Advertisements
Advertisements
प्रश्न
Give reasons Acetylation of aniline reduces its activation effect.
Advertisements
उत्तर
The lone pair of nitrogen will get involved in resonance with the carbonyl group. Hence it will reduce the activity of benzene ring in aniline. The resonance involved is shown below:
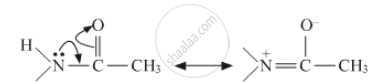
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
Complete the synthesis by giving missing starting material, reagent or product.
\[\begin{array}{cc}
\ce{C6H5CHO}\phantom{............}\\
\phantom{........}\ce{+\phantom{......}\ce{->[dil.NaOH][\Delta]}}\phantom{...}\\
\ce{CH3CH2CHO}\phantom{............}
\end{array}\]
Write chemical equations of the following reaction :
Propanone is treated with dilute Ba (OH)2-.
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
Explain aldol condensation reaction in detail.
Cannizaro’s reaction is not given by ______.
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
What is aldol condensation? Explain it with suitable examples.
