Advertisements
Advertisements
Question
Write the structures of compounds A, B and C in the following reactions
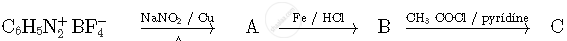
Advertisements
Solution
A: C6H5NO2 (Nitrobenzene)
B: C6H5NH2 (Aniline)
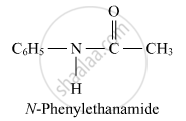
C:
APPEARS IN
RELATED QUESTIONS
Write structures of compounds A, B and C in of the following reactions

Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Write the structures of compounds A, B and C in the following reactions:

In which of the following compounds the oxidation state of the nickel atom is 0?
Which of the following is an ionic ligand?
Which one of the following ligands forms a chelate?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
What is a chelate complex?
What is meant by the chelate effect? Give an example.
What is meant by ambidentate ligand?
