Advertisements
Advertisements
प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Advertisements
उत्तर
Oxidation state = +2
Coordination number = 4
3d7 configuration = \[\ce{t^3_{2g}e^4_g}\]
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
IUPAC name of the following compound is
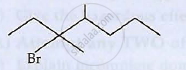
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write structures of compounds A, B and C in of the following reactions

Write structures of compounds A, B and C in of the following reactions

What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Which of the following is non-ionizable?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
In which of the following compounds, the oxidation number of iodine is fractional?
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
Which one of the following ligands forms a chelate?
According to IUPAC nomenclatures, sodium nitroprusside is named as
Which of the following ligands can exhibit linkage isomerism?
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Statement I: [Co(NH3)6]3+ is a homoleptic compleх whereas [Co(NH3)4Cl2]+ is a heteroleptic complex.
Statement II: Complex [Co(NH3)6]3+ has only one kind of ligands but [Co(NH3)4Cl2]+ has more than one kind of ligands.
In the light of the above statements, Choose the correct answer form the options given below.
