Advertisements
Advertisements
प्रश्न
Cannizaro’s reaction is not given by ______.
पर्याय
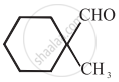
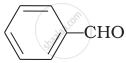
\[\ce{HCHO}\]
\[\ce{CH3CHO}\]
Advertisements
उत्तर
Cannizaro’s reaction is not given by \[\ce{CH3CHO}\].
Explanation:
\[\ce{CH3CHO}\] will not give Cannizzaro’s reaction because it contains a-hydrogen while other three compounds have no a-hydrogen. Hence, they will give Cannizzaro’s reaction.
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction and which neither? Write the structures of the expected products of aldol condensation and Cannizzaro reaction.
- Methanal
- 2-Methylpentanal
- Benzaldehyde
- Benzophenone
- Cyclohexanone
- 1-Phenylpropanone
- Phenylacetaldehyde
- Butan-1-ol
- 2, 2-Dimethylbutanal
How will you convert ethanal into the following compound?
Butane-1, 3-diol
How will you convert ethanal into the following compound?
But-2-enal
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Describe the following:
Cross aldol condensation
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
What is substituted imine called?
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Why are carboxylic acids more acidic than alcohols or phenols although all of them have hydrogen atom attached to a oxygen atom \[\ce{(-O-H)}\]?
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
Convert the following:
Acetaldehyde to But-2-enal
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
Explain Aldol condensation of ethanal.
What is aldol condensation? Explain it with suitable examples.
Which one of the following undergoes reaction with 50% sodiumhydroxide solution to give the corresponding alcohol and acid:
