Advertisements
Advertisements
Question
Give two examples of unidentate ligands.
Advertisements
Solution
- Chloride ion (Cl−)
- Ammonia (NH3)
APPEARS IN
RELATED QUESTIONS
Explain the following, giving two examples:
Coordination entity
What is meant by unidentate ligand?
IUPAC name of the following compound is
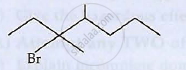
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write structures of compounds A, B and C in of the following reactions

Write structures of compounds A and B of the following reaction :

Ligand (en) is an example of ___________.
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
Match the compounds given in Column I with the oxidation state of cobalt present in it (given in Column II) and assign the correct code:
| Column I (Compound) | Column II (Oxidation state of Co) |
| A. \[\ce{[Co(NCS)(NH3)5](SO3)}\] | 1. + 4 |
| B. \[\ce{[Co(NH3)4 CL2]SO4}\] | 2. 0 |
| C. \[\ce{Na4[Co(S2O3)3]}\] | 3. + 1 |
| D. \[\ce{[Co2(CO)8]}\] | 4. + 2 |
| 5. + 3 |
Which one of the following does not achieve an octet of electrons in the central atom?
Oxidation number of carbon in CH2Cl2 is
In which of the following compounds, the oxidation number of iodine is fractional?
Which one of the following ligands forms a chelate?
Why chelate complexes are more stable than complexes with unidentate ligands?
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
What is meant by the chelate effect? Give an example.
Give two examples of ambidentate ligands.
Homoleptic complex from the following complex is ____.
