Advertisements
Advertisements
प्रश्न
Write structures of compounds A and B of the following reaction :

Advertisements
उत्तर
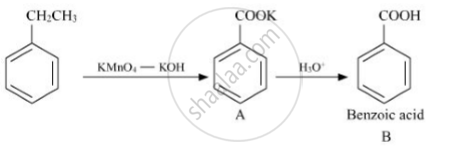
APPEARS IN
संबंधित प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
IUPAC name of the following compound is
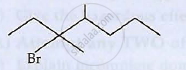
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Complete the following reactions
NH3+3Cl2(excess) ---->
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Oxidation number of cobalt in K[Co(CO)4] is
Which one of the following ligands forms a chelate?
What are Heteroleptic complexes?
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
Statement I: [Co(NH3)6]3+ is a homoleptic compleх whereas [Co(NH3)4Cl2]+ is a heteroleptic complex.
Statement II: Complex [Co(NH3)6]3+ has only one kind of ligands but [Co(NH3)4Cl2]+ has more than one kind of ligands.
In the light of the above statements, Choose the correct answer form the options given below.
