Advertisements
Advertisements
प्रश्न
Action of soap is due to emulsification and micelle formation. Comment.
Advertisements
उत्तर
Soap is a sodium or potassium salt of a higher fatty acid and may be represented as RCOO–Na+ (e.g., sodium stearate CH3(CH2 )16COO Na+ which is a major component of many bar soaps). When dissolved in water, it dissociates into RCOO– and Na+ ions. The RCOO– ions, however, consist of two parts – a long hydrocarbon chain R (also called the non-polar ‘tail’) which is hydrophobic (water repelling), and a polar group COO– (also called the polar-ionic ‘head’), which is hydrophilic (water loving).
The RCOO– ions are, therefore, present on the surface with their COO– groups in water and the hydrocarbon chains R stay away from it and remain at the surface. But at critical micelle concentration, the anions are pulled into the bulk of the solution and aggregate to form a spherical shape with their hydrocarbon chains pointing towards the center of the sphere with COO– part remaining outward on the surface of the sphere. An aggregate thus formed is known as an ‘ionic micelle’.
The cleansing action of soap is due to the fact that soap molecules form micelles around the oil droplet in such a way that the hydrophobic part of the stearate ions is in the oil droplet and the hydrophilic part projects out of the grease droplet like the bristles. Since the polar groups can interact with water, the oil drop that is surrounded by stearate ions is now pulled into the water and removed from the dirty surface. Thus, soap helps in emulsification and washing away of oils and fats. The negatively charged sheath around the globules prevents them from coming together and forming aggregates.

(a) Grease on cloth
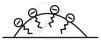
(b) Stearate ions (from soap) arranging around the grease droplets
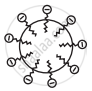
(c) Micelle formed
संबंधित प्रश्न
Give reasons for the following observations: Addition of alum purifies the water
What are emulsions?
What are the different types of emulsions? Give one example of each type.
What are the dispersed phase and dispersion medium in milk?
How do emulsifires stabilise emulsion? Name two emulsifiers.
Give four uses of emulsions.
Give one example each of 'oil in water' and 'water in oil' emulsion.
Answer the following question.
What is the difference between an emulsion and a gel?
Give three uses of emulsions.
Match the types of colloidal systems given in Column I with the name given in Column II.
| Column I | Column II |
| (i) Solid in liquid | (a) Foam |
| (ii) Liquid in solid | (b) Sol |
| (iii) Liquid in liquid | (c) Gel |
| (iv) Gas in liquid | (d) Emulsion |
What is the role of adsorption in froth floatation process used especially for concentration of sulphide ores?
During preparation of ice-cream gelatin is added in ice-creams. What could be the role of gelatin in the process?
