Advertisements
Advertisements
प्रश्न
The reaction between A and B is first order with respect to A and zero order with respect to B. Fill in the blanks in the following table:
| Experiment | A/mol L−1 | B/mol L−1 | Initial rate/mol L−1 min−1 |
| I | 0.1 | 0.1 | 2.0 × 10−2 |
| II | ______ | 0.2 | 4.0 × 10−2 |
| III | 0.4 | 0.4 | ______ |
| IV | ______ | 0.2 | 2.0 × 10−2 |
Advertisements
उत्तर
| Experiment | A/mol L−1 | B/mol L−1 | Initial rate/mol L−1 min−1 |
| I | 0.1 | 0.1 | 2.0 × 10−2 |
| II | 0.2 | 0.2 | 4.0 × 10−2 |
| III | 0.4 | 0.4 | 8.0 × 10−2 |
| IV | 0.1 | 0.2 | 2.0 × 10−2 |
The rate of the reaction is given by,
Rate = k [A]1 [B]0
⇒ Rate = k [A]
From experiment I, we obtain
2.0 × 10−2 mol L−1 min−1 = k (0.1 mol L−1)
⇒ k = 0.2 min−1
From experiment II, we obtain
4.0 × 10−2 mol L−1 min−1 = 0.2 min−1 [A]
⇒ [A] = 0.2 mol L−1
From experiment III, we obtain
Rate = 0.2 min−1 × 0.4 mol L−1
= 0.08 mol L−1 min−1
= 8.0 × 10−2 mol L−1 min−1
From experiment IV, we obtain
2.0 × 10−2 mol L−1 min−1 = 0.2 min−1 [A]
⇒ [A] = 0.1 mol L−1
APPEARS IN
संबंधित प्रश्न
The decomposition of NH3 on platinum surface is zero order reaction. What are the rates of production of N2 and H2 if k = 2.5 × 10−4 mol−1 L s−1?
The decomposition of NH3 on a platinum surface is a zero-order reaction. If the rate constant (k) is 4 x 10-3 ms-1, how long will it take to reduce the initial concentration of NH3 from 0.1 M to 0.064 M?
For which of the following reaction the units of rate constant and rate of the reaction are same?
At high pressure the following reaction is zero order.
\[\ce{2NH3(g) ->[1130 K][Platinum catalyst] N2(g) + 3H2(g)}\]
Which of the following options are correct for this reaction?
(i) Rate of reaction = Rate constant.
(ii) Rate of the reaction depends on concentration of ammonia.
(iii) Rate of decomposition of ammonia will remain constant until ammonia disappears completely.
(iv) Further increase in pressure will change the rate of reaction.
Which of the following graphs is correct for a zero order reaction?
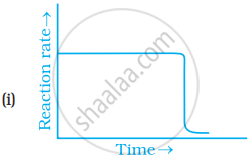

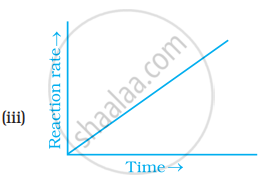

Write the rate equation for the reaction `2A + B -> C` if the order of the reaction is zero.
For a zero order reaction will the molecularity be equal to zero? Explain.
For a zero-order reaction, the plot of [A]t vs t is linear with a ______
Write the expression of integrated rate equation for zero order reaction.
The slope in the plot of [R] Vs. time for a zero-order reaction is ______.
If the initial concentration of substance A is 1.5 M and after 120 seconds the concentration of substance A is 0.75 M, the rate constant for the reaction if it follows zero-order kinetics is ______.
What is zeroth order reaction? Derive its integrated rate Law. What are the units of rate constant?
Derive integrated rate law for zero order reaction.
Write the unit of rate constant of zero order reaction.
What is zero order reaction?
