Advertisements
Advertisements
प्रश्न
Give two examples of didentate ligands.
Advertisements
उत्तर
- Ethane-1, 2-diamine (H2NCH2CH2NH2)
- oxalate \[\ce{(C2O^{2-}_4)}\]
APPEARS IN
संबंधित प्रश्न
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
IUPAC name of the following compound is
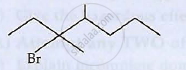
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write the IUPAC name of the following coordination compound:
[PtCl2(NH3)4][PtCl4]
Which of the following represents a chelate ligand?
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
The nature of hybridisation in the ammonia molecule is
In which of the following compounds, the oxidation number of iodine is fractional?
Which one of the following ligands forms a chelate?
According to IUPAC nomenclatures, sodium nitroprusside is named as
Why chelate complexes are more stable than complexes with unidentate ligands?
The following ligand is:

What is a chelate complex?
Which of the following species cannot act as a ligand? Give reason.
Statement I: [Co(NH3)6]3+ is a homoleptic compleх whereas [Co(NH3)4Cl2]+ is a heteroleptic complex.
Statement II: Complex [Co(NH3)6]3+ has only one kind of ligands but [Co(NH3)4Cl2]+ has more than one kind of ligands.
In the light of the above statements, Choose the correct answer form the options given below.
