Advertisements
Advertisements
प्रश्न
Write structures of compounds A and B of the following reaction :

आकृति
Advertisements
उत्तर
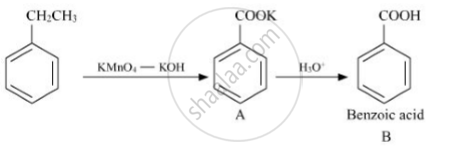
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Write the structures of compounds A, B and C in the following reactions:

In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
In which of the following compounds, the oxidation number of iodine is fractional?
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
The following ligand is:

The one that will show optical activity is: (en = ethane 1, 2-diamine)
What is meant by a didentate ligand?
What is meant by ambidentate ligand?
Homoleptic complex from the following complex is ____.
