Advertisements
Advertisements
प्रश्न
Give two examples of unidentate ligands.
Advertisements
उत्तर
- Chloride ion (Cl−)
- Ammonia (NH3)
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
cis-[CrCl2(en)2]Cl
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Write IUPAC name of the following Complex [Cr(NH3)3Cl3]
Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Write the structures of compounds A, B and C in the following reactions
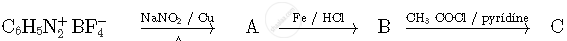
Write the IUPAC name of the following coordination compound:
[PtCl2(NH3)4][PtCl4]
Which of the following represents a chelate ligand?
Which of the following is non-ionizable?
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
The nature of hybridisation in the ammonia molecule is
Oxidation number of carbon in CH2Cl2 is
In which of the following compounds, the oxidation number of iodine is fractional?
The oxidation number d-arbitral occupation and co-ordination number of Cr in the complex cis [Cr(en)2Cl2]Cl are respectively.
What are Heteroleptic complexes?
The one that will show optical activity is: (en = ethane 1, 2-diamine)
What is meant by a didentate ligand?
