Advertisements
Advertisements
प्रश्न
Write the structures of compounds A, B and C in the following reactions
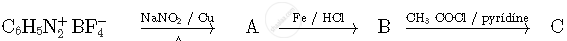
Advertisements
उत्तर
A: C6H5NO2 (Nitrobenzene)
B: C6H5NH2 (Aniline)
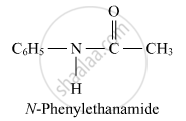
C:
APPEARS IN
संबंधित प्रश्न
What happens when PCl5 is heated? Write the equations involved.
Write structures of compounds A, B and C in of the following reactions

Complete the following reactions
NH3+3Cl2(excess) ---->
Write structures of compounds A and B of the following reaction :

The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
Which of the following ligands can exhibit linkage isomerism?
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
What is a chelate complex?
Which of the following species cannot act as a ligand? Give reason.
Give two examples of didentate ligands.
