Advertisements
Chapters
1: Solid State
2: Solutions
▶ 3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
Inorganic Chemistry
6: General Principles and Processes of Isolation of Elements
7: p-Block Elements
8: d-and ƒ-Block Elements
9: Coordination Compounds
Organic Chemistry
10: Haloalkanes and Haloarenes
Chapter 11: Alcohols, Phenols and Ethers
Chapter 12: Aldehydes, Ketones and Carboxylic Acids
Chapter 13: Organic Compounds containing Nitrogen
Chapter 14: Biomolecules
15: Polymers
Chapter 16: Chemistry in Everyday Life
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 3 - Electrochemistry Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 3 - Electrochemistry - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Advertisements
Solutions for Chapter 3: Electrochemistry
Below listed, you can find solutions for Chapter 3 of CISCE Nootan for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry REVIEW EXERCISES [Pages 148 - 187]
Define a galvanic cell.
Define reference electrode.
Define standard electrode potential.
What is understood by a normal hydrogen electrode?
Give the significance of the normal hydrogen electrode.
Define standard electrode potential.
What do you understand by electrochemical series?
How does the electrochemical series help in predicting whether a given reaction is feasible in a given direction or not?
It is impossible to measure the potential of a single electrode. Comment.
What is standard hydrogen electrode?
Give the reaction that occurs at standard hydrogen electrode when it acts as positive electrode in an electrochemical cell.
What is the effect of change in concentration on the electrode potential of a given half-cell?
What is the effect of change in temperature on the electrode potential of a given half cell?
Write the Nernst equation and explain the terms involved.
State the values of `E_(Ca//Ca^(2+))^circ, E_(Ni//Ni^(2+))^circ` and `E_(Cl^-//1/2Cl_2)^circ` if `E_(Ca^(2+)//Ca)^circ` = −2.87 volts, `E_(Ni^(2+)//Ni)^circ` = −0.25 volts and `E_(1/2Cl_2//Cl^-)^circ` = +1.36 volts.
A potential difference of 0.25 volts is observed when an electrode system M/M2+ (1 mol L−1) is connected to a standard hydrogen electrode. If the direction of the flow of current is from hydrogen electrode to the metal electrode, calculate the standard potential of the electrode.
The standard reduction potential of the electrode X/X+ (1 mol L−1) is +0.80 volts. Predict the direction of the flow of electrons when it is connected to a standard hydrogen electrode.
Predict whether the following reaction is feasible under standard conditions or not:
\[\ce{Zn_{(s)} + Cu{^{2+}_{(aq)}} -> Zn{^{2+}_{(aq)}} + Cu_{(s)}}\]
Given that `E_(Zn^(2+)//Zn)^circ` = −0.76 volts and `E_(Cu^(2+)//Cu)^circ` = +0.34 volts.
On the basis of electrochemical series, explain why Fe can displace hydrogen from dilute HCl but silver cannot.
On the basis of electrochemical series, explain why HCl cannot be stored in an aluminium vessel.
On the basis of electrochemical series, explain why Au2O3 decomposes on heating.
On the basis of electrochemical series, explain why a coating of copper is deposited on an iron nail when placed in an aqueous solution of copper sulphate.
Can we store copper sulphate solution in zinc vessel? Give suitable explanation.
Given: \[\ce{E_{Cu^{2+}/Cu}}\] = +0.34 V, \[\ce{E_{Zn^{2+}/Zn}}\] = −0.76 V, \[\ce{E_{Ag^{+}/Ag}}\] = +0.80 V
Can we store copper sulphate solution in silver vessel? Give suitable explanation.
Given: \[\ce{E{^{\circ}_{Cu^{2+}/Cu}}}\] = +0.34 V, \[\ce{E{^{\circ}_{Zn^{2+}/Zn}}}\] = −0.76 V, \[\ce{E{^{\circ}_{Ag^{+}/Ag}}}\] = +0.80 V.
The two half cell reactions and their oxidation potentials are
- \[\ce{Pb_{(s)} - 2e- -> Pb{^{2+}_{(aq)}}, E^{\circ}_{Pb/Pb^{2+}}}\] = +0.13 V
- \[\ce{Ag_{(s)} - e- -> Ag{^+_{(aq)}}, E^{\circ}_{Ag/Ag^{+}}}\] = −0.80 V
Write the cell reaction and calculate the cell emf.
A cell is prepared by dipping a copper rod in 1 M copper sulphate solution and a zinc rod in 1 M zinc sulphate solution. The standard reduction potentials of copper and zinc are 0.34 V and −0.76 V respectively.
- What will be the cell reaction?
- What will be the standard emf of the cell?
- Which electrode will be positive?
- How can the cell be represented?
Calculate at 25°C the electrode potential of Mg2+/Mg electrode in which the concentration of Mg2+ ions is 0.1 M. Given \[\ce{E^{\circ}_{Mg^{2+}/Mg}}\] = −2.36 V, R = 8.314 JK−1, F = 96500 coulombs mol−1.
Why does the blue colour of copper sulphate solution get discharged when zinc rod is dipped into it?
Given: \[\ce{E^{\circ}_{Cu^{2+}/Cu}}\] = +0.34 V and \[\ce{E^{\circ}_{Zn^{2+}/Zn}}\] = −0.76 V.
Using the standard electrode potentials, predict if the reaction between the following is feasible:
\[\ce{Fe^{3+}_{ (aq)}}\] and \[\ce{I^-_{ (aq)}}\]
Using the standard electrode potentials, predict if the reaction between the following is feasible:
\[\ce{Ag^+_{ (aq)}}\] and Cu(s)
Using the standard electrode potentials, predict if the reaction between the following is feasible:
\[\ce{Fe^{3+}_{ (aq)}}\] and \[\ce{Br^-_{ (aq)}}\]
Using the standard electrode potential, predict if the reaction between the following is feasible:
Ag(s) and \[\ce{Fe^{3+}_{ (aq)}}\]
Using the standard electrode potential, predict if the reaction between the following is feasible:
Br2(aq) and \[\ce{Fe^{2+}_{ (aq)}}\]
Calculate the electrode potentials of the following half cell at 25°C.
Pt, Cl2 (10 atm) | HCl (0.1 M)
Given: \[\ce{E^{\circ}_{\frac{1}{2}Cl_2/Cl^-}}\] = +1.36 V and \[\ce{E{^{\circ}_{H^+/\frac{1}{2}H_2}}}\] = 0.00 V.
Calculate the electrode potentials of the following half cell at 25°C.
Pt, H2 (5 atm) | HCl (0.5 M)
Given: \[\ce{E^{\circ}_{\frac{1}{2}Cl_2/Cl^-}}\] = +1.36 V and \[\ce{E{^{\circ}_{H^+/\frac{1}{2}H_2}}}\] = 0.00 V.
Calculate the electrode potentials of the following half cells at 298 K.
Ag(s) | AgNO3 (0.1 M)
Given: \[\ce{E^{\circ}_{Ag^+/Ag}}\] = +0.80 V, \[\ce{E^{\circ}_{{Co^{2+}/Co}}}\] = −0.28 V
Calculate the electrode potentials of the following half cells at 298 K.
Co(s) | Co2+ (0.01 M)
Given: \[\ce{E^{\circ}_{Ag^+/Ag}}\] = +0.80 V, \[\ce{E^{\circ}_{{Co^{2+}/Co}}}\] = −0.28 V
Write Nernst equation for the following cell reaction:
\[\ce{2 A + 3 B -> C + 2 D}\]
How can the cell potential be used in predicting the feasibility of a cell reaction?
Calculate the EMF of the cell at 298 K:
Pt, Br2 | Br− (0.01 M) || H+ (0.03 M) | H2 (1 atm), Pt
Given: \[\ce{E^{\circ}_{\frac{1}{2}Br/Br^-}}\] = +1.08 V.
Calculate the standard electrode potential of the Ni2+/Ni electrode, if the cell potential of the cell is 0.59 V.
Ni | Ni2+ (0.01 M) || Cu2+ (0.1 M) | Cu
Given: \[\ce{E^{\circ}_{Cu{^{2+}/Cu}}}\] = +0.34 V
Calculate the emf of the following cell at 298 K:
Mg | Mg2+ (0.001 M) || Cu2+ (0.0001 M) | Cu
Given: \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}}}\] = 0.337 V, \[\ce{E^{\circ}_{{Mg^{2+}/{Mg}}}}\] = −2.37 V, F = 96500 C mol−1.
Calculate the EMF of the following cell at 298 K:
Mg | Mg2+ (0.1 M) || Ag+ (0.1 M) | Ag
\[\ce{E^{\circ}_{{Mg^{2+}/{Mg}}}}\] = −2.37 V, \[\ce{E^{\circ}_{{Ag^{+}/{Ag}}}}\] = +0.80 V, R = 8.31 J K−1, F = 96500 C mol−1.
Consider a cell composed of two half cells:
- \[\ce{Cu_{(s)} | Cu{^{2+}_{(aq)}}}\] and
- \[\ce{Ag_{(s)} | Ag{^+_{(aq)}}}\].
Calculate: (a) the standard cell potential, and (b) the cell potential when [Cu2+] is 2M and [Ag+] is 0.05 M.
Given: \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}}}\] = +0.33 V, \[\ce{E^{\circ}_{{Ag^{+}/{Ag}}}}\] = +0.80 V, R = 8.31 J K−1 mol−1, F = 96500 C mol−1.
Consider a cell composed of the following two half cells:
- \[\ce{Mg_{(s)} | Mg{^{2+}_{(aq)}}}\], and
- \[\ce{Ag_{(s)} | Ag{^{+}_{(aq)}}}\]
The EMF of the cell is 2.96 V, [Mg2+] = 0.130 M and [Ag+] = 1.0 × 10−4 M. Calculate the standard EMF of the cell. (R = 8.31 J K−1 mol−1, F = 96500 C mol−1)
Calculate the cell potential at 298 K:
\[\ce{Zn_{(s)} | Zn^{2+} (0.1 M) || Sn^{2+} (0.001 M) | Sn_{(s)}}\]
(Given: \[\ce{E{^{\circ}_{Zn^{2+}/Zn}}}\] = −0.76 V, \[\ce{E{^{\circ}_{Sn^{2+}/Sn}}}\] = −0.14 V, Gas constant, R = 8.314 J K−1 mol−1, Faraday constant, F = 96500 C mol−1)
The standard potentials are given as:
\[\ce{E{^{\circ}_{Cu^{2+}/Cu}}}\] = +0.34 V, and \[\ce{E{^{\circ}_{Ag^{+}/Ag}}}\] = +0.80 V.
Calculate the cell potential (E) for the cell containing 0.100 M Ag+ and 4.00 M Cu2+ at 298 K.
Predict whether the following reaction will occur spontaneously at 298 K:
\[\ce{Co_{(s)} + Fe{^{2+}_{(aq)}} -> Co{^{2+}_{(aq)}} + Fe_{(s)}}\]
Given: \[\ce{E^{\circ}_{{Co^{2+}/{Co}}}}\] = −0.28 V and \[\ce{E^{\circ}_{{Fe^{2+}/{Fe}}}}\] = −0.44 V.
The half reactions are:
- \[\ce{Fe^3+ + e- -> Fe^2+}\]; E° = 0.76 V
- \[\ce{Ag+ + e- -> Ag}\]; E° = 0.80 V
Calculate Kc for the following reaction at 25°C:
\[\ce{Ag+ + Fe^2+ -> Fe^3+ + Ag}\] (F = 96500 C mol−1)
Calculate the emf of the cell:
Mg(s) | Mg2+ (0.1 M) || Cu2+ (1 × 10−3 M) | Cu(s)
Given \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}}}\] = +0.34 V, \[\ce{E^{\circ}_{{Mg^{2+}/{Mg}}}}\] = −2.37 V
What are electrolytic conductors?
Define the following term:
Specific conductance
Define the term equivalent conductance.
Define the term cell constant.
Define “Molar conductivity”.
What is the SI unit of molar conductivity?
Define “Molar conductivity”.
Mention the effect of temperature on molar conductivity.
Define the term equivalent conductance.
Define “Molar conductivity”.
What are the physical significances of equivalent conductivity?
The resistance of 0.01 M NaCl solution in a conductivity cell was found to be 210 Ω. The specific conductance of this solution is 4.5 × 10−3 S cm−1. What is the cell constant of the cell?
