Advertisements
Advertisements
प्रश्न
How does a catalyst increase the rate of a reaction?
Advertisements
उत्तर
A catalyst is a material that speeds up a reaction while maintaining its amount and chemical composition at the end.
The rate of a reaction is accelerated by a catalyst. This is due to the fact that it offers a different route with a lower energy barrier. A greater number of reactant molecules can pass through the energy barrier as a result of its reduction. The rate of reaction rises as a result.

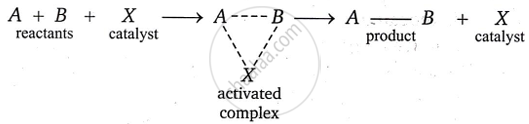
It is thought that the catalyst and reactants combine to generate an active compound. It is at lower potential energy that this activated compound forms. As a result, the reaction’s activation energy drops. As a result, more molecules are able to reach this energy and pass through the energy barrier. As a result, the rate of reaction rises significantly. Let’s look at the hypothetical reaction below, where material X serves as a catalyst.
\[\ce{A + B ->[X] AB}\]
The reaction is supposed to take place as follows.
The figure shows the energy diagram for both the catalysed and uncatalysed processes. The activation energies of uncatalysed forward and backward processes are shown in this figure by Ea(f) and Ea(b), respectively, whereas the comparable values for catalysed reactions are represented by E'a(f) and E'a(b).
