Advertisements
Advertisements
प्रश्न
Cannizaro’s reaction is not given by ______.
विकल्प
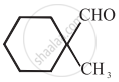
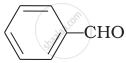
\[\ce{HCHO}\]
\[\ce{CH3CHO}\]
Advertisements
उत्तर
Cannizaro’s reaction is not given by \[\ce{CH3CHO}\].
Explanation:
\[\ce{CH3CHO}\] will not give Cannizzaro’s reaction because it contains a-hydrogen while other three compounds have no a-hydrogen. Hence, they will give Cannizzaro’s reaction.
APPEARS IN
संबंधित प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Aldol
How will you convert ethanal into the following compound?
Butane-1, 3-diol
Describe the following:
Cross aldol condensation
Give reasons Acetylation of aniline reduces its activation effect.
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write chemical equations of the following reaction :
Propanone is treated with dilute Ba (OH)2-.
Explain aldol condensation reaction in detail.
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Compound ‘A’ was prepared by oxidation of compound ‘B’ with alkaline \[\ce{KMnO4}\]. Compound ‘A’ on reduction with lithium aluminium hydride gets converted back to compound ‘B’. When compound ‘A’ is heated with compound B in the presence of \[\ce{H2SO4}\] it produces fruity smell of compound C to which family the compounds ‘A’, ‘B’ and ‘C’ belong to?
When liquid ‘A’ is treated with a freshly prepared ammoniacal silver nitrate solution, it gives bright silver mirror. The liquid forms a white crystalline solid on treatment with sodium hydrogensulphite. Liquid ‘B’ also forms a white crystalline solid with sodium hydrogensulphite but it does not give test with ammoniacal silver nitrate. Which of the two liquids is aldehyde? Write the chemical equations of these reactions also.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Cross aldol condensation occurs between
Which of the following gives aldol con~ensation reaction?
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
The major product of the following reaction is:

Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.

Identify A and B:
Write a note on the aldol condensation reaction of acetaldehyde.