The resistance of 0.01 N NaCl solution is 200 Ω at 25°C. Cell constant of the conductivity cell is unity. Calculate the equivalent conductance of the solution.
The specific conductance of `N/50` solution of KCl at 25°C is 0.002765 mho cm−1. If the resistance of a cell containing this solution is 400 ohms, find out the cell constant.
Calculate the equivalent conductance of 1 M H2SO4 solution, if its conductivity is 2.6 × 10−2 ohm−1 cm−1.
The resistance of a decinormal solution of a salt occupying a volume between two platinum electrodes 1.80 cm apart and 5.4 cm2 in area was found to be 32 ohms.
Calculate the specific and equivalent conductances of the solution.
A conductivity cell has its electrodes 1 cm apart and each electrode has a cross section of 2 cm2. When filled with `N/50` solution of an electrolyte MX, the cell shows a resistance of 166.5 ohms. Calculate the equivalent conductivity of MX at the given concentration.
A potential difference of 20 volts applied to the ends of a column of `N/10` AgNO3 solution, 4 cm in diameter and 12 cm in length, gives a current of 1.198 amperes. Calculate the specific and equivalent conductivity of the solution.
A conductivity cell when filled with 0.05 M solution of KCl records a resistance of 410.5 ohm at 25°C. When filled with CaCl2 solution (11 g in 500 mL), it records a resistance of 990 ohms. If specific conductance of 0.05 M KCl is 0.00189 ohm−1 cm−1, calculate
- cell constant;
- specific conductance of CaCl2;
- equivalent conductance of CaCl2 solution;
- molar conductance of CaCl2 solution.
A cell with N/50 KCl solution showed a resistance of 550 ohms at 25°C. The specific conductivity of N/50 KCl at 25°C is 0.002768 ohm−1,cm−1. The cell filled with N/10 ZnSO4 solution at 25°C shows a resistance of 72.18 ohms. Calculate the cell constant and molar conductivity of ZnSO4 solution.
How does molar conductance of a strong electrolyte vary with its concentration in solution?
How does molar conductivity of a weak electrolyte vary with its concentration in solution?
What is the effect of decreasing concentration on the molar conductivity of a weak electrolyte?
State and explain Kohlrausch’s law.
How can the degree of dissociation of acetic acid be calculated from its molar conductance data?
At 298 K, the molar conductivities at infinite dilution of sodium propionate (CH3CH2COONa), HCl and NaCl are 85.9, 426.1 and 126 ohm−1 cm2 mol−1 respectively. Calculate the molar conductivity of propionic acid at infinite dilution.
The molar conductivity of a weak monobasic acid at infinite dilution is 387 ohm−1 cm2 mol−1. The 0.02 M solution of the same acid has a specific conductivity of 3.3 × 10−4 ohm−1 cm2 mol−1. Calculate the degree of dissociation of the acid at the given concentration.
The molar conductivity of CH3COOH at infinite dilution is found to be 387 ohm−1 cm2 mol−1 but at a dilution of 1 g mol in 1000 litres it is 55 ohm−1 cm2 mol−1. What is the percent dissociation of the acid at the given dilution?
Calculate the molar conductivity at infinite dilution for CH3COOH if the molar conductivity at infinite dilution for NaCl, HCl and CH3COONa are 126.45, 426.16 and 91.0 ohm−1 cm2 mol−1 respectively.
The molar conductance at infinite dilution of Al2(SO4)3 is 858 ohm−1 cm2 mol−1. Calculate the molar conductance at infinite dilution of Al3+ ion if that of \[\ce{SO^{2-}_{4}}\] ion is 160 ohm−1 cm2 mol−1.
What is electrolysis?
Explain the mechanism of electrolysis by taking a suitable example.
Define the following term:
Faraday
Define the following term:
Electrochemical equivalent
Explain the criteria for product formation during electrolysis taking the example of aqueous sodium chloride.
State Faraday’s Laws of electrolysis.
Predict the products of electrolysis of an aqueous solution of CuBr2 using inert electrodes.
Given: \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}}}\] = 0.34 V, \[\ce{E^{\circ}_{{H_{2}O}/H_{2}}}\], OH− = −0.83 V, \[\ce{E^\circ_{Br/Br^-}}\] = 1.08 V and \[\ce{E^{\circ}_{\frac{1}{2} O_{2}, H{^{+}}/{H_{2}O}}}\] = 1.23 V.
How many coulombs of electricity are required for reduction of 1 mole of \[\ce{Cr2O^{2-}_{ 7}}\] to Cr3+?
How much electricity is required in coulomb for the oxidation of 1 mol of H2O to O2?
How many coulombs of electricity are required for reduction of 1 mole of Sn4+ to Sn2+?
Calculate the number of coulombs required to deposit 5.4 g of Al when the electrode reaction is Al3 + 3e– + Al.
(Given: Atomic mass of Al = 27 g mol−1, F = 96500 C mol−1)
Calculate the quantity of electricity that would be required to reduce 12.3 g of nitrobenzene to aniline, if the current efficiency is 50%.
How many molecules of chlorine should be deposited from sodium chloride in 1 minute by a current of 300 mA?
A constant current was passed through a solution of \[\ce{AuCl^-_4}\] between gold electrodes. After a period of 14 minutes, the cathode recorded an increase in mass of 1.808 g. Calculate the quantity of current passed in solution. What was the magnitude of current? (Atomic mass of gold = 197.0)
A current of 4 amperes was passed for 2 hours through a solution of copper sulphate when 5.0 g of copper was deposited. Calculate the current efficiency.
Two voltameters containing CuSO4 and acidulated water respectively are connected in series and the same current is passed for some time. 0.3177 g copper is deposited in the first one. Calculate the weight of liberated hydrogen in the second. (At. wt. of Cu = 63.5 and H = 1.008)
An aqueous solution of NaCl on electrolysis gives H2(g), Cl2(g) and NaOH according to the reaction
\[\ce{2Cl^-_{ (aq)} + 2H2O -> 2OH^-_{ (aq)} + H2_{(g)} + Cl2_{(g)}}\].
A direct current of 25 amperes with a current efficiency of 62% is passed through 20 litres of NaCl solution (20% by weight).
- Write down the reactions taking place at the electrodes.
- How long will it take to produce 1 kg of Cl2?
- What will be the molarity of solution with respect to OH−? Assume no loss in volume due to evaporation.
Explain, why electrolysis of aqueous solution of NaCl gives H2 at cathode and Cl2 at anode. Write overall reaction.
[Given: \[\ce{E^{\circ}_{{Na^{+}/{Na}}}}\] = 2.71 V, \[\ce{E^{\circ}_{Cl_2/2Cl^-}}\] = 1.36 V and \[\ce{\frac{1}{2} O2_{(g)} + 2H+_{ (aq)} + 2e- -> H2O_{(l)}}\]; E° = 1.23 V]
Silver is electrodeposited on a metallic vessel of surface area 800 cm2 by passing current of 0.2 amp for 3 hours. Calculate the thickness of silver deposited.
(Density of silver = 10.47 g cm−3, atomic mass of silver = 107.92 amu)
Predict the products of electrolysis obtained at the electrodes in the case when the electrodes used are of platinum:
An aqueous solution of AgNO3.
Predict the product of electrolysis in the following:
A dilute solution of H2SO4 with platinum electrodes.
How much charge is required for the following reduction?
1 mol of Cu2+ to Cu.
What is a primary cell?
What is a secondary cell?
Write the differences between the primary cell and the secondary cell.
What do you understand by the term fuel-cell?
Give electrode reactions of hydrogen-oxygen fuel cell.
What is a mercury cell?
Give the electrode reactions of the mercury cell.
What is corrosion?
Give a brief account of the mechanism of corrosion.
What is corrosion?
How is the cathodic protection of iron different from its galvanisation?
What is galvanisation?
In what respect is galvanisation better than tin coating used for the protection of iron from corrosion?
Write two applications of fuel cells.
Describe the limitations of fuel cells.
Explain why tin protects copper more efficiently as compared to iron?
Discuss the factors which govern the rate of corrosion of a metal.
Give a brief account of the mechanism of corrosion.
Give an example of a fuel cell and write the anode and cathode reactions for it.
How does a fuel cell operate?
Write the cell reactions which occur in lead storage battery when the battery is in use.
Write the cell reactions which occur in a lead storage battery when the battery is on charging.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry VERY SHORT ANSWER TYPE QUESTIONS [Pages 192 - 193]
Differentiate between conductors and insulators.
What do you understand by non-electrolytes?
What is the effect of temperature on metallic conduction?
What type of species are responsible for electrolytic conduction?
Can electric current be passed through a solid ionic compound?
How does electrolytic conduction vary with temperature?
What is meant by the term ‘electrolyte’?
Give two examples of electrolytes.
Distinguish between strong electrolyte and weak electrolyte.
Define the degree of dissociation.
How does solute-solute interaction affect electrolytic conduction?
What is the effect of solute-solvent interaction on electrolytic conduction?
State Ostwald’s dilution law.
Define the following term:
Specific conductance
What are the units of specific conductivity?
How is equivalent conductivity related to specific conductivity?
Define the term equivalent conductance.
Mention the units of equivalent conductivity.
What is the relationship between molar conductivity and specific conductivity?
Define “Molar conductivity”.
What is the SI unit of molar conductivity?
What is cell constant?
Define molar conductivity at infinite dilution.
Why is the molar conductivity of a weak electrolyte much lower than that of a strong electrolyte at moderate concentrations?
What is specific resistance?
How would you represent a Daniell cell?
What is the polarity of anode in a galvanic cell and why?
What do you understand by a half cell?
How would you represent a half cell in which H+ ions of conc. 0.2 M get reduced into hydrogen gas at atmospheric pressure?
What is the direction of flow of electrons in the outer circuit in a galvanic cell?
Define electrode potential.
What is a null electrode?
Name the electrode and the potential when the electrode reaction involves reduction.
What do you understand by standard electrode potential?
What is the reference electrode generally used in the measurement of standard electrode potential of an electrode assembly?
What is the function of platinised platinum in the standard hydrogen electrode?
Represent a standard hydrogen electrode.
What is the direction of flow of electrons in the outer circuit when a standard hydrogen electrode is connected to a copper electrode?
How does the tendency to gain electrons vary when one moves from the top to the bottom in the electrochemical series?
It is impossible to measure the potential of a single electrode. Comment.
Predict whether F2 and Na will react together:
\[\ce{E^{\circ}_{F/F^-}}\] = +2.87 V, \[\ce{E^{\circ}_{{Na^{+}/{Na}}}}\] = −2.71 V
Which substance can be used to oxidise fluorides to fluorine?
How does the reducing power of elements vary on moving down the electrochemical series?
Can copper displace zinc from its salt solution?
\[\ce{E^{\circ}_{{Zn^{2+}/{Zn}}}}\] = −0.76 V, \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}}}\] = +0.34 V
Can nickel displace hydrogen from hydrochloric acid?
\[\ce{E^{\circ}_{{Ni^{2+}/{Ni}}}}\] = −0.25 V
Write the Nernst equation for a reduction electrode.
Define emf of a galvanic cell.
What is the convention for the emf of a cell?
Mention the relationship between the standard cell potential and standard electrode potentials of the two electrodes.
Write the Nernst equation for a cell having the cell reaction.
\[\ce{aA + bB -> cC + dD}\].
The standard reduction potential for \[\ce{Zn{^{2+}_{(aq)}} | Zn_{(s)}}\] half cell is −0.76 V. Write the reactions occurring at the electrodes when coupled with NHE.
Define electrolysis.
Why does an aqueous solution of sodium chloride on electrolysis give H2 at cathode instead of sodium?
What is overvoltage?
Why do Cl− ions get oxidised at anode in preference to H2O during the electrolysis of an aqueous solution of sodium chloride?
Predict the products of electrolysis of a dilute aqueous solution of sulphuric acid using inert electrodes.
State Faraday’s Laws of electrolysis.
What is electrochemical equivalent?
How can electrochemical equivalent be calculated for a particular substance?
What is the quantity of electricity needed to liberate one equivalent of a substance?
Calculate the electric charge carried by one electron on the basis of Faraday’s constant.
How much charge is required to reduce 8 g of H+ ions to H2 gas?
How can you increase the reduction potential of an electrode?
Electrolysis of KBr(aq) gives Br2 at anode but that of KF(aq) does not give F2. Give reason.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry SHORT ANSWER TYPE QUESTIONS [Pages 194 - 195]
Define electrochemistry.
