Advertisements
Advertisements
प्रश्न
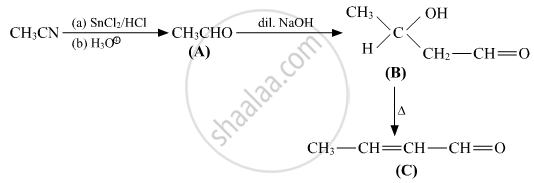
Write structures of compounds A, B and C in of the following reactions

Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
IUPAC name of the following compound is
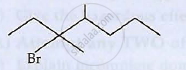
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
How is Benzonitrile converted to Benzophenone?
The coordination number of the central ion may be obtained from:
The nature of hybridisation in the ammonia molecule is
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Glycinato ligand is ______.
What is a chelate complex?
What is meant by the chelate effect? Give an example.
Explain the following, giving two examples:
Homoleptic
