 |
- What is the overall order of reaction?
- Write the rate law equation.
25% of first order reaction is completed in 30 minutes. Calculate the time taken in minutes for the reaction to go to 90% completion.
Show that for a first-order reaction the time required to complete 75% of reaction is about 2 times more than that required to complete 50% of the reaction.
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
Fill in the blanks:
The half life period of ______ order reaction is ______ of the initial concentration of the reactant.
Answer the following questions:
A first order reaction takes 100 minutes for completion of 60 per cent of reaction. Find the time when 90% of the reaction will be completed.
In a reaction \[\ce{mA + nB -> products}\], when the concentration of A is doubled, the rate of reaction is also doubled. When the concentration of B is doubled, the rate of reaction becomes four times. Find the overall order of reaction.
Answer the following questions:
Write two factors that affect the rate of reaction.
In a first order reaction, 20% of the reactant is consumed in 30 minutes. Calculate the following:
- The half life period of the reaction.
- The time required for completing 93.75% of the reaction.
A first order reaction is 50% complete in 30 minutes at 27°C and the same reaction is again 50% complete in 10 minutes at 47°C. Calculate the activation energy of the reaction.
(R = 8.314 JK−1 mol−1)
Solutions for 4: Chemical Kinetics
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 4 - Chemical Kinetics Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 4 - Chemical Kinetics - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 4 - Chemical Kinetics
Shaalaa.com has the CISCE Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE 4 (Chemical Kinetics) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 4 Chemical Kinetics are Overview of Chemical Kinetics.
Using Nootan केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी solutions Chemical Kinetics exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 4, Chemical Kinetics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी additional questions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.