Advertisements
Advertisements
प्रश्न
Discuss the variation of conductivity and molar conductivity with concentration.
Advertisements
उत्तर
Both conductivity and molar conductivity change with changes in the concentration of the electrolyte. Conductivity always decreases as the concentration of both weak and strong electrolytes decreases. This can be explained by the fact that with dilution, the number of ions carrying electric current per unit volume decreases. The conductivity of a solution at any concentration is the conductivity of a unit volume of the solution placed between two platinum electrodes having a unit cross-sectional area and situated at a unit distance from each other.
This is clear from the following equation:
C = `(kappa A)/l` = κ ...(Both A and l are in appropriate units m or cm)
The molar conductivity of a solution at a given concentration is the conductivity of volume (V) of the solution containing one mole of electrolyte dissolved in it and placed between two electrodes of cross-sectional area (A), located at a unit distance from each other. So,
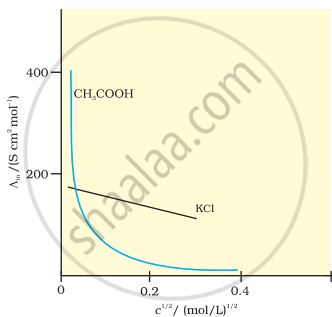
Plot of c1/2 versus molar conductivity for potassium chloride (a strong electrolyte) in aqueous solution.
∧m = `(kappa A)/l` = κ
Since l = 1 and A = V (volume in which one mole of electrolyte is dissolved)
∧m = κ V
Molar conductivity increases with decreasing concentration. This is because the total volume (V) in which one mole of electrolyte is present also increases. It has been found that, upon dilution of the solution, the increase in volume is much greater than the decrease in κ.
Strong Electrolytes: For strong electrolytes, the value of ∧m increases gradually with dilution, and it can be represented by the following equation:
∧m = `Lambda_m^0 - Ac^(1//2)`
It can be seen that if ∧m is plotted against c1/2, we get a straight line with intercept A and slope equal to ‘A’. The value of constant ‘A’ at a given solvent and temperature depends on the type of electrolyte, i.e., on the charges of the cation and anion produced on dissociation of the electrolyte in solution. Thus, NaCl, CaCl2, and MgSO4 are known as 1-1, 2-1 and 2-2 electrolytes, respectively. The value of ‘A’ is the same for all electrolytes of the same type.
APPEARS IN
संबंधित प्रश्न
The conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm−1. Calculate its molar conductivity.
The conductivity of 0.20 mol L−1 solution of KCl is 2.48 × 10−2 S cm−1. Calculate its molar conductivity and degree of dissociation (α). Given λ0 (K+) = 73.5 S cm2 mol−1 and λ0 (C1−) = 76.5 S cm2 mol−1.
Define the following terms: Molar conductivity (⋀m)
The molar conductivity of 0.025 mol L−1 methanoic acid is 46.1 S cm2 mol−1. Calculate its degree of dissociation and dissociation constant. Given \[\ce{λ^0_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{λ^0_{(HCOO^-)}}\] = 54.6 S cm2 mol−1.
Write mathematical expression of molar conductivity of the given solution at infinite dilution.
The S.I. unit of cell constant for conductivity cell is __________.
Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes because of the fact that ____________.
Molar conductivity of ionic solution depends on:
(i) temperature.
(ii) distance between electrodes.
(iii) concentration of electrolytes in solution.
(iv) surface area of electrodes.
When acidulated water (dil.H2SO4 solution) is electrolysed, will the pH of the solution be affected? Justify your answer.
Why on dilution the m Λm of \[\ce{CH3COOH}\] increases very fast, while that of \[\ce{CH3COONa}\] increases gradually?
Assertion: Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason: For weak electrolytes degree of dissociation increases with dilution of solution.
Which of the following halogen acids is the strongest reducing agent?
The molar conductivity of CH3COOH at infinite dilution is 390 Scm2/mol. Using the graph and given information, the molar conductivity of CH3COOK will be:

Which of the following solutions of KCl will have the highest value of molar conductivity?
Assertion (A) : Conductivity decreases with decrease in concentration of electrolyte.
Reason (R) : Number of ions per unit volume that carry the current in a solution decreases on dilution.
The unit of molar conductivity is ______.
Which of the following solutions will have the highest conductivity at 298 K?
The resistance of a conductivity cell with a 0.1 M KCl solution is 200 ohm. When the same cell is filled with a 0.02 M NaCl solution, the resistance is 1100 ohm. If the conductivity of 0.1 M KCl solution is 0.0129 ohm-1 cm-1, calculate the cell constant and molar conductivity of 0.02 M NaCl solution.
