Advertisements
Advertisements
Questions
How is Benzonitrile converted to Benzophenone?
How is benzophenone prepared from benzonitrile?
Advertisements
Solution
Benzonitrile reacts with phenyl magnesium bromide in equimolecular proportion in the presence of dry ether to give an adduct, which on acid hydrolysis gives benzophenone

APPEARS IN
RELATED QUESTIONS
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Amongst the following, the most stable complex is:
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
IUPAC name of the following compound is
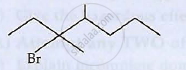
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write IUPAC names of the following compounds:

Write structures of compounds A, B and C in of the following reactions

Write the IUPAC name of the following complex:
[Co(NH3)5(CO3)]Cl
Complete the following reactions
NH3+3Cl2(excess) ---->
A group of atoms can function as a ligand only when:
Which of the following is non-ionizable?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
When 0.1 mol \[\ce{CoCl3 (NH3)5}\] is treated with excess of \[\ce{AgNO3}\], 0.2 mol of \[\ce{AgCl}\] are obtained. The conductivity of solution will correspond to ______.
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
A complex of the type \[\ce{[M(AA)2X2]^{n+}}\] is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
The nature of hybridisation in the ammonia molecule is
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
The most stable ion is ______.
Glycinato ligand is ______.
What is a chelate complex?
Explain the ligand.
Explain the following, giving two examples:
Coordination polyhedron
What is meant by a didentate ligand?
What is meant by ambidentate ligand?
