Advertisements
Advertisements
प्रश्न
Describe the graphical representation of first order reaction.
Describe the graphical method for the determination of the order of a first order reaction.
How would you represent a first order reaction graphically?
Advertisements
उत्तर १
The rate constant for first order reaction is,
kt = ln `([A_0])/([A])`
kt = In [A0] – In [A]
In [A] = In [A0] – kt
y = c + mx
If we follow the reaction by measuring the concentration of the reactants at regular time intervals ‘t’, a plot of ln [A] against ‘t’ yields a straight line with a negative slope. From this, the rate constant is calculated.

A plot of ln [A] vs t for a first-order reaction, \[\ce{A -> product}\] with initial concentration of [A] = 1.00 M and k = 2.5 × 10−2 min−1
उत्तर २
A first order reaction can be represented graphically in several ways. Some important graphical representations and their characteristics are as follows.
Reaction rate vs concentration plot: The rate law for a first-order reaction is given by
Rate = k[A]

A plot of reaction rate vs concentration of reactant for a first order reaction.
The equation y = mx indicates a straight line that passes through the origin. Plotting reaction rates against reactant concentration yields a straight line that passes through the origin, as illustrated in the picture above. The slope of the line corresponds to the rate constant of the reaction.
\[\ce{Slope of the line = \frac{CB}{AC} = k}\]
Concentration vs. time plot: We know that
[A] = [A]0 e−kt

Exponential decay of the conc. of reactant as a function of time for a first order reaction.
The equation shows that a first-order process is exponential, with the concentration of reactant decreasing exponentially over time. Graphing reactant concentrations over time yields an exponential decay curve, as illustrated in the image above.
log10 [A] vs time plot: We know that
\[\ce{k = \frac{2.303}{t} log_10 \frac{[A]_0}{[A]}}\]
This equation can be written as
\[\ce{log_10 [A] = - \frac{kt}{2.303} + log_10[A]_0}\]
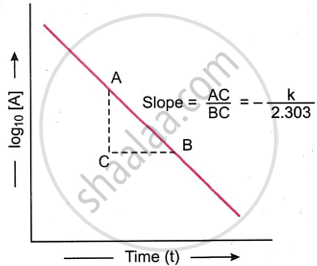
The above equation is of the type y = mx + c and represents a straight line. Therefore, on plotting log10 [A] against t, a straight line (as shown in the figure above) is obtained. The slope of the line is equal to \[\ce{\frac{-k}{2.303}}\] while the intercept of the line on log10 [A] axis is equal to log10 [A]0. Thus,
\[\ce{Slope of the line = \frac{AC}{BC} = - \frac{k}{2.303}}\]
Intercept on log10 [A] axis = log10 [A]0
Thus, the value of k can be obtained from the slope of the line.
संबंधित प्रश्न
Answer the following in one or two sentences.
How do the half-lives of the first order and zero-order reactions change with the initial concentration of reactants?
Answer the following in brief.
Obtain the relationship between the rate constant and half-life of a first-order reaction.
In a first-order reaction, the concentration of the reactant decreases from 20 mmol dm−3 to 8 mmol dm−3 in 38 minutes. What is the half-life of reaction?
For the reaction 2NOBr → 2NO2 + Br2, the rate law is rate = k[NOBr]2. If the rate of a reaction is 6.5 × 10–6 mol L–1 s–1, when the concentration of NOBr is 2 × 10–3 mol L–1. What would be the rate constant of the reaction?
Write the rate law for the following reaction.
A reaction that is second order in NO and first order in Br2.
The rate constant of a first order reaction is 3 × 10−6 per s. If the initial concentration is 0.10 mol dm−3, the initial rate of reaction is ____________.
In the reaction \[\ce{2SO2 + O2 -> 2SO3}\] the rate of appearance of SO3 is 4 × 10−4 M/s, the rate of disappearance of O2 is ____________.
Obtain the expression for half-life and rate constant of the first-order reaction.
Consider the following reaction.
\[\ce{SO2(g) + 1/2 O2(g) <=>[K1] SO3(g)}\]
\[\ce{2SO3(g)<=>[K2] 2SO2(g) + O2(g)}\]
What is the relation between K1 and K2?
A first order reaction takes 10 minute for 30% completion. Find rate constant of the reaction.