Discuss the scope of electrochemistry.
What is metallic conduction?
How does metallic conduction differ from electrolytic conduction?
Discuss the factors which affect electrolytic dissociation.
What is specific resistance?
What do you understand by specific conductance of a solution?
Derive the units for specific resistance of a solution.
Derive the units for conductance of a solution.
Derive the units of specific conductivities of an electrolytic solution and write expressions which show their mutual relationship.
Define the term equivalent conductance.
How is equivalent conductivity related to specific conductivity?
Derive the units of equivalent conductivity of a solution.
Define “Molar conductivity”.
What is the SI unit of molar conductivity?
What is the relationship between molar conductivity and specific conductivity?
Derive a relationship between molar conductivity and equivalent conductivity.
What is conductivity cell?
For what purpose is a conductivity cell used?
What is wheatstone bridge?
What is the wheatstone bridge’s significance in the determination of the conductivity of a solution?
What is cell constant?
How is cell constant determined?
Can a direct current be used for the measurement of electrical conductivity of a solution? If not, why?
How does the molar conductivity for the solution of a strong electrolyte vary with concentration? Explain graphically.
What is Debye-Huckel-Onsagar equation and what is its significance?
Explain why the molar conductivity of a strong electrolytic solution increases only slightly when concentration is decreased.
What do you understand by molar conductivity at infinite dilution?
How does the molar conductivity for the solution of a strong electrolyte vary with concentration? Explain graphically.
How does the molar conductivity of a weak electrolytic solution vary with concentration?
The molar conductivity at infinite dilution of a strong electrolyte can be obtained by extrapolating the curve Λm vs `sqrt c`, but the same cannot be done for a weak electrolyte. Explain why.
Why does the molar conductivity of a weak electrolytic solution increase sharply at very low concentrations?
How does the molar conductivity of a weak electrolytic solution vary with concentration?
What is the effect of dilution on the specific conductivity of a solution?
What is the effect of dilution on the equivalent conductivity of a solution?
What is the effect of dilution on the molar conductivity of a solution?
What is a galvanic cell?
How can a galvanic cell be constructed?
What do you understand by a half cell?
What are the half cells present in a Daniell cell?
How would you represent a Daniell cell?
How would you represent a Daniell cell?
Explain why the anode is of negative polarity in a galvanic cell.
Explain why the cathode is of positive polarity in a galvanic cell.
What type of reactions do occur at the two electrodes at a galvanic cell and what is the nature of the net cell reaction? Explain with an example.
What is electrode potential?
How does electrode potential come into existence?
What is a salt bridge?
What are the functions of a salt bridge in a galvanic cell?
Write the ion electron equations for the half cell reactions and overall cell reaction for the cell.
\[\ce{Pt, H2_{(g)} | HCl_{(aq)} || Fe^{3+}, Fe^{2+} | Pt}\]
Depict the electrochemical cell in which the cell reaction is
\[\ce{2Cr_{(s)} + 3Cu{^{2+}_{(aq)}} -> 2Cr{^{3+}_{(aq)}} + 3Cu_{(s)}}\]
- mark the anode and the cathode;
- show movement of electrons and flow of current;
- write the half cell reactions.
Write the symbolic representation of the electrochemical cell with the cell reaction.
\[\ce{Zn_{(s)} + 2Ag{^+_{(aq)}} -> Zn{^{2+}_{(aq)}} + 2Ag_{(s)}}\]
Indicate the oxidation electrode and the direction of movement of electrons and write the electrode reactions.
What is standard hydrogen electrode?
What is standard hydrogen electrode significance?
Define electrode potential.
It is impossible to measure the potential of a single electrode. Comment.
How are standard electrode potentials measured?
When a Zn2+/Zn electrode is connected to a SHE, the electrons flow from zinc electrode to hydrogen electrode but the flow of electrons is in opposite direction when a Cu2+/Cu electrode is connected to a SHE. Explain giving reasons.
How does the tendency to gain electrons vary when one moves from the top to the bottom in the electrochemical series?
How does the following property vary on moving down the electrochemical series?
Reducing power of the reduced form.
How does the following property vary on moving down the electrochemical series?
Oxidising power of the oxidised form.
Justify the statement:
If \[\ce{E^{\circ}_{{M^{n+}/{M}}}}\] is positive, Mn+ can be reduced to M by H2 gas under standard conditions.
Comment on the statement:
If \[\ce{E^{\circ}_{{M^{n+}/{M}}}}\] is negative, H+ ions cannot be reduced to H2(g) by the metal M.
On what factor does the reactivity of a metal depend and why?
Explain, why can zinc displace Cu from an aqueous solution of CuSO4 but Cu cannot displace zinc from aqueous ZnSO4 solution?
What is meant by reduction electrode potentials of zinc and copper being −0.76 V and + 0.34 V respectively? Can an aqueous solution of CuSO4 be stored in a zinc vessel? Answer with reason.
What do you understand by electrochemical series?
How does the electrochemical series help in predicting whether a given reaction is feasible in a given direction or not?
What do you understand by electrochemical series?
How does the electrochemical series help in calculating the emf of a standard cell?
Give reason why the blue colour of copper sulphate solution is discharged slowly when an iron rod is dipped in it.
Given: \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}} = 0.34 V}\], \[\ce{E^{\circ}_{{Fe^{2+}/{Fe}}} = -0.44 V}\].
Write the equation which correlates the electrode potential of an electrode assembly with the concentration of ions and the temperature. What is the name of the equation and what is its significance?
What do you understand by EMF of a galvanic cell?
How can EMF of a galvanic cell be calculated if the cell is in standard condition?
Write the Nernst equation for the cell potential of a galvanic cell.
Apply the Nernst equation to a Daniell cell.
Write the Nernst equation for a cell having the cell reaction.
\[\ce{aA + bB -> cC + dD}\].
What is an electrolytic cell?
How does electrolytic cell differ from a galvanic cell?
Define electrolysis.
Discuss the electrolysis mechanism by taking the example of fused sodium chloride.
What is overvoltage?
How does overvoltage affect the electrolysis of an aqueous solution of sodium chloride?
Predict the products of electrolysis obtained at the electrodes in the case when the electrodes used are of platinum:
An aqueous solution of AgNO3.
Predict the product of electrolysis in the following:
An aqueous solution of AgNO3 with silver electrodes.
What are the criteria for product formation during electrolysis?
Why does the anode dissolve when an aqueous solution of CuSO4 is subjected to electrolysis using copper electrodes?
What are the products of electrolysis of an aqueous solution of dilute sulphuric acid? Explain with reasons.
State Faraday’s Laws of electrolysis.
How much charge is necessary to liberate one equivalent of a substance and why? Calculate the value of this charge.
All the energy released from the reaction \[\ce{X -> Y}\], ΔrG° = −193 KJ mol−1 is used for oxidizing \[\ce{M+ -> M^{3+} + 2e−}\], E° = −0.25 V.
Under standard conditions, the number of moles of M+ oxidized when one mole of X is converted to Y is [F = 96500 C mol−1].
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry LONG ANSWER TYPE QUESTIONS [Page 196]
What are electrolytic conductors?
How are electrolytic conductors classified?
Define the following term:
Specific conductance
Define the term equivalent conductance.
Define “Molar conductivity”.
Derive the units of specific conductivities of an electrolytic solution and write expressions which show their mutual relationship.
Derive the units of equivalent conductivity of a solution.
How is equivalent conductivity related to specific conductivity?
What is the SI unit of molar conductivity?
Derive a relationship between molar conductivity and equivalent conductivity.
What is the relationship between molar conductivity and specific conductivity?
What is cell constant?
How is cell constant determined?
What is the significance of the cell constant in the measurement of conductivity of an unknown electrolytic solution?
Discuss the method used to measure the electrolytic conductivity of a solution.
How would you determine the equivalent and molar conductivities of a given solution?
How does molar conductance of a strong electrolyte vary with its concentration in solution?
How does the molar conductivity of a weak electrolytic solution vary with concentration?
How does the molar conductivity for the solution of a strong electrolyte vary with concentration? Explain graphically.
What do you understand by molar conductivity at infinite dilution?
How does the molar conductivity of a weak electrolytic solution vary with concentration?
State and explain Kohlrausch’s law.
How would you determine the molar conductivity at infinite dilution of NH4OH with the help of Kohlrausch’s law?
What is a galvanic cell?
Describe the construction and working of a Daniell cell.
Draw a labelled diagram of Daniell cell and show
- electrodes working as anode and cathode and their polarity;
- direction of flow of electrons and direction of flow of conventional current;
- half cell reactions and the net cell reaction.
What is electrode potential?
How does electrode potential come into existence?
Explain with examples the term oxidation potential.
Explain with examples the term reduction potential.
What do you understand by standard electrode potential?
How are standard electrode potentials measured?
What do you understand by electrochemical series?
What are the important features of the electrochemical series?
How does the electrochemical series help in comparing the relative reducing or oxidising powers of different elements?
How does the electrochemical series help in predicting the displacement of hydrogen by metals from dilute acids?
On the basis of electrochemical series, how would you predict displacement of metals from salt solutions?
On the basis of electrochemical series, how would you predict feasibility of a redox reaction?
On the basis of electrochemical series, how would you predict thermal stability of oxides of different metals?
What is Nernst equation for the potential of an electrode?
Write the Nernst equation for a cell having the cell reaction.
\[\ce{aA + bB -> cC + dD}\].
What do you understand by EMF of a galvanic cell?
Write the IUPAC convention for calculating the standard emf of a cell.
Write the Nernst equation for the cell potential of a galvanic cell.
What is the significance of electrode potentials in predicting the products of electrolysis of an aqueous solution of an electrolyte? Explain with a suitable example.
Discuss the electrolysis of an aqueous solution of sodium chloride and explain the products formed at the two electrodes.
Mention the criteria to predict the formation of products during electrolysis of an aqueous solution of an electrolyte. Apply these criteria to the electrolysis of an aqueous solution of copper sulphate using inert electrodes.
State Faraday’s Laws of electrolysis.
How are Faraday’s laws of electrolysis helpful in exploring the quantitative aspect of electrolysis?
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry INTEGER TYPE QUESTIONS [Page 196]
The molar conductivities at infinite dilution of barium chloride, sulphuric acid and hydrochloric acid are 280, 860 and 426 S cm2 mol−1 respectively. The molar conductivity at infinite dilution of barium sulphate is ______ S cm2 mol−1.
(Round off to the nearest integer)
A KCl solution of conductivity 0.14 S m−1 shows a resistance of 4.19 Ω in a conductivity cell. If the same cell is filled with an HCl solution, the resistance drops to 1.03 Ω. The conductivity of the HCl solution is ______ × 10−2 S m−1
(Round off to the nearest integer)
At 298 K, the limiting molar conductivity of a weak mono basic acid is 4 × 102 S cm2 mol−1. At 298 K, for an aqueous solution of the acid, the degree of dissociation is ‘α’ and the molar conductivity is y × 102 S cm2 mol−1. At 298 K, upon 20 times dilution with water, the molar conductivity of the solution becomes 3y × 102 S cm2 mol−1.
- The value of ‘α’ is ______.
- The value of ‘y’ is ______.
The cell potential for the following cell:
\[\ce{Pt | H2_{(g)} | H{^+_{(aq)}} || Cu^+2 (0.01 M) | Cu}\] is 0.576 at 298 K.
The pH of the solution is ______. (Nearest integer)
The cell potential for the given cell at 298 K.
\[\ce{Pt | H2_{(g)} (1 bar) | H{^+_{(aq)}} || Cu{^{+2}_{(aq)}} | Cu_{(s)}}\] is 0.31 V.
The pH of the acidic solution is found to be 3 whereas the concentration of Cu+2 is 10−x M. The value of x is ______ (Nearest integer).
(Given, \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}} = 0.34 V}\] and \[\ce{\frac{2.303 RT}{F} = 0.06 V}\]).
A solution of Fe2(SO4)3 is electrolysed for ‘x’ min with a current of 1.5 A to deposit 0.3482 g of Fe. The value of ‘x’ is ______. (Nearest integer)
(Given, 1 F = 96500 C mol−1, Atomic mass of Fe = 56)
\[\ce{Cu_{(s)} + Sn{^{+2}} (0.001 M) -> Cu^{+2} (0.01 M) + Sn_{(s)}}\]
The Gibbs free energy change for the above reaction at 298 K is x × 10−1 KJ mol−1. The value of x is ______ (Nearest integer).
