Advertisements
Advertisements
प्रश्न
Which of the following species cannot act as a ligand? Give reason.
पर्याय
OH−
`"NH"_4^+`
CH3NH2
H2O
Advertisements
उत्तर
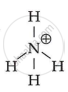
`bb("NH"_4^+)`
Explanation:
Because it is unable of providing a core metal ion with a lone pair of electrons.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Amongst the following, the most stable complex is:
What happens when PCl5 is heated? Write the equations involved.
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
A group of atoms can function as a ligand only when:
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
What are Heteroleptic complexes?
What is a chelate complex?
Give two examples of didentate ligands.
