Advertisements
Advertisements
Question
Which of the following species cannot act as a ligand? Give reason.
Options
OH−
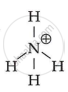
`"NH"_4^+`
CH3NH2
H2O
Advertisements
Solution
`bb("NH"_4^+)`
Explanation:
Because it is unable of providing a core metal ion with a lone pair of electrons.
APPEARS IN
RELATED QUESTIONS
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
cis-[CrCl2(en)2]Cl
A group of atoms can function as a ligand only when:
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
In which of the following compounds, the oxidation number of iodine is fractional?
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
Explain the following, giving two examples:
Coordination polyhedron