(Given \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}} = 0.34 V}\], \[\ce{E^{\circ}_{{Sn^{2+}/{Sn}}} = -0.14 V}\], F = 96500 C mol−1)
For the given reactions
\[\ce{Sn^{+2} + 2e- -> Sn}\]
\[\ce{Sn^{+4} + 2e- -> Sn}\]
The electrode potentials are \[\ce{E^{\circ}_{{Sn^{+2}/{Sn}}}}\] = −0.14 V and \[\ce{E^{\circ}_{{Sn^{+4}/{Sn}}}}\] = 0.010 V. The magnitude of standard electrode potential for \[\ce{Sn^{+4}/Sn^{+2}}\] i.e., \[\ce{E^{\circ}_{{Sn^{+4}/{Sn^{+2}}}}}\] is ______ × 10−2 V (Nearest integer).
The reduction potential (E° in V) of \[\ce{MnO^-_{4(aq)}/Mn_{(s)}}\] is ______.
Given \[\ce{E^{\circ}_{MnO^-_{4(aq)}/MnO_{2(s)}}}\] = 1.68 V
\[\ce{E^{\circ}_{MnO_{2(s)}/Mn^{+2}_{(aq)}}}\] = 1.21 V
\[\ce{E^{\circ}_{Mn^{+2}_{(aq)}/Mn_{(s)}}}\] = −1.03 V
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry OBJECTIVE (MULTIPLE CHOICE) TYPE QUESTIONS [Pages 197 - 203]
Choose the correct option in the following questions.
Molten sodium chloride conducts electricity due to the presence of ______.
free electrons
free ions
free molecules
free atoms of Na and Cl
When one Faraday of electric current is passed, the mass deposited is equal to ______.
one gram equivalent
one gram mole
half of electrochemical equivalent
half gram equivalent
A current liberated 0.50 g of hydrogen in 2 hours. How many grams of copper can be liberated by the same current flowing for the same time in CuSO4 solution?
12.7 g
15.9 g
31.8 g
63.5 g
One Faraday of charge was passed through the electrolytic cells placed in series containing solutions of Ag+, Ni2+ and Cr3+ respectively. The amounts of Ag (At. mass = 108), Ni (At. mass = 59) and Cr (At. mass = 52) deposited will be:
Ag Ni Cr 108 g 29.5 g 17.5 g Ag Ni Cr 108 g 59.0 g 52.0 g Ag Ni Cr 108 g 108.0 g 108.0 g Ag Ni Cr 108 g 117.5 g 166.0 g
Which one is the correct equation that represents the first law of electrolysis?
mZ = ct
Where, c represents the magnitude of current in amperes.
m = cZt
Where, c represents the magnitude of current in amperes.
mc = Zt
Where, c represents the magnitude of current in amperes.
c = mZt
Where, c represents the magnitude of current in amperes.
When electricity is passed through a solution of AlCl3, 13.5 g of Al is discharged, the amount of charge passed is ______.
1.5 F
0.5 F
1.0 F
2.0 F
The cathodic reaction in the electrolysis of dilute H2SO4 with platinum electrodes is ______.
oxidation
reduction
both oxidation and reduction
neutralisation
The reaction, \[\ce{\frac{1}{2} H2_{(g)} + AgCl_{(s)} -> H^+_{ (aq)} + Cl^-_{ (aq)} + Ag_{(s)}}\], occurs in the galvanic cell
\[\ce{Ag | AgCl_{(s)} | KCl (soln) || AgNO3 (soln) | Ag}\]
\[\ce{Pt | H2_{(g)} | HCl (soln) || AgNO3 (soln) | Ag}\]
\[\ce{Pt | H2_{(g)} | HCl (soln) || AgCl_{(s)} | Ag}\]
\[\ce{Pt | H2_{(g)} | KCl (soln) || AgCl_{(s)} | Ag}\]
The standard oxidation potentials, \[\ce{E{^{\circ}_{oxi}}}\] for the half cell reactions are as follows:
\[\ce{Zn -> Zn^2+ + 2e-}\]; \[\ce{E{^{\circ}_{oxi}}}\] = +0.76 V
\[\ce{Fe -> Fe^2+ + 2e-}\]; \[\ce{E{^{\circ}_{oxi}}}\] = +0.41 V
The emf of the cell, \[\ce{Fe^2+ + Zn -> Zn^2 + Fe}\], is ______.
+0.35 V
−0.35 V
+1.17 V
−1.17 V
If a spoon of copper metal is placed in a solution of ferrous sulphate, ______.
Cu will precipitate out
iron will precipitate
Cu and Fe will precipitate
no reaction will take place
Which one will liberate Br2 from KBr?
H2
I2
Cl2
SO2
The standard reduction potentials at 25°C for the following half cell reactions are given against each:
\[\ce{Zn{^{2+}_{(aq)}} + 2e- -> Zn_{(s)}}\] = −0.762 V
\[\ce{Cr{^{3+}_{(aq)}} + 3e- -> Cr_{(s)}}\] = −0.740 V
\[\ce{2H+ + 2e- -> H2_{(g)}}\] = 0.000 V
\[\ce{Fe^3+ + e- -> Fe^2+}\] = 0.770 V
Which is the strongest reducing agent?
Zn
Cr
H2(g)
\[\ce{Fe{^{2+}_{(aq)}}}\]
Which is not true for a standard hydrogen electrode?
The pressure of hydrogen is 1 atm.
The hydrogen ion concentration is 1 M.
Temperature is 25°C.
H2 gas is not absorbed by platinum electrode.
The standard electrode potentials of four elements A, B, C, D are −3.05, 1.66, −0.40 and 0.80 volts respectively. The highest chemical activity will be shown by ______.
A
B
C
D
Which one of the following will increase the voltage of the cell whose cell reaction is given below?
\[\ce{Sn + 2Ag+ -> Sn^2+ + 2Ag}\]
Increase in the size of silver rod.
Increase in the concentration of Sn2+ ions.
Increase in the concentration of Ag+ ions.
None of the above.
When electric current is passed through a cell having an electrolyte, the positive ions move towards the cathode and the negative ions towards the anode. If the cathode is pulled out of the solution ______.
the positive and the negative ions will move towards the anode
the positive ions will start moving towards the anode
the negative ions will continue to move towards the anode; the positive ions will stop moving
the positive ions and the negative ions will start moving randomly
If a salt bridge is removed from the two half cells, the voltage ______.
drops to zero
does not change
increases gradually
increases rapidly
1 Faraday of electricity will liberate 1 g atom of the metal from ______.
fused NaCl
aqueous BaCl2
aqueous CuSO4
aqueous AlCl3
The metal that cannot be obtained by electrolysis of the aqueous solution of its salts is ______.
Ag
Cr
Cu
Al
The standard reduction electrode potential values of the elements A, B and C are +0.68, −2.50 and −0.50 V respectively. The order of their reducing power is ______.
A > B > C
A > C > B
C > B > A
B > C > A
How many electrons carry one coulomb of charge?
6.02 × 1021
6.24 × 1018
6.24 × 1015
6.02 × 1016
If \[\ce{E^{\circ}_{{Ag^{+}/{Ag}}}}\] = +0.80 V and \[\ce{E^{\circ}_{{Sn^{2+}/{Sn}}}}\] = −0.14 V, the standard emf of the cell \[\ce{Sn | Sn^2+ || Ag+ | Ag}\], will be ______.
0.66 V
0.80 V
0.94 V
1.80 V
The hydrogen electrode is dipped in a solution of pH = 3 at 25°C. The reduction potential of the electrode would be ______.
0.177 V
−0.177 V
0.087 V
0.059 V
The standard EMF for the cell reaction,
\[\ce{Zn + Cu^{2+} -> Zn^{2+} + Cu}\],
is 1.10 V at 25°C. The EMF of the cell reaction, when 0.1 M Cu2+ and 0.1 M Zn2+ solutions are used at 25°C is:
1.10 V
0.110 V
−1.10 V
−0.110 V
The number of Faradays required to reduce one mol of Cu2+ to metallic copper is ______.
one
two
three
four
A standard hydrogen electrode has zero electrode potential because ______.
hydrogen is easiest to oxidise.
the electrode potential is assumed to be zero.
hydrogen has only one electron.
hydrogen is the lightest element.
The standard reduction potential values of three metallic cations, X, Y, Z are 0.52, −3.03 and −1.18 V respectively. The order of reducing power of the corresponding metals is ______.
Y > Z > X
X > Y > Z
Z > Y > X
Z > X > Y
A gas X at 1 atm is bubbled through a solution containing a mixture of 1 MY− and 1 MZ− at 25°C. If the reduction potential of Z > Y > X, then ______.
Y will oxidise X and not Z
Y will oxidise Z and not X
Y will oxidise both X and Z
Y will reduce both X and Z
For the electrochemical cell:
\[\ce{M | M+ || X- | X}\];
\[\ce{E^{\circ}_{{M^{+}/{M}}}}\] = 0.44 V,
\[\ce{E^{\circ}_{X/X^-}}\] = 0.33 V
Which of the following is TRUE for this data?
\[\ce{E^{\circ}_{cell}}\] = −0.77V
\[\ce{E^{\circ}_{cell}}\] = 0.77V
\[\ce{M+ + X- -> M + X}\] is a spontaneous reaction.
\[\ce{M + X -> M+ + X-}\] is a spontaneous reaction.
At 25°C, the standard e.m.f. of a cell having reaction involving two electron change is found to be 0.295 V. The equilibrium constant of the reaction is ______.
29.5 × 10−2
10
1 × 1010
29.5 × 1010
Saturated solution of KNO3 is used to make salt bridge because:
velocity of K+ is greater than that of \[\ce{NO^-_3}\].
velocity of \[\ce{NO^-_3}\] is greater than that of K+.
velocities of both K+ and \[\ce{NO^-_3}\] are nearly the same.
KNO3 is highly soluble in water.
The correct order of equivalent conductance at infinite dilution of LiCl, NaCl and KCl is ______.
LiCl > NaCl > KCl
KCl > NaCl > LiCl
NaCl > KCl > LiCl
LiCl > KCl > NaCl
For a cell given below:
\[\ce{Ag | Ag+ || Cu^2+ | Cu}\]
\[\ce{Ag+ + e- -> Ag}\]; E° = x
\[\ce{Cu^2+ + 2e- -> Cu}\]; E° = y
\[\ce{E{^{\circ}_{cell}}}\] is ______.
x + 2y
2x + y
y − x
y − 2x
Conductivity (siemens, S) is directly proportional to area of the vessel and the concentration of the solution in it and is inversely proportional to the length of the vessel, then constant of proportionality is expressed in ______.
S m mol−1
S2 m2 mol−2
S m2 mol−1
S2 m2 mol
When during electrolysis of a solution of AgNO3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposited on the cathode will be ______.
10.8 g
21.6 g
108 g
1.08 g
For the redox reaction,
\[\ce{Zn_{(s)} + Cu^2+ (0.1 M) -> Zn^2+ (1M) + Cu_{(s)}}\],
taking place in a cell, \[\ce{E{^{\circ}_{cell}}}\] is 1.10 volt. Ecell for the cell will be: \[\ce{(2.303 \frac{RT}{F} = 0.0591)}\]
2.14 V
1.80 V
1.07 V
0.82 V
For a cell reaction in involving a two electron change, the Stanford emf of the cell is found to be 0.295 V at 25°C. The equilibrium constant of the reaction at 25°C will be:
1 × 10−10
29.5 × 10−2
10
1 × 1010
Standard reduction electrode potentials of three metals A, B and C are +0.5 V, −3.0 V and −1.2 V respectively. The reducing power of these metals is ______.
B > C > A
A > B > C
C > B > A
A > C > B
In a hydrogen-oxygen fuel cell, combustion of hydrogen occurs to ______.
Produce high purity water
Created defection difference between two electrodes
Generated heat
Remove absorbed oxygen form electron surface
Consider the following E° values:
\[\ce{E^{\circ}_{{Fe^{3+}/{Fe^{2+}}}}}\] = + 0.77 V
\[\ce{E^{\circ}_{{Sn^{2+}/{Sn}}}}\] = −0.14 V.
