Advertisements
Advertisements
Question
Complete the following reactions
NH3+3Cl2(excess) ---->
Advertisements
Solution
Complete chemical reactions are:
NH3 + 3 Cl2 (excess) → NCl3 + 3 HCl
APPEARS IN
RELATED QUESTIONS
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Write the structures of compounds A, B and C in the following reactions
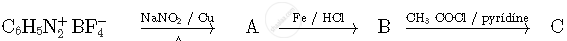
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
The correct \[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2Cl2]}\] is ______.
Which of the following complexes are heteroleptic?
(i) \[\ce{[Cr(NH3)6]^{3+}}\]
(ii) \[\ce{[Fe(NH3)4]Cl2]^+}\]
(iii) \[\ce{[Mn(CN)6]^{4-}}\]
(iv) \[\ce{[Co(NH3)4]Cl2]}\]
Ethylene diamine tetraacetate (EDTA) ion is ______.
In the complex ion \[\ce{[CoCl(en)2ONO]+}\], the coordination number and the oxidation number of the central metal ion are ______ and ______.
Explain the following, giving two examples:
Coordination polyhedron
