Advertisements
Advertisements
प्रश्न
What is Schottky defect in a solid?
What is Schottky defect?
दीर्घउत्तर
Advertisements
उत्तर
This defect arises when some of the lattice points in a crystal are unoccupied. The unoccupied lattice points are called lattice vacancies or holes. The Schottky defect is common in ionic crystals. The vacancy at a cation site in ionic solids is always followed by a vacancy at a nearby anion site. Consequently, a Schottky defect is always characterised by the absence of an equal number of cations and anions (Schottky pairs) in an ionic solid. This ensures that the crystal remains electrically neutral, despite the absence of ions. Figure illustrates the Schottky defect.
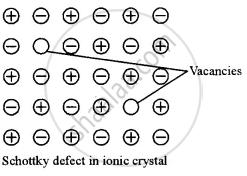
shaalaa.com
States of Matter - Point Defects – F Centers
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