Under standard conditions the potential for the reaction:
\[\ce{Sn_{(s)} + 2Fe^{3+}_{ (aq)} -> 2Fe^{2+}_{ (aq)} + Sn^{2+}_{ (aq)}}\] is:
1.68 V
1.40 V
0.91 V
0.63 V
The standard emf of a cell, involving one electron charge is found to be 0.591 V at 25°C. The equilibrium constant of the reaction is ______. (F = 96,500 C mol−1)
1.0 × 101
1.0 × 105
1.0 × 1010
1.0 × 1030
The limiting molar conductivities Λ° for NaCl, KBr and KCl are 126, 152 and 150 S cm2 mol–1 respectively. The limiting molar conductivity Λ° for NaBr is ______.
278 S cm2 mol–1
176 S cm2 mol–1
128 S cm2 mol–1
302 S cm2 mol–1
In a cell that utilises the reaction \[\ce{Zn_{(s)} + 2H{^{+}_{(aq)}} -> Zn{^{2+}_{(aq)}} + H2_{(g)}}\] addition of H2SO4 to cathode compartment will ______.
lower the E and shift equilibrium to the left.
lower the E and shift the equilibrium to the right.
increase the E and shift the equilibrium to the right.
increase the E and shift the equilibrium to the left.
The \[\ce{E^{\circ}_{M^{3+}/M^{2+}}}\] values for Cr, Mn, Fe and Co are −0.41, +1.57, +0.77 and +1.97 V respectively. For which one of these metals the change in oxidation state from +2 to +3 is easiest?
Cr
Mn
Fe
Co
Aluminium oxide may be electrolysed at 1000°C to furnish aluminium metal.
(Atomic mass = 27 amu; 1 Faraday = 96,500 coulombs)
The cathode reaction is:
\[\ce{Al^3+ + 3e- -> Al^0}\]
To prepare 5.12 kg of aluminium metal by this method would require:
5.49 × 101 C of electricity
5.49 × 104 C of electricity
1.83 × 107 C of electricity
5.49 × 107 C of electricity
Given the data at 25°C,
\[\ce{Ag + I- -> AgI + e-}\]; E° = – 0.152 V
\[\ce{Ag -> Ag+ + e-}\]; E° = – 0.800 V.
What is the value of Ksp for AgI?
\[\ce{\left(2.303 \frac{RT}{F} = 0.059 V\right)}\]
−8.12
+8.612
−37.83
−16.13
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1 M is 100 Ω. The conductivity of this solution is 1.29 S m−1. Resistance of the same cell when filled with 0.02 M of the same solution is 520 Ω. The molar conductivity of 0.02 M solution of the electrolyte will be ______.
124 × 10−4 S m2 mol−1
1240 × 10−4 S m2 mol−1
1.24 × 10−4 S m2 mol−1
12.4 × 10−4 S m2 mol−1
The cell, \[\ce{Zn | Zn^{2+} (1 M) || Cu^{2+} (1 M) | Cu}\], (\[\ce{E^{\circ}_{cell} = 1.10 V}\]), was allowed to be completely discharged at 298 K. The relative concentration of Zn2+ to \[\ce{Cu^{2+}\left(\frac{[Zn^{2+}]}{[Cu{2+}]}\right)}\] is:
antilog (24.08)
37.3
1037.3
9.65 × 104
The equivalent conductances of two strong electrolytes at infinite dilution in H2O (where ions move freely through a solution) at 25°C are given below:
\[\ce{\Lambda^{\circ}_{CH_3COONa}}\] = 91.0 S cm2/equiv.
\[\ce{\Lambda^{\circ}_{HCl}}\] = 426.2 S cm2/equiv.
What additional information/quantity does one need to calculate Λ° of an aqueous solution of acetic acid?
Λ° of NaCl
Λ° of CH3COOK
The limiting equivalent conductance of H+.
Λ° of chloroacetic acid (ClCH2COOH)
Given \[\ce{E^{\circ}_{Cr^{3+}/Cr}}\] = −0.72 V, \[\ce{E^{\circ}_{Fe^{2+}/Fe}}\] = −0.42 V. The potential for the cell \[\ce{Cr | Cr^{3+} (0.1 M) || Fe^{2+} (0.01 M) | Fe}\] is:
0.26 V
0.339 V
−0.339 V
−0.26 V
For the reduction of silver ions with copper metal, the standard cell potential was found to be +0.46 V at 25°C. The value of standard Gibbs energy, ΔG° will be ______. (F = 96500 C mol−1)
−89.0 kJ
−89.0 J
−44.5 kJ
−98.0 kJ
Standard electrode potential of three metals X, Y and Z are –1.2 V, +0.5 V and –3.0 V, respectively. The reducing power of these metals will be ______.
Y > Z > X
X > Y > Z
Z > X > Y
Z > X > Y
Y > X > Z
If the \[\ce{E^{\circ}_{cell}}\] for a given reaction has a negative value, then which of the following gives the correct relationships for the values of ΔG° and Keq?
ΔG° < 0; Keq > 1
ΔG° < 0; Keq < 1
ΔG° > 0; Keq < 1
ΔG° > 0; Keq > 1
The electrode potentials for \[\ce{Cu{^{2+}_{(aq)}} + e- -> Cu{^+_{(aq)}}}\] and \[\ce{Cu{^{+}_{(aq)}} + e- -> Cu_{(s)}}\] are +0.15 V and +0.50 V respectively. The value of \[\ce{E^{\circ}_{{Cu^{2+}/{Cu}}}}\] will be:
0.325 V
0.650 V
0.150 V
0.500 V
A buffer solution is prepared in which the concentration of NH3 is 0.30 M and the concentration of \[\ce{NH^+_4}\] is 0.20 M. If the equilibrium constant, Kb for NH3 equals 1.8 × 10−5, what is the pH of this solution? (log 2.7 = 0.43)
9.43
11.72
8.73
9.08
Standard electrode potential for Sn4+/Sn2+ couple is +0.15 V and that for the Cr3+/Cr couple is −0.74. These two couples in their standard state are connected to make a cell. The cell potential will be ______.
+0.89 V
+0.18 V
+1.83 V
+1.199 V
\[\ce{\Lambda^{\circ}_m(NH_4OH)}\] [i.e., \[\ce{\Lambda^{\circ}_m(NH4OH)}\]] is equal to ______.
\[\ce{\Lambda^{\circ}_m(NH4Cl) + \Lambda^{\circ}_m(NaCl) - \Lambda^{\circ}_m(NaOH)}\]
\[\ce{\Lambda^{\circ}_m(NaOH) + \Lambda^{\circ}_m(NaCl) - \Lambda^{\circ}_m(NH4Cl)}\]
\[\ce{\Lambda^{\circ}_m(NH4OH) + \Lambda^{\circ}_m(NH4Cl) - \Lambda^{\circ}_m(HCl)}\]
\[\ce{\Lambda^{\circ}_m(NH4Cl) + \Lambda^{\circ}_m(NaOH) - \Lambda^{\circ}_m(NaCl)}\]
When 0.1 mol \[\ce{MnO^{2-}_4}\] is oxidised, the quantity of electricity required to completely oxidise \[\ce{MnO^{2-}_4}\] to \[\ce{MnO^-_4}\] is ______.
96500 C
2 × 96500 C
9650 C
96.50 C
The weight of silver (at. wt. = 108) displaced by a quantity of electricity which displaces 5600 mL of O2 at STP will be ______.
5.4 g
10.8 g
54.0 g
108.0 g
Resistance of 0.2 M solution of an electrolyte is 50 Ω. The specific conductance of the solution is 1.4 S m−1. The resistance of 0.5 M solution of the same electrolyte is 280 Ω. The molar conductivity of 0.5 M solution of the electrolyte in S mol−1 is ______.
5 × 10−4
5 × 10−3
5 × 103
5 × 102
The equivalent conductance of NaCl at concentration C and at infinite dilution are λc and λ∞, respectively. The correct relationship between λc and λ∞ is given as:
λc = λ∞ + (B)C
λc = λ∞ − (B)C
λc = `lambda_infty - (B) sqrt C`
λc = `lambda_infty + (B) sqrt C`
The metal that cannot be obtained by the electrolysis of an aqueous solution of its salts is ______.
Ag
Ca
Cu
Cr
Given below are the half-cell reactions:
\[\ce{Mn^2+ + 2e- -> Mn}\], E° = −1.18 V
\[\ce{2(Mn^3+ + e- -> Mn^2+)}\], E° = +1.51 V
The E° for \[\ce{3Mn^2+ -> Mn + 2Mn^3+}\] will be ______.
−2.69 V, the reaction will not occur.
−2.69 V, the reaction will occur.
−0.33 V, the reaction will not occur.
−0.33 V, the reaction will occur.
The electrochemical equivalent is the amount of substance which gets deposited from its solution on passing electrical charge equal to ______.
96,500 coulombs
1 coulomb
60 coulombs
965 coulombs
Two Faraday of electricity is passed through a solution of CuSO4. The mass of copper deposited at the cathode is ______. (at. mass of Cu = 63.5 amu)
0 g
63.5 g
2 g
127 g
Galvanization is applying a coating of ______.
Cu
Zn
Pb
Cr
The pressure of H2 required to make the potential of H2-electrode zero in pure water at 298 K is ______.
10−14 atm
10−12 atm
10−10 atm
10−4 atm
In the electrochemical cell, Zn | ZnSO4 (0.01 M) || CuSO4 (1.0 M) | Cu, the emf of this Daniell cell is E1. When the concentration of ZnSO4 is changed to 1.0 M and that of CuSO4 changed to 0.01 M, the emf changes to E2. From the following, which one is the relationship between E1 and E2?
`("Given", (RT)/F = 0.059)`
E1 = E2
E1 < E2
E1 > E2
E2 = 0 ≠ E1
Given: \[\ce{E^{\circ}_{Cl_2/Cl^-}}\] = 1.36 V, \[\ce{E^{\circ}_{Cr^{3+}/Cr}}\] = −0.74 V
\[\ce{E^{\circ}_{Cr_2O^{2-}_7/Cr^{3+}}}\] = 1.33 V, \[\ce{E^{\circ}_{MnO^-_4/Mn^{2+}}}\] = 1.51 V
Among the following, the strongest reducing agent is:
Cr3+
Cl−
Cr
Mn2+
How long (approximate) should water be electrolysed by passing through 100 amperes current so that the oxygen is released can completely burn 27.66 g of diborane?
(Atomic weight of B = 10.8 u)
6.4 hours
0.8 hours
3.2 hours
1.6 hours
Consider the change in the oxidation state of Bromine corresponding to different emf values as shown in the expression below:
\[\ce{BrO^-_4 ->[1.82 V] BrO^-_3 ->[1.5 V] HBrO ->[1.595 V] Br2 ->[1.0652 V] Br^-}\]
Then the species undergoing disproportionation is:
Br2
\[\ce{BrO^-_4}\]
\[\ce{BrO^-_3}\]
HBrO
Consider the statements S1 and S2.
S1: Conductivity always increases with decrease in the concentration of electrolyte.
S2: Molar conductivity always increases with decrease in the concentration of electrolyte.
The correct option among the following is:
Both S1 and S2 are wrong.
S1 is correct and S2 is wrong.
Both S1 and S2 are correct.
S1 is wrong and S2 is correct.
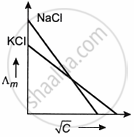
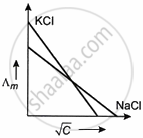
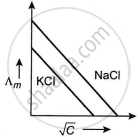
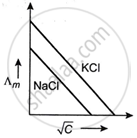
Which one of the following graphs between molar conductivity (Λm) versus `sqrt C` is correct?
Following limiting molar conductivities are given as:
\[\ce{\lambda^{\circ}_{m} (H2SO4)}\] = x S cm2 mol−1
\[\ce{\lambda{^{\circ}_{m}} (K2SO4)}\] = y S cm2 mol−1
\[\ce{\lambda{^{\circ}_{m}} (CH3COOK)}\] = z S cm2 mol−1
\[\ce{\lambda^{\circ}_{m}}\] (in S cm2 mol−1) for CH3COOH will be:
\[\ce{\frac{(x - y)}{2} + z}\]
x − y + 2z
x + y + z
x − y + z
The standard electrode potential (E°) values of Al3+/Al, Ag+/Ag, K+/K and Cr3+/Cr are −1.66 V, 0.80 V, −2.93 V and −0.74 V, respectively. The correct decreasing order of reducing power of the metal is ______.
