Advertisements
Advertisements
प्रश्न
The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below:
| Concentration/M | 0.001 | 0.010 | 0.020 | 0.050 | 0.100 |
| 102 × κ/S m−1 | 1.237 | 11.85 | 23.15 | 55.53 | 106.74 |
Calculate ∧m for all concentrations and draw a plot between ∧m and c1/2. Find the value of `∧_m^0`.
Advertisements
उत्तर
| Concentration (M) | C1/2 (M1/2) |
κ(S m−1) | κ(S cm−1) | `Lambda_m = (kappa xx 1000)/M` (S cm2 mol−1) |
| 0.001 | 0.032 | 1.237 × 10−2 | 1.237 × 10−4 | `Lambda_m = (1.237 xx 10^-4 xx 1000)/0.001` = 123.7 |
| 0.010 | 0.100 | 11.85 × 10−2 | 11.85 × 10−4 | `Lambda_m = (11.85 xx 10^-4 xx 1000)/(0.010)` = 118.5 |
| 0.020 | 0.141 | 23.15 × 10−2 | 23.15 × 10−4 | `Lambda_m = (23.15 xx 10^-4 xx 1000)/0.020` = 115.8 |
| 0.050 | 0.224 | 55.53 × 10−2 | 55.53 × 10−4 | `Lambda_m = (55.53 xx 10^-4 xx 1000)/(0.050)` = 111.1 |
| 0.100 | 0.316 | 106.74 × 10−2 | 106.74 × 10−4 | `Lambda_m = (106.74 xx 10^-4 xx 1000)/(0.100)` = 106.7 |
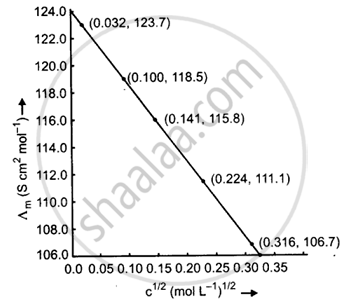
When the straight line is drawn backwards, it meets the ∧m axis at 124.0 S cm2 mol−1, which is the value of `∧_"m"^0`.
APPEARS IN
संबंधित प्रश्न
State Kohlrausch Law.
The conductivity of 0.001 mol L-1 solution of CH3COOH is 3.905× 10-5 S cm-1. Calculate its molar conductivity and degree of dissociation (α) Given λ°(H+)= 349.6 S cm2 mol-1 and λ°(CH3COO)= 40.9S cm2mol-1.
10.0 grams of caustic soda when dissolved in 250 cm3 of water, the resultant gram molarity of solution is _______.
(A) 0.25 M
(B) 0.5 M
(C) 1.0 M
(D) 0.1 M
Write mathematical expression of molar conductivity of the given solution at infinite dilution.
Calculate the degree of dissociation (α) of acetic acid if its molar conductivity (Λm) is 39.05 S cm2 mol−1.
(Given \[\ce{\lambda^{\circ}_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{\lambda^{\circ}_{(CH_3COO^-)}}\] = 40.95 S cm2 mol−1)
Define the following terms :
Limiting molar conductivity
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:

Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes because of the fact that ____________.
When acidulated water (dil.H2SO4 solution) is electrolysed, will the pH of the solution be affected? Justify your answer.
Write the cell reaction of a lead storage battery when it is discharged. How does the density of the electrolyte change when the battery is discharged?
Why on dilution the m Λm of \[\ce{CH3COOH}\] increases very fast, while that of \[\ce{CH3COONa}\] increases gradually?
The limiting molar conductivities Λ° for NaCl, KBr and KCl are 126, 152 and 150 S cm2 mol–1 respectively. The limiting molar conductivity Λ° for NaBr is ______.
The molar conductance of NaCl, HCl and CH3COONa at infinite dilution are 126.45, 426.16 and 91.0 S cm2 mol−1 respectively. The molar conductance of CH3COOH at infinite dilution is ______.
Choose the right option for your answer.
The molar conductivity of 0.007 M acetic acid is 20 S cm2 mol−1. What is the dissociation constant of acetic acid? Choose the correct option.
\[\begin{array}{cc}
\end{array}\]\[\begin{bmatrix}
\ce{\Lambda^{\circ}_{H^+} = 350 S cm^2 mol^{-1}}\\
\ce{\Lambda^{\circ}_{CH_3COO^-} = 50 S cm^2 mol^{-1}}
\end{bmatrix}\]
The molar conductance of NaCl, HCl, and CH3COONa at infinite dilution are 126.45, 426.16, and 91.0 S cm2 mol−1 respectively. The molar conductance of CH3COOH at infinite dilution is. Choose the right option for your answer.
Given below are two statements:
Statements I: The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH3COOH (weak electrolyte).
Statement II: Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:
Conductivity of 2 × 10−3 M methanoic acid is 8 × 10−5 S cm−1. Calculate its molar conductivity and degree of dissociation if `∧_"m"^0` for methanoic acid, is 404 S cm2 mol−3.
Which of the following solutions will have the highest conductivity at 298 K?
