Advertisements
Advertisements
प्रश्न
Amongst the following, the most stable complex is:
विकल्प
[Fe(H2O)6]3+
[Fe(NH3)6]3+
[Fe(C2O4)3]3−
[FeCl6]3−
Advertisements
उत्तर
[Fe(C2O4)3]3−
Explanation:
[Fe(C2O4)3]3− is the most stable complex because oxalate ion \[\ce{(C2O^{2-}_4)}\] is a bidentate ligand. Bidentate ligands form chelate rings with the metal ion, which increases the stability of the complex due to the chelate effect. The other ligands (H2O, NH3, Cl−) are monodentate ligands and form comparatively less stable complexes.
APPEARS IN
संबंधित प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
IUPAC name of the following compound is
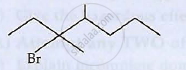
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Write the structures of compounds A, B and C in the following reactions:

Write structures of compounds A and B of the following reaction :

Which of the following represents a chelate ligand?
A group of atoms can function as a ligand only when:
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
In which of the following compounds, the oxidation number of iodine is fractional?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
What are Homoleptic complexes?
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
What is a chelate complex?
Explain the following, giving two examples:
Heteroleptic