Al > K > Ag > Cr
Ag > Cr > Al > K
K > Al > Cr > Ag
K > Al > Ag > Cr
For the cell reaction:
\[\ce{2Fe^{3+}_{ (aq)} + 2l^-_{ (aq)} -> 2Fe^{2+}_{ (aq)} + l2_{(aq)}}\]
\[\ce{E{^{\circ}_{cell}}}\] = 0.24 V at 298 K. The standard Gibbs energy (ΔrG°) of the cell reaction is:
[Given that Faraday constant, F = 96500 C mol−1]
−23.16 kJ mol−1
46.32 kJ mol−1
23.16 kJ mol−1
−46.32 kJ mol−1
For a cell involving one electron \[\ce{E^{\circ}_{cell}}\] = 0.59 V at 298 K, the equilibrium constant for the cell reaction is:
[Given that \[\ce{\frac{2.303 RT}{F}}\] = 0.059 V at T = 298 K]
1.0 × 102
1.0 × 105
1.0 × 1010
1.0 × 1030
On electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be ______.
Hydrogen gas
Oxygen gas
Hydrogen sulphide (H2S) gas
Sulphur dioxide (SO2) gas
The number of Faradays (F) required to produce 20 g of calcium from molten CaCl2 (Atomic mass of Ca = 40 g mol−1) is ______.
1
2
3
4
Let CNaCl and \[\ce{C{_{BaSO_4}}}\] be the conductances (in S) measured for saturated aqueous solutions of NaCl and BaSO4 respectively, at a temperature T. Which of the following is false?
Ionic mobilities of ions from both salts increase with T.
CNaCl >> \[\ce{C{_{BaSO_4}}}\], at a given T
\[\ce{C{_{BaSO_4}}}\] (T2) > \[\ce{C{_{BaSO_4}}}\] (T1) for T2 > T1
CNaCl (T2) > CNaCl (T1) for T2 > T1
250 mL of a waste solution obtained from the workshop of a goldsmith contains 0.1 M AgNO3 and 0.1 M AuCl. The solution was electrolyzed at 2 V by passing a current of 1 A for 15 minutes. The metal/metals electrodeposited will be ______.
(\[\ce{E^{\circ}_{Ag^+/Ag}}\] = 0.80 V, \[\ce{E^{\circ}_{Au^{2+}/Au}}\] = 1.69 V)
silver and gold in proportion to their atomic weights
silver and gold in equal mass proportion
only silver
only gold
The molar conductance of NaCl, HCl and CH3COONa at infinite dilution are 126.45, 426.16 and 91.0 S cm2 mol−1 respectively. The molar conductance of CH3COOH at infinite dilution is ______.
Choose the right option for your answer.
540.48 S cm2 mol−1
201.28 S cm2 mol−1
390.71 S cm2 mol−1
698.28 S cm2 mol−1
The pKb of dimethylamine and pKa of acetic acid are 3.27 and 4.77 respectively at T(K). The correct option for the pH of dimethylammonium acetate solution is ______.
6.25
8.50
5.50
7.75
The molar conductivity of 0.007 M acetic acid is 20 S cm2 mol−1. What is the dissociation constant of acetic acid? Choose the correct option.
\[\begin{array}{cc}
\end{array}\]\[\begin{bmatrix}
\ce{\Lambda^{\circ}_{H^+} = 350 S cm^2 mol^{-1}}\\
\ce{\Lambda^{\circ}_{CH_3COO^-} = 50 S cm^2 mol^{-1}}
\end{bmatrix}\]
2.50 × 10−5 mol L−1
1.75 × 10−4 mol L−1
2.50 × 10−4 mol L−1
1.75 × 10−5 mol L−1
Some standard electrode potentials at 298 K are given below:
| Pb+2/Pb | −0.13 V |
| Ni+2/Ni | −0.24 V |
| Cd+2/Cd | −0.40 V |
| Fe+2/Fe | −0.44 V |
To a solution containing 0.001 M of X+2 and 0.1 M of Y+2, the metal rods X and Y are inserted (at 298 K) and connected by a conducting wire. This resulted in dissolution of X. The correct combination(s) of X and Y respectively is (are):
(Given R = 8.314 J K−1 mol−1, F = 96500 (mol−1)
Cd and Ni
Cd and Fe
Ni and Pb
Ni and Fe
At 298 K, the standard electrode potentials of Cu+2/Cu, Zn+2/Zn, Fe+2/Fe and Ag+/Ag are 0.34 V, −0.76 V, −0.44 V and −0.80 V respectively.
On the basis of standard electrode potential, predict which of the following reaction cannot occur?
\[\ce{CuSO4_{(aq)} + Fe_{(s)} -> FeSO4_{(aq)} + Cu_{(s)}}\]
\[\ce{FeSO4_{(aq)} + Zn_{(s)} -> ZnSO4_{(aq)} + Fe_{(s)}}\]
\[\ce{2CuSO4_{(aq)} + 2Ag_{(s)} -> 2Cu_{(s)} + Ag2SO4_{(aq)}}\]
\[\ce{CuSO4_{(aq)} + Zn_{(s)} -> ZnSO4_{(aq)} + Cu_{(s)}}\]
Given below are half cell reactions:
\[\ce{MnO^-_4 + 8H+ + 5e- -> Mn^{+2} + 4H2O}\]
\[\ce{E^{\circ}_{Mn^{+2}/MnO^-_4}}\] = −1.510 V
\[\ce{\frac{1}{2} O2 + 2H+ + 2e- -> H2O}\]
\[\ce{E^{\circ}_{O_2/H_2O}}\] = +1.223 V
Will the permanganate ion \[\ce{MnO^-_4}\] liberate O2 from water in presence of an acid?
No, because \[\ce{E^{\circ}_{cell}}\] = −0.287 V
Yes, because \[\ce{E^{\circ}_{cell}}\] = +2.733 V
No, because \[\ce{E^{\circ}_{cell}}\] = −2.733 V
Yes, because \[\ce{E^{\circ}_{cell}}\] = +0.287 V
Find the emf of the cell in which the following reaction takes place at 298 K.
\[\ce{Ni_{(s)} + 2Ag+ (0.001 M) -> Ni^{+2} (0.001 M) + 2Ag_{(s)}}\]
(Given that \[\ce{E{^{\circ}_{cell}}}\] = 1.05 V, \[\ce{\frac{2.303 RT}{F}}\] = 0.059 at 298 K)
1.385 V
0.9615 V
1.05 V
1.0385 V
The `((del E)/(del T))_P`of different types of half cells are as follows:
| A | B | C | D |
| 1 × 10−4 | 2 × 10−4 | 0.1 × 10−4 | 0.2 × 10−4 |
(where E is the electromotive force)
Which of the above half cells would be preferred to be used as reference electrode?
A
B
C
D
The correct order of reduction potentials of the following pairs is:
- Cl2/Cl
- I2/I−
- Ag+/Ag
- Na+/Na
- Li+/Li
Choose the correct answer from the options given below:
A > C > B > D > E
A > B > C > D > E
A > C > B > E > D
A > B > C > E > D
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry FILL IN THE BLANKS TYPE QUESTIONS [Pages 205 - 206]
The conduction of current by a conductor is either due to flow of ______ or due to the movement of ______.
The electrolytic conduction ______ with a rise in temperature.
The fraction of total number of moles which undergoes dissociation is called ______.
The ions remain ______ in solution.
Higher the interaction between solute and solvent particles ______ is the salvation of ions and ______ is the current conducted by ions.
The properties of an electrolytic solution are the properties of ______ present in the solution.
The degree of dissociation of a weak electrolyte is ______ proportional to the ______ of its molar concentration.
According to Ohm’s law, the potential difference across a conductor is ______ proportional to the ______ flowing through it.
The unit of conductance is ______ which is also expressed as ______.
Equivalent conductivity is the conducting power of all the ions furnished by one ______ of an electrolyte present in a definite ______ of the solution.
D.C. cannot be used to measure the electrical conductivity of an electrolytic solution because it causes ______ effect.
Conductivity water has a very ______ conductance.
According to Debye-Huckel-Onsagar equation, `Lambda_m = Lambda_m^infty -` ______.
The molar conductivity of a weak electrolytic solution is much ______ than that of a strong electrolytic solution.
The salt bridge used in a galvanic cell completes the ______ and maintains the ______ of the solutions present in the two ______.
While representing a galvanic cell, the oxidation electrode is always written at the ______ while the reduction electrode at the ______.
The electrode potential of a standard hydrogen electrode is ______.
When a SHE is connected to a Zn2+/Zn electrode, the electrons flow from ______ electrode to the ______ electrode and the current flows in the ______ direction.
The electrode systems having positive values of standard electrode potentials act as ______ when connected to a standard hydrogen electrode.
F2 is the ______ oxidising agent because it possesses the ______ value of standard electrode potential.
A metal can displace any other metal placed ______ it in the electrochemical series from its salt solution.
A cell reaction is feasible when the EMF of the cell is ______.
The cell which converts ______ energy into ______ energy is called an electrolytic cell.
The electrolysis of fused sodium chloride gives ______ at the cathode and ______ at the anode, whereas the products obtained on electrolysis of an aqueous solution of sodium chloride are ______ at the cathode and ______ at the anode.
During the electrolysis of an aqueous solution of sodium chloride, the chloride ions get oxidised at anode in preference to water due to ______.
If the cation produced from the electrolyte has higher reduction potential than that of water, the ______ will get reduced at the cathode.
The electrochemical equivalent is the amount of substance liberated by a current of ______ ampere passed for ______ second.
The quantity of electricity required to liberate one equivalent of any substance is ______ Faraday.
Reduction of 1 mole of \[\ce{MnO^-_4}\] to Mn2+ requires ______ coulombs of charge.
The cell potential of a dry cell is approximately ______ V to ______ V.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry ASSERTION-REASON TYPE QUESTIONS [Page 206]
Assertion: When a Zn | Zn2+ electrode is connected to a standard hydrogen electrode, the electrons flow from zinc electrode to the hydrogen electrode.
Reason: The standard reduction potential of zinc electrode is lower than that of hydrogen electrode.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The specific conductivity of an electrolytic solution decreases with increase in dilution.
Reason: Specific conductivity is the conductance due to the ions present in 1 cm3 of the solution.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: The electrode potential of a standard hydrogen electrode is equal to zero.
Reason: The value has been obtained on the basis of experimental observations.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: Copper can displace zinc from the solution of zinc sulphate.
Reason: Copper is placed below zinc in the electrochemical series.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Assertion: When \[\ce{E{^{\circ}_{cell}}}\] is positive, the cell reaction is feasible and proceeds spontaneously.
Reason: When \[\ce{E{^{\circ}_{cell}}}\] is positive, ΔG° is negative which makes the cell reaction feasible.
If both Assertion and Reason are CORRECT and Reason is the CORRECT explanation of the Assertion.
If both Assertion and Reason are CORRECT but Reason is not the CORRECT explanation of the Assertion.
If Assertion is CORRECT but Reason is INCORRECT.
If Assertion is INCORRECT but Reason is CORRECT.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry NUMERICAL PROBLEMS [Pages 206 - 207]
When a conductivity cell was filled with 0.02 M KCl, it had a resistance of 82.4 ohm at 25°C and when filled with 0.005 N K2SO4 it had a resistance of 326 ohm. Calculate
- cell constant
- specific conductivity, and
- equivalent conductivity of 0.005 N K2SO4 solution.
The specific conductance of 0.02 M KCl is 0.002768 ohm−1 cm−1.
The resistance of a 0.5 M solution of an electrolyte in a cell was found to be 45 ohm. Calculate the molar conductivity of the solution if the electrodes in the cell are 2.2 cm apart and have an area of 3.8 cm2.
During electrolysis, 4828.4 mg of iodine requires 3671.3 coulombs of electricity for its deposition at the anode. Calculate the value of Faraday’s constant.
Calculate the volume of Cl2 at STP produced during electrolysis of MgCl2 which produces 6.50 g of Mg. (At. wt. of Mg = 24.3)
A current of 1.70 amp is passed through 500 mL of 0.16 M solution of ZnSO4 for 230 seconds with a current efficiency of 90%. Find the molarity of Zn2+ after the deposition of zinc. Assume that the volume of the solution remains constant during electrolysis.
