Advertisements
Advertisements
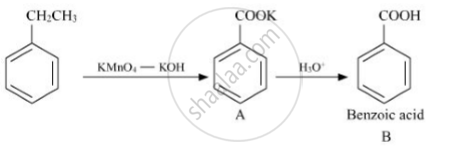
Question
Write structures of compounds A and B of the following reaction :

Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
cis-[CrCl2(en)2]Cl
How is Benzonitrile converted to Benzophenone?
What happens when PCl5 is heated? Write the equations involved.
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
The most stable ion is ______.
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
Explain the following, giving two examples:
Coordination polyhedron
Give two examples of unidentate ligands.
