Advertisements
Advertisements
प्रश्न
Write structures of compounds A, B and C in of the following reactions

Advertisements
उत्तर
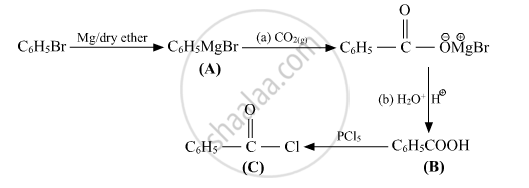
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the structures of compounds A, B and C in the following reactions:

A group of atoms can function as a ligand only when:
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
In which of the following compounds the oxidation state of the nickel atom is 0?
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
Oxidation number of cobalt in K[Co(CO)4] is
Which of the following ligands can exhibit linkage isomerism?
What are Homoleptic complexes?
Explain the following, giving two examples:
Coordination polyhedron