The reaction \[\ce{Zn_{(s)} + Co^{2+} -> Co_{(s)} + Zn^{2+}}\] occurs in a cell. Write the electrode reactions and compute the standard emf of the cell.
Given that \[\ce{E^{\circ}_{{Zn/{Zn}^{2+}}}}\] = +0.76 V and \[\ce{E^{\circ}_{{Co/{Co}^{2+}}}}\] = 0.2 V.
There are two hydrogen electrodes in a cell. The anode is in contact with the hydrogen ion concentration of 10−6 M. The emf of the cell at 25°C is 0.118 volt. Calculate the hydrogen ion concentration at cathode.
How many grams of silver could be plated out on a serving tray by electrolysis of a solution containing silver in +1 oxidation state for a period of 8.0 hours at a current of 8.46 amperes? What is the area of the tray if the thickness of the silver plating is 0.00254 cm? Density of silver is 10.5 g cm−3.
Calculate the quantity of electricity required to reduce 6.15 g of nitrobenzene to aniline if the current efficiency is 68 per cent. If potential drop across the cell is 7.0 volts, calculate the energy consumed in the process.
Calculate the pH of the following half cell solutions (at 25°C):
\[\ce{Pt, H2 (1 atm) | HCl; E_{H/H^+}}\] = 0.25 V
Calculate the pH of the following half cell solutions (at 25°C):
\[\ce{Pt, H2 (1 atm) | H2SO; E_{H/H^+}}\] = 0.3 V
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry 'NCERT TEXT-BOOK' Exercises [Pages 209 - 212]
Arrange the following metals in the order in which they displace each other from the solution of their salts.
Al, Cu, Fe, Mg and Zn.
Given the standard electrode potentials,
K+/K = −2.93 V, Ag+/Ag = 0.80 V,
Hg2+/Hg = 0.79 V
Mg2+/Mg = −2.37 V, Cr3+/Cr = −0.74 V
Arrange these metals in their increasing order of reducing power.
Depict the galvanic cell in which the reaction takes place.
\[\ce{Zn_{(s)} + 2Ag+_{ (aq)} -> Zn^{2+}_{ (aq)} + 2Ag_{(s)}}\]
Further show:
- Which of the electrode is negatively charged?
- The carriers of the current in the cell.
- Individual reaction at each electrode.
Calculate the standard cell potential of a galvanic cell in which the following reaction takes place:
\[\ce{2Cr_{(s)} + 3Cd{^{2+}_{(aq)}} -> 2Cr{^{3+}_{(aq)}} + 3Cd}\]
Calculate the ΔrG° and equilibrium constant of the reaction.
Calculate the standard cell potential of a galvanic cell in which the following reaction takes place:
\[\ce{Fe{^{2+}_{(aq)}} + Ag{^{+}_{(aq)}} -> Fe{^{3+}_{(aq)}} + Ag_{(s)}}\]
Calculate the ΔrG° and equilibrium constant of the reaction.
Write the Nernst equation and emf of the following cell at 298 K:
Mg(s) | Mg2+ (0.001 M) || Cu2+ (0.0001 M) | Cu(s)
Write the Nernst equation and emf of the following cell at 298 K:
Fe(s) | Fe2+ (0.001 M) || H+ (1 M) | H2(g) (1 bar) | Pt(s)
Write the Nernst equation and emf of the following cell at 298 K:
\[\ce{Sn_{(s)} | Sn^{2+} (0.050 M) || H^+ (0.020 M) | H2_{(g)} (1 bar) | Pt_{(s)}}\]
Write the Nernst equation and emf of the following cell at 298 K:
Pt(s) | Br− (0.010 M) | Br2(l) || H+ (0.030 M) | H2(g) (1 bar) | Pt(s)
In the button cells widely used in watches and other devices, the following reaction takes place:
\[\ce{Zn_{(s)} + Ag2O_{(s)} + H2O_{(l)} -> Zn{^{2+}_{(aq)}} + 2Ag_{(s)} + 2OH^-_{ (aq)}}\]
Determine ΔrG° and E° for the reaction.
Define conductivity for the solution of an electrolyte.
Define “Molar conductivity”.
Discuss the variation of conductivity and molar conductivity with concentration.
The conductivity of 0.20 M solution of KCl at 298 K is 0.0248 S cm−1. Calculate its molar conductivity.
The resistance of a conductivity cell containing 0.001 M KCl solution at 298 K is 1500 Ω. What is the cell constant if the conductivity of 0.001 M KCl solution at 298 K is 0.146 × 10−3 S cm−1.
The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below:
| Concentration/M | 0.001 | 0.010 | 0.020 | 0.050 | 0.100 |
| 102 × κ/S m−1 | 1.237 | 11.85 | 23.15 | 55.53 | 106.74 |
Calculate ∧m for all concentrations and draw a plot between ∧m and c1/2. Find the value of `∧_m^0`.
Conductivity of 0.00241 M acetic acid is 7.896 × 10−5 S cm−1. Calculate its molar conductivity and if `∧_m^0` for acetic acid is 390.5 S cm2 mol−1, what is its dissociation constant?
How much charge is required for the following reduction:
1 mol of Al3+ to Al?
How much charge is required for the following reduction?
1 mol of Cu2+ to Cu.
How much charge is required for the following reduction:
1 mol of \[\ce{MnO^-_4}\] to Mn2+?
How much electricity in terms of Faraday is required to produce 20 g of Ca from molten CaCl2?
(Given: Molar mass of Calcium is 40 g mol−1.)
How much electricity in terms of Faraday is required to produce 40.0 g of Al from molten Al2O3?
(Given: Molar mass of Aluminium is 27 g mol−1.)
How much electricity is required in coulomb for the oxidation of 1 mol of H2O to O2?
How much electricity is required in coulomb for the oxidation of 1 mol of FeO to Fe2O3?
A solution of Ni(NO3)2 is electrolysed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of Ni is deposited at the cathode?
Three electrolytic cells A, B, C containing solutions of ZnSO4, AgNO3 and CuSO4, respectively, are connected in series. A steady current of 1.5 amperes was passed through them until 1.45 g of silver deposited at the cathode of cell B. How long did the current flow? What mass of copper and zinc were deposited?
Using the standard electrode potentials, predict if the reaction between the following is feasible:
\[\ce{Fe^{3+}_{ (aq)}}\] and \[\ce{I^-_{ (aq)}}\]
Using the standard electrode potentials, predict if the reaction between the following is feasible:
\[\ce{Ag{^{+}_{(aq)}}}\] and Cu(aq)
Using the standard electrode potentials, predict if the reaction between the following is feasible:
\[\ce{Fe^{3+}_{ (aq)}}\] and \[\ce{Br^-_{ (aq)}}\]
Using the standard electrode potential, predict if the reaction between the following is feasible:
Ag(s) and \[\ce{Fe^{3+}_{ (aq)}}\]
Using the standard electrode potential, predict if the reaction between the following is feasible:
Br2(aq) and \[\ce{Fe^{2+}_{ (aq)}}\]
Predict the product of electrolysis in the following:
An aqueous solution of AgNO3 with silver electrodes.
Predict the product of electrolysis in the following:
An aqueous solution of AgNO3 with platinum electrodes.
Predict the product of electrolysis in the following:
A dilute solution of H2SO4 with platinum electrodes.
Predict the product of electrolysis in the following:
An aqueous solution of CuCl2 with platinum electrodes.
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी 3 Electrochemistry QUESTIONS FROM ISC EXAMINATION PAPERS [Pages 213 - 216]
Correct the following statement:
Copper displaces both zinc and silver from their solutions.
A conductivity cell has a cell constant of 0.5 cm−1. This cell when filled with 0.01 M sodium chloride solution, has a resistance of 384 ohms at 25°C. Calculate the equivalent conductivity of 0.01 M sodium chloride at 25°C.
For the cell, Cu | Cu2+ (0.13 M) || Ag+ (0.01 M) | Ag,
- Calculate the reduction potential of each electrode if the standard reduction potential for copper and silver electrodes are 0.34 V and 0.80 V respectively.
- Calculate the emf of the cell.
- Write the cell reaction.
- Is this cell reaction spontaneous? Why?
Correct the following statement:
For a strong electrolyte, the plot of equivalent conductance versus concentration of the solution is a straight line.
A current of 4 amperes is passed through a molten solution for 45 minutes. 2.977 g of metal is deposited. Calculate the charge carried by the metal cation if its atomic mass is 106.4 g mol−1.
Fill in the blanks:
As concentration of an electrolyte solution increases, its ______ conductance increases but ______ conductance decreases.
A cell is constructed by dipping a zinc rod in 0.1 M zinc nitrate solution and a lead rod in 0.2 M lead nitrate solution.
\[\ce{E^{\circ}_{Pb^{2+}/Pb}}\] = −0.13 V and \[\ce{E^{\circ}_{Zn^{2+}/Zn}}\] = −0.76 V
- Write the spontaneous cell reaction.
- Calculate standard emf and emf of the cell.
The number of Faradays which will be required to reduce 4 gram equivalents of Cu2+ to Cu metal is ______.
1
2
3
4
For the cell:
Zn | Zn2+ (a = 1) || Cu2+ (a = 1) | Cu
Given that \[\ce{E^{\circ}_{Zn/Zn^{2+}}}\] = 0.761 volt; \[\ce{E^{\circ}_{Cu^{2+}/Cu}}\] = +0.339 volt
- Write the cell reaction.
- Calculate the emf and free energy change at 298 K involved in the cell.
[Faraday’s constant, F = 96500 coulomb mol−1]
Calculate the equivalent conductivity of 1 M H2SO4, whose specific conductivity is 26 × 10−2 ohm−1 cm−1.
A current of 10 A is passed for 80 min. and 27 seconds through a cell containing dilute sulphuric acid.
- How many moles of oxygen gas will be liberated at the anode?
- Calculate the amount of zinc deposited at the cathode when another cell containing ZnSO4 solution is connected in series (Zn = 65).
Calculate:
Ecell at 25°C for the reaction:
\[\ce{Zn + Cu{^{2+}} (0.20 M) -> Zn{^{2+}} (0.50 M) + Cu}\]
Given: \[\ce{E^{\circ}_{({Zn^{2+}/{Zn}})}}\] = −0.76 volt, \[\ce{E^{\circ}_{({Cu^{2+}/{Cu}})}}\] = 0.34 volt
Give a reason for the following:
Specific conductance decreases with dilution, whereas equivalent conductance increases with dilution.
Complete the following sentence by selecting the correct alternative from the choice given:
When zinc granule is dipped in copper sulphate solution, copper is precipitated because ______.
Both copper and zinc have a positive reduction potential.
Reduction potential of copper is higher than that of zinc.
Reduction potential of zinc is higher than that of copper.
Both zinc and copper have a negative reduction potential.
fill in the blanks:
In a galvanic cell, the movement of electrons in the external circuit is from ______ to ______.
Zinc displaces hydrogen from acid solution (\[\ce{E^{\circ}_{Zn^{2+}/Zn}}\] = −0.76 V). Explain.
A 0.05 M NaOH solution offered a resistance of 31.6 ohms in a conductivity cell. If the cell constant of the conductivity cell is 0.378 cm−1, determine the molar conductivity of sodium hydroxide solution at this temperature.
Mention any two factors affecting the electrode potential of a metal.
For the following cell, calculate the emf:
\[\ce{Al | Al^{3+} (0.01 M) || Fe^{2+} (0.02 M) | Fe}\]
Given: \[\ce{E^{\circ}_{{Al^{3+}/{Al}}}}\] = −1.66 V, \[\ce{E^{\circ}_{{Fe^{2+}/{Fe}}}}\] = −0.44 V
When zinc granule is dipped in copper sulphate solution, copper is precipitated because ______.
Both copper and zinc have a positive reduction potential.
Reduction potential of copper is higher than that of zinc.
Reduction potential of zinc is higher than that of copper.
Both zinc and copper have a negative reduction potential.
State Faraday’s first law of electrolysis.
How many electrons will flow when a current of 5 amperes is passed through a solution for 200 seconds?
Consider the reaction:
\[\ce{2Ag+ + Cd -> 2Ag + Cd^{2+}}\]
The reduction potentials of Ag+/Ag and Cd2+/Cd are +0.80 volt and −0.40 volt respectively.
- Give the cell representation.
- What is the standard cell emf, E°?
