Advertisements
Advertisements
Question
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
Advertisements
Solution
No, ionization isomers are possible by exchange of ligand with counter ion only and not by exchange of central metal ion.
APPEARS IN
RELATED QUESTIONS
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Write IUPAC names of the following compounds:

Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Write the structures of compounds A, B and C in the following reactions
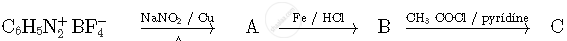
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
Oxidation number of carbon in CH2Cl2 is
Which of the following ligands can exhibit linkage isomerism?
Is the central metal atom in coordination complexes a Lewis acid or a Lewis base? Explain.
Explain the following, giving two examples:
Coordination polyhedron
Homoleptic complex from the following complex is ____.
