Advertisements
Advertisements
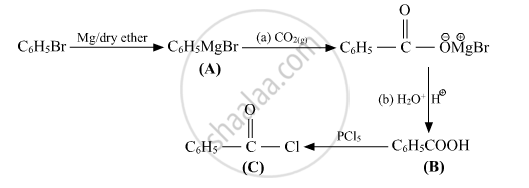
प्रश्न
Write structures of compounds A, B and C in of the following reactions

Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Explain the following, giving two examples:
Coordination entity
How is Benzonitrile converted to Benzophenone?
Write applications of co-ordination compounds in medicine and electroplating.
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the IUPAC name of the following coordination compound:
[Cr(NH3)4(H2O)2]Cl3
Write structures of compounds A and B of the following reaction :

In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
What is meant by a didentate ligand?
