Advertisements
Advertisements
Question
Give reasons Acetylation of aniline reduces its activation effect.
Advertisements
Solution
The lone pair of nitrogen will get involved in resonance with the carbonyl group. Hence it will reduce the activity of benzene ring in aniline. The resonance involved is shown below:
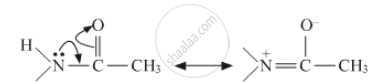
APPEARS IN
RELATED QUESTIONS
Describe the following:
Cross aldol condensation
Complete the synthesis by giving missing starting material, reagent or product.
\[\begin{array}{cc}
\ce{C6H5CHO}\phantom{............}\\
\phantom{........}\ce{+\phantom{......}\ce{->[dil.NaOH][\Delta]}}\phantom{...}\\
\ce{CH3CH2CHO}\phantom{............}
\end{array}\]
What is substituted imine called?
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Why is there a large difference in the boiling points of butanal and butan-1-ol?
When liquid ‘A’ is treated with a freshly prepared ammoniacal silver nitrate solution, it gives bright silver mirror. The liquid forms a white crystalline solid on treatment with sodium hydrogensulphite. Liquid ‘B’ also forms a white crystalline solid with sodium hydrogensulphite but it does not give test with ammoniacal silver nitrate. Which of the two liquids is aldehyde? Write the chemical equations of these reactions also.
Cross aldol condensation occurs between
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
Write a note on the aldol condensation reaction of acetaldehyde.
Which one of the following undergoes reaction with 50% sodiumhydroxide solution to give the corresponding alcohol and acid:
