Advertisements
Advertisements
प्रश्न
Write the structures of compounds A, B and C in the following reactions
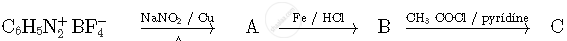
Advertisements
उत्तर
A: C6H5NO2 (Nitrobenzene)
B: C6H5NH2 (Aniline)
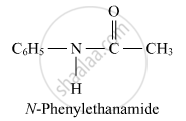
C:
APPEARS IN
संबंधित प्रश्न
Amongst the following, the most stable complex is:
Write IUPAC names of the following compounds:

In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
What are Homoleptic complexes?
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
What is meant by the chelate effect? Give an example.
Explain the ligand.
