Advertisements
Advertisements
प्रश्न
The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below:
| Concentration/M | 0.001 | 0.010 | 0.020 | 0.050 | 0.100 |
| 102 × κ/S m−1 | 1.237 | 11.85 | 23.15 | 55.53 | 106.74 |
Calculate ∧m for all concentrations and draw a plot between ∧m and c1/2. Find the value of `∧_m^0`.
Advertisements
उत्तर
| Concentration (M) | C1/2 (M1/2) |
κ(S m−1) | κ(S cm−1) | `Lambda_m = (kappa xx 1000)/M` (S cm2 mol−1) |
| 0.001 | 0.032 | 1.237 × 10−2 | 1.237 × 10−4 | `Lambda_m = (1.237 xx 10^-4 xx 1000)/0.001` = 123.7 |
| 0.010 | 0.100 | 11.85 × 10−2 | 11.85 × 10−4 | `Lambda_m = (11.85 xx 10^-4 xx 1000)/(0.010)` = 118.5 |
| 0.020 | 0.141 | 23.15 × 10−2 | 23.15 × 10−4 | `Lambda_m = (23.15 xx 10^-4 xx 1000)/0.020` = 115.8 |
| 0.050 | 0.224 | 55.53 × 10−2 | 55.53 × 10−4 | `Lambda_m = (55.53 xx 10^-4 xx 1000)/(0.050)` = 111.1 |
| 0.100 | 0.316 | 106.74 × 10−2 | 106.74 × 10−4 | `Lambda_m = (106.74 xx 10^-4 xx 1000)/(0.100)` = 106.7 |
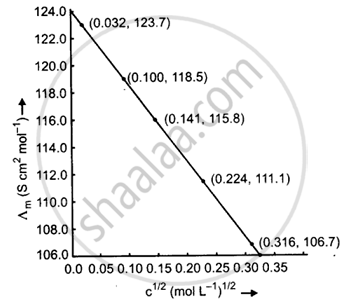
When the straight line is drawn backwards, it meets the ∧m axis at 124.0 S cm2 mol−1, which is the value of `∧_"m"^0`.
APPEARS IN
संबंधित प्रश्न
Resistance of conductivity cell filled with 0.1 M KCl solution is 100 ohms. If the resistance of the same cell when filled with 0.02 M KCl solution is 520 ohms, calculate the conductivity and molar conductivity of 0.02 M KCl solution. [Given: Conductivity of 0.1 M KCl solution is 1.29 S m-1 .]
The conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm−1. Calculate its molar conductivity.
State Kohlrausch Law.
10.0 grams of caustic soda when dissolved in 250 cm3 of water, the resultant gram molarity of solution is _______.
(A) 0.25 M
(B) 0.5 M
(C) 1.0 M
(D) 0.1 M
The conductivity of 0.02M AgNO3 at 25°C is 2.428 x 10-3 Ω-1 cm-1. What is its molar
conductivity?
Define the following terms :
Limiting molar conductivity
The S.I. unit of cell constant for conductivity cell is __________.
A steady current of 2 amperes was passed through two electrolytic cells X and Y connected in series containing electrolytes FeSO4and ZnSO4 until 2.8g of Fe deposited at the cathode of cell X. How long did the current flow? Calculate the mass of Zn deposited at the cathode of cell Y.
(Molar mass: Fe=56g mol-1,Zn=65.3g mol-1,1F=96500C mol-1)
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:

Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes because of the fact that ____________.
Kohlrausch law of independent migration of ions states ____________.
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer.
Match the items of Column I and Column II on the basis of data given below:
`E_("F"_2//"F"^-)^Θ` = 2.87 V, `"E"_(("Li"^(+))//("Li"^-))^Θ` = − 3.5V, `"E"_(("Au"^(3+))//("Au"))^Θ` = 1.4 V, `"E"_(("Br"_(2))//("Br"^-))^Θ` = 1.09 V
| Column I | Column II |
| (i) F2 | (a) metal is the strongest reducing agent |
| (ii) Li | (b) metal ion which is the weakest oxidising agent |
| (iii) Au3+ | (c) non metal which is the best oxidising agent |
| (iv) Br– | (d) unreactive metal |
| (v) Au | (e) anion that can be oxidised by Au3+ |
| (vi) Li+ | (f) anion which is the weakest reducing agent |
| (vii) F– | (g) metal ion which is an oxidising agent |
Assertion: Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason: For weak electrolytes degree of dissociation increases with dilution of solution.
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer. Graphically show the behavior of ‘A’ and ‘B’.
The molar conductivity of 0.007 M acetic acid is 20 S cm2 mol−1. What is the dissociation constant of acetic acid? Choose the correct option.
\[\begin{array}{cc}
\end{array}\]\[\begin{bmatrix}
\ce{\Lambda^{\circ}_{H^+} = 350 S cm^2 mol^{-1}}\\
\ce{\Lambda^{\circ}_{CH_3COO^-} = 50 S cm^2 mol^{-1}}
\end{bmatrix}\]
Given below are two statements:
Statements I: The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH3COOH (weak electrolyte).
Statement II: Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:
Assertion (A): Molar conductivity decreases with increase in concentration.
Reason (R): When concentration approaches zero, the molar conductivity is known as limiting molar conductivity.
Discuss the variation of conductivity and molar conductivity with concentration.
