Advertisements
Advertisements
प्रश्न
If the radius of the octahedral void is r and the radius of the atoms in close packing is R, derive the relation between r and R.
The radius of an octahedral void is r and the radius of the atom is R when these are in close-packing. Derive a relationship between the two.
Advertisements
उत्तर
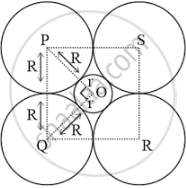
A sphere with center O is fitted into the octahedral void as shown in the above figure. It can be observed from the figure that ΔPOQ is right-angled
∠POQ = 90°
Now, applying the Pythagorean theorem, we can write:
PQ2 = PO2 + OQ2
⇒ (2R)2 = (R + r)2 + (R + r)2
⇒ (2R)2 = 2(R + r)2
⇒ 2R2 = (R + r)2
⇒ `sqrt 2R = R + r`
⇒ `r = sqrt 2R - R`
⇒ `r = (sqrt 2 - 1)R`
⇒ `r = (1.414 - 1)R`
⇒ r = 0.414 R
संबंधित प्रश्न
What is the formula of a compound in which the element Y forms hcp lattice and atoms of X occupy 2/3rd of tetrahedral voids?
How will you distinguish between the following pair of terms?
Tetrahedral and octahedral voids
How many tetrahedral voids can exist per unit cell in a hexagonal close packing sphere?
The unit cell of a substance has cations A+ at the corners of the unit cell and the anions B− at the center. The simplest formula of the substance is ____________.
Total number of voids in 0.5 mole of a compound which forms hexagonal close packed structure is ____________.
What is the coordination number of sodium in Na2O?
The Ca2+ and F– are located in CaF2 crystal, respectively at face centered cubic lattice points and in ____________.
Which set of following characteristics for ZnS crystal is correct?
Percentages of free space in cubic close packed structure and in the body-centered packed structure are respectively:
The number of octahedral voids present in a lattice is A. The number of closed packed articles, the number of tetrahedral voids generated is B the number of closed packed particles:
In which of the following arrangements octahedral voids are formed?
(i) hcp
(ii) bcc
(iii) simple cubic
(iv) fcc
The number of tetrahedral voids per unit cell in NaCl crystal is:
(i) 4
(ii) 8
(iii) twice the number of octahedral voids.
(iv) four times the number of octahedral voids.
Show that in a cubic close packed structure, eight tetrahedral voids are present per unit cell.
A solid compound XY has Nacl structure. If the radium of cation (X+) is 100 pm, the radium of anion (r–) will be:-
In which of the following structures coordination number for cations and anions in the packed structure will be same?
