Advertisements
Advertisements
प्रश्न
In the cubic crystal of CsCl (d = 3.97 g/cm3), the eight corners are occupied by Cl− with a Cs+ at the centre and vice-versa. Calculate the distance between the neighbouring Cs+ and Cl− ions. What is the radius ratio of the two ions? (At. masses: Cs = 132.91, Cl = 35.45)
Advertisements
उत्तर
Number of Cl− ions per unit cell = `8 xx 1/8 = 1`
Number of Cs+ ions per unit cell = 1 × 1 = 1
Therefore, the unit cell of caesium chloride contains one Cs+ Cl− unit, i.e., Z = 1
Given that ρ = 3.97 g/cm3,
M = 132.91 + 35.45 = 168.36
∴ `rho = (Z xx M)/(a^3 xx N_A)`
∴ `a = ((Z xx M)/(rho xx N_A))^(1/3)`
= `((1 xx 168.36)/(3.97 xx 6.022 xx 10^23))^(1/3)`
= `((1 xx 168.36)/(23.90 xx 10^23))^(1/3)`
= `(7.044 xx 10^(-23))^(1/3)`
= 4.13 × 10−8 cm
Since the crystal lattice is body-centred,
body diagonal AD = `sqrt 3 a`
= `sqrt 3 xx 4.13`
= `1.73 xx 4.13`
= 7.15 Å
Also, AD = 2r+ + 2r− = 7.15
or `r_+ + r_- = 7.15/2`
= 3.57 Å
Hence, the distance between the neighbouring Cs+ and Cl− ions is 3.57 Å.
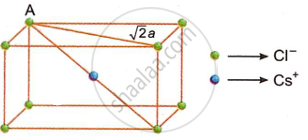
If two Cl− ion touch each other,
2r− = a
= 4.13 Å
or, `r_- = 4.13/2 = 2.06` Å
r+ = 3.57 − 2.06
= 1.51 Å
Hence, the radius ratio
`r_+/r_- = 1.51/2.05`
= 0.73 Å
