Advertisements
Advertisements
प्रश्न
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Advertisements
उत्तर
Oxidation state = +3
Coordination number = 6
3d6 configuration = \[\ce{t^6_{2g}e^0_g}\]
APPEARS IN
संबंधित प्रश्न
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
IUPAC name of the following compound is
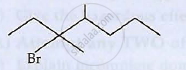
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
What happens when PCl5 is heated? Write the equations involved.
Write the structures of compounds A, B and C in the following reactions
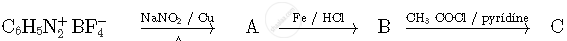
Which of the following represents a chelate ligand?
The coordination number of the central ion may be obtained from:
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Which one of the following ligands forms a chelate?
The most stable ion is ______.
Metal attached with EDTA in an octahedral complex, has coordination number ______.
Ethylene diamine tetraacetate (EDTA) ion is ______.
The following ligand is:

Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
Assertion (A): EDTA is a hexadentate ligand.
Reason (R): EDTA has 2 nitrogen and 4 oxygen donor atoms.
What is meant by the chelate effect? Give an example.
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
Give two examples of unidentate ligands.
Give two examples of didentate ligands.