- What will be the emf of the cell if concentration of Cd2+ is 0.1 M and Ag+ is 0.2 M?
- Will the cell work spontaneously for the condition given in (c) above?
Fill in the blanks:
Zinc can displace ______ from CuSO4 solution, but cannot displace ______ from MgSO4 solution.
The quantity of electricity required to deposit 1.15 g of sodium from molten NaCl (Na = 23, Cl = 35.5) is ______.
1 F
0.5 F
0.05 F
1.5 F
The conductivity of 0.2 M KCl solution is 3 × 10−2 ohm−1 cm−1. Calculate its molar conductance.
Magnesium displaces hydrogen from dilute acid solution because ______.
The oxidation potential of magnesium is less than that of hydrogen.
The reduction potential of magnesium is less than that of hydrogen.
Both magnesium and hydrogen have same oxidation potential.
Both magnesium and hydrogen have same reduction potential.
Calculate the number of coulombs required to deposit 20.25 g of aluminium (at. mass = 27) from a solution containing Al+3.
What do you understand by specific conductance of a solution?
What are the units of specific conductivity?
How is equivalent conductivity related to specific conductivity?
2.5 amperes of current is passed through copper sulphate solution for 30 minutes. Calculate the number of copper atoms deposited at the cathode (Cu = 63.54).
Four metals W, X, Y and Z have the following values of Ered:
Ered
W = −0.140 V
X = −2.93 V
Y = +0.80 V
Z = +1.50 V
Arrange them in the increasing order of reducing power.
A current liberates 0.50 g of hydrogen in 2 hours. The weight of copper (at. wt. = 63.5) deposited at the same time by the same current through copper sulphate solution is ______.
63.5 g
31.8 g
15.9 g
15.5 g
What happens when a nickel rod is dipped into a copper sulphate solution? Justify your answer.
[\[\ce{E{^{\circ}_{Ni^{+2}/Ni}}}\] = − 0.25 V and \[\ce{E{^{\circ}_{Cu^{+2}/Cu}}}\] = +0.34 V]
What is standard hydrogen electrode?
0.05 M NaOH solution offered resistance of 31.6 ohms in a conductivity cell at 298 K. If the cell constant of the cell is 0.367 cm−1, calculate the molar conductivity of the NaOH solution.
0.3605 g of a metal is deposited on the electrode by passing 1.2 amperes of current for 15 minutes through its salt solution. The atomic weight of the metal is 96. What is the valency of the metal?
How many hours does it take to reduce 3 moles of Fe3+ to Fe2+ with 2.0 A current intensity?
The specific conductance of a 0.01 M solution of acetic acid at 298 K is 1.65 × 10−4 ohm−1 cm−1. The molar conductance at infinite dilution for H+ ion and CH3COO− ion is 349.1 ohm−1 cm2 mol−1 and 40.9 ohm−1 cm2 mol−1 respectively.
Calculate:
- Molar conductance of the solution.
- The degree of dissociation of CH3COOH.
- A dissociation constant for acetic acid.
Calculate the e.m.f. of the following cell reaction at 298 K:
\[\ce{Mg_{(s)} + Cu{^{2+}} (0.0001 M) -> Mg{^{2+}} (0.001M) + Cu_{(s)}}\]
The standard potential (E°) of the cell is 2.71 V.
Consider the following cell reaction at 298 K:
\[\ce{2Ag+ + Cd -> 2Ag + Cd^{2+}}\]
The standard reduction potentials (E°) for Ag+/Ag and Cd2+/Cd are 0.80 V and −0.40 V respectively.
- Write the cell representation.
- What will be the emf of the cell if concentration of Cd2+ is 0.1 M and that of Ag+ is 0.2 M?
- Will the cell work spontaneously for the condition given in (b) above?
A 0.05 M NH4OH solution offers the resistance of 50 ohms to a conductivity cell at 298 K. If the cell constant is 0.50 cm−1 and molar conductance of NH4OH at infinite dilution is 471.4 ohm−1 cm2 mol−1, calculate:
- Specific conductance
- Molar conductance
- Degree of dissociation
Fill in the blanks:
On dilution of a solution, its specific conductance ______ while its equivalent conductance ______.
More ______ the standard reduction potential of a substance, the ______ is its ability to displace hydrogen from acids.
For a spontaneous reaction ΔG° and \[\ce{E{^{\circ}_{cell}}}\] will be respectively:
−ve and +ve
+ve and −ve
+ve and +ve
−ve and −ve
Specific conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm−1. Calculate its molar conductivity.
The following electrochemical cell is set up at 298 K:
\[\ce{Zn | Zn^{2+}_{ (aq)} (1 M) || Cu^{2+}_{ (aq)} (1 M) | Cu}\]
Given: \[\ce{E^{\circ}_{Zn^{2+}/Zn}}\] = −0.761 V, \[\ce{E^{\circ}_{Cu^{2+}/Cu}}\] = +0.339 V
Write the cell reaction.
The following electrochemical cell is set up at 298 K:
\[\ce{Zn | Zn^{2+}_{ (aq)} (1 M) || Cu^{2+}_{ (aq)} (1 M) | Cu}\]
Given: \[\ce{E^{\circ}_{Zn^{2+}/Zn}}\] = −0.761 V, \[\ce{E^{\circ}_{Cu^{2+}/Cu}}\] = +0.339 V
Calculate the emf and free energy change at 298 K.
Answer the following:
What is the effect of temperature on the ionic product of water (Kw)?
What happens to the ionic product of water (Kw) if some acid is added to it?
Name the law or principle to which the following observations confirm:
When 9650 coulombs of electricity is passed through a solution of copper sulphate, 3.175 g of copper is deposited on the cathode (at. wt. of Cu = 63.5).
Fill in the blanks:
A galvanic cell converts ______ energy into ______ energy.
Calculate the emf and ΔG° for the cell reaction at 25°C:
\[\ce{\underset{(0.1 M)}{Zn_{(s)} | Zn^{2+}_{ (aq)}} || \underset{(0.01 M)}{Cd^{2+}_{ (aq)} | Cd_{(s)}}}\]
Given \[\ce{E^{\circ}_{Zn^{2+}/Zn}}\] = −0.763 and \[\ce{E^{\circ}_{Cd^{2+}/Cd}}\] = −0.403
Define the term equivalent conductance.
Define the following term:
Corrosion of metals
The specific conductivity of a solution containing 5 g of anhydrous BaCl2 (mol. wt. = 208) in 1000 cm3 of a solution is found to be 0.0058 ohm−1 cm−1. Calculate the molar and equivalent conductivity of the solution.
What do you understand by electrochemical series?
How is the electrochemical series useful in predicting whether a metal can liberate hydrogen from acid or not?
Calculate the mass of silver deposited at cathode when a current of 2 amperes is passed through a solution of AgNO3 for 15 minutes.
(at. wt. of Ag = 108, 1 F = 96500 C)
Calculate the emf and ΔG for the cell reaction at 298 K:
Mg(s) | Mg2+ (0.1 M) || Cu2+ (0.01 M) | Cu(s)
Given: \[\ce{E^{\circ}_{cell}}\] = 2.71 V
1 F = 96500 C
Define the following term:
Specific conductance
Define the following term:
Kohlrausch’s Law.
The resistance of a conductivity cell containing 0.001 M KCl solution at 298 K is 1500 ohm. What is the cell constant and molar conductivity of 0.001 M KCl solution, if the conductivity of this solution is 0.146 × 10−3 ohm−1 cm−1 at 298 K?
Calculate the molar conductivity at infinite dilution for CH3COOH if the molar conductivity at infinite dilution for NaCl, HCl and CH3COONa are 126.45, 426.16 and 91.0 ohm−1 cm2 mol−1 respectively.
Calculate the emf and ΔG for the given cell at 25°C:
Cr(s) | Cr3+ (0.1 M) || Fe2+ (0.01 M) || Fe(s)
Given: \[\ce{E^{\circ}_{Cr^{3+}/Cr}}\] = −0.74 V, \[\ce{E^{\circ}_{Fe^{2+}/Fe}}\] = −0.44 V
(1 F = 96500 C mol−1, R = 8.314 JK−1 mol−1)
Calculate the degree of dissociation (α) of acetic acid if its molar conductivity (Λm) is 39.05 S cm2 mol−1.
(Given \[\ce{\lambda^{\circ}_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{\lambda^{\circ}_{(CH_3COO^-)}}\] = 40.95 S cm2 mol−1)
Select and write the correct alternative from the choices given below:
Which of the following statements is true for electrochemical cell?
Cations move towards zinc electrode.
Cations move towards copper electrode.
Current flows from zinc electrode to copper electrode.
Electrons flow from copper electrode to zinc electrode.
Electrochemical equivalent (z) is the amount of substance which gets deposited from its solutions on passing electrical charge equal to solutions on passing electrical charge equal to ______.
96500 coulombs
9650 coulombs
965 coulombs
1 coulomb
The standard electrode potential of Cu+2/Cu is +0.34 V and that of Cr+3/Cr is −0.74 V. These two electrodes are connected in their standard state to make an electrochemical cell. What will be the standard electrode potential (E°) of this cell?
1.19 V
1.08 V
0.69 V
1.83 V
The ionic conductance at infinite dilution for Ba+2 and Cl− ions are 127 ohm−1 cm2 mol−1 and 76 ohm−1 cm2 mol−1 respectively. What will be the molar conductance of BaCl2 at infinite dilution?
139.5 ohm−1 cm2 mol−1
279.0 ohm−1 cm2 mol−1
203.0 ohm−1 cm2 mol−1
101.5 ohm−1 cm2 mol−1
A conductivity cell is filled with 0.05 M NaOH solution offering a resistance of 31.6 ohm. If the cell constant of the cell is 0.347 cm−1, calculate the following:
The value of specific conductance:
1.29 × 10−4 ohm−1 cm−1
12.9 × 10−4 ohm−1 cm−1
1.16 × 10−2 ohm−1 cm−1
11.6 × 10−2 ohm−1 cm−1
A conductivity cell is filled with 0.05 M NaOH solution offering a resistance of 31.6 ohm. If the cell constant of the cell is 0.347 cm−1, calculate the following:
The value of molar conductance:
232.20 ohm−1 cm2 mol−1
23.22 ohm−1 cm2 mol−1
119.07 ohm−1 cm2 mol−1
165.36 ohm−1 cm2 mol−1
The standard electrode potential for Sn+4/Sn+2 couple is +0.15 V and that for the Cr+3/Cr couple is −0.74 V. These two couples in their standard states are connected to make a cell (1 Faraday = 96500 mol−1)
What will be the value of \[\ce{E{^{\circ}_{cell}}}\]?
+1.19 V
+0.89 V
+0.18 V
+1.83 V
The standard electrode potential for Sn+4/Sn+2 couple is +0.15 V and that for the Cr+3/Cr couple is −0.74 V. These two couples in their standard states are connected to make a cell. (1 Faraday = 96500 mol−1)
What will be the value of standard Gibbs energy (ΔG°)?
−650.3 kJ
−515.3 kJ
−226.4 kJ
−406.8 kJ
A solution containing 2 g of anhydrous barium chloride in 400 cm3 of water has a specific conductivity of 0.0058 S cm−1. (at. wt. of Ba = 137, Cl = 35.5)
What is the molarity of the above solution?
0.204 M
0.024 M
0.420 M
4.021 M
A solution containing 2 g of anhydrous barium chloride in 400 cm3 of water has a specific conductivity of 0.0058 S cm−1. (at. wt. of Ba = 137, Cl = 35.5)
What is the molar conductivity of the above solution?
241.67 S cm2/mol
261.47 S cm2/mol
247.17 S cm2/mol
361.47 S cm2/mol
Solutions for 3: Electrochemistry
![Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 3 - Electrochemistry Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 3 - Electrochemistry - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12-isc_6:a6407550bf9a46188b122e0d354548f8.png)
Nootan solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 3 - Electrochemistry
Shaalaa.com has the CISCE Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Nootan solutions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE 3 (Electrochemistry) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Nootan textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी chapter 3 Electrochemistry are Overview of Electrochemistry.
Using Nootan केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी solutions Electrochemistry exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Nootan Solutions are essential questions that can be asked in the final exam. Maximum CISCE केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी students prefer Nootan Textbook Solutions to score more in exams.
Get the free view of Chapter 3, Electrochemistry केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी additional questions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ आईएससी CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.