Advertisements
Advertisements
प्रश्न
For the reaction:
\[\ce{2A + B → A2B}\]
the rate = k[A][B]2 with k = 2.0 × 10−6 mol−2 L2 s−1. Calculate the initial rate of the reaction when [A] = 0.1 mol L−1, [B] = 0.2 mol L−1. Calculate the rate of reaction after [A] is reduced to 0.06 mol L−1.
Advertisements
उत्तर
Given: [A] = 0.1 mol L−1,
[B] = 0.2 mol L−1,
k = 2.0 × 10−6
The initial rate of the reaction is
Rate = k [A][B]2
= 2.0 × 10−6 × 0.1 × (0.2)2
= 8 × 10−9 mol L−1 s−1
When [A] reduces to 0.06 mol L−1 i.e. 0.04 mol L−1 of A has reacted, then the reactant B
= `1/2 xx 0.04`
= 0.02 mol L−1
Hence, the new [B] = 0.2 − 0.02 = 0.18 mol L−1
Thus, the new concentrations of A and B are
[A] = 0.06 mol L−1,
[B] = 0.18 mol L−1,
Now rate = 2.0 × 10−6 × (0.06) × (0.18)2
= 3.89 × 10−9 mol L−1 s−1
APPEARS IN
संबंधित प्रश्न
For a reaction A + B ⟶ P, the rate is given by
Rate = k [A] [B]2
What is the overall order of reaction if A is present in large excess?
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is doubled?
Write resonating structures of ozone.
Write the principle behind the following methods of refining:
Hydraulic washing
Define the following terms:
Half-life period of reaction (t1/2).
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?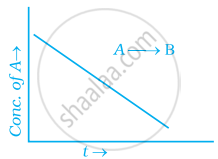
Why is the probability of reaction with molecularity higher than three very rare?
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Assertion: Order of the reaction can be zero or fractional.
Reason: We cannot determine order from balanced chemical equation.
Assertion: Rate constants determined from Arrhenius equation are fairly accurate for simple as well as complex molecules.
Reason: Reactant molecules undergo chemical change irrespective of their orientation during collision.
Use Molecular Orbital theory to determine the bond order in each of species, [He2j+ and [He2]2+?
The role of a catalyst is to change
In the presence of a catalyst, the heat evolved or absorbed during the reaction.
For a first order A → B, the reaction rate at reactant concentration of 0.01 m is found to be 2.0 × 10–5. The half-life period of reaction.
The following data was obtained for chemical reaction given below at 975 K.
\[\ce{2NO(g) + 2H2(g) -> N2(g) + 2H2O(g)}\]
| [NO] | [H2] | Rate | |
| Mol L-1 | Mol L-1 | Mol L-1 s-1 | |
| (1) | 8 × 10-5 | 8 × 10-5 | 7 × 10-9 |
| (2) | 24 × 10-5 | 8 × 10-5 | 2.1 × 10-8 |
| (3) | 24 × 10-5 | 32 × 10-5 | 8.4 × 10-8 |
The order of the reaction with respect to NO is ______. (Integer answer)
