Advertisements
Advertisements
प्रश्न
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
Advertisements
उत्तर
When white light falls on the complex, some part of it is absorbed. Higher the crystal field splitting, lower will be the wavelength absorbed by the complex. The observed colour of complex is the colour generated from the wavelength left over.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
[Mn(H2O)6]SO4
Write the structures of compounds A, B and C in the following reactions
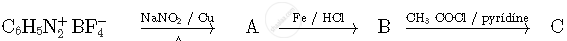
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
Oxidation number of cobalt in K[Co(CO)4] is
Oxidation number of carbon in CH2Cl2 is
Which one of the following ligands forms a chelate?
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
What are Homoleptic complexes?
Why chelate complexes are more stable than complexes with unidentate ligands?
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
Which of the following species cannot act as a ligand? Give reason.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
What is meant by a didentate ligand?
