Advertisements
Advertisements
प्रश्न
Describe the collision theory of reaction rate.
विस्तार में उत्तर
Advertisements
उत्तर
A theory called the Collision Theory of Reaction Rate was put up to explain how temperature affects reaction rates. The following are this theory’s key characteristics.
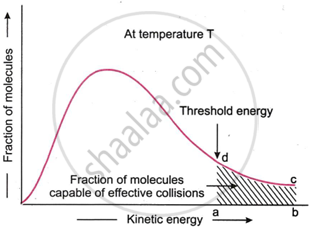
- Only when the molecules of the reactants come into contact with one another, or collide, does a reaction take place.
- Every collision that occurs between the molecules that are interacting is incapable of causing a chemical change. A small percentage of all collisions are actually effective. and cause a reaction that yields the form N goods.
- Only when reacting molecules gain a specific amount of energy do collisions between them become effective. Threshold energy is the bare minimum of energy that interacting molecules need to have in order to collide effectively.
Collisions that result in the creation of products are known as effective collisions. The following two variables control the quantity of effective collisions.
- Energy barrier: As mentioned above, the collisions between the reacting molecules cannot be effective until they possess a definite minimum amount of energy called threshold energy. This indicates that only when two reacting molecules have energy greater than or equal to the threshold energy will their collisions be effective and result in the production of products. This figure shows how energy is distributed among colliding molecules at a specific temperature. The percentage of molecules with energy above or equal to the threshold energy that can collide effectively is shown by the darkened region abed.
- Orientation barrier: To promote the production of new bonds and the breaking of existing ones, the reactant molecules must clash in a favourable orientation. Collisions are effective when the atoms of the molecules involved make direct touch with one another thanks to the proper orientation.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
