Advertisements
Advertisements
Question
What happens when (NH4)2Cr2O7 is heated? Write the equations.
Advertisements
Solution
When (NH4)2Cr2O7 is heated its decompose to produces Cr2O3, H2O and N2
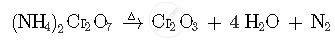
APPEARS IN
RELATED QUESTIONS
Describe the laboratory method of preparation of ammonia
What is the action of Excess of air on ammonia ?
Mention the conditions required to maximise the yield of ammonia.
How does ammonia react with a solution of Cu2+?
What is the action of the following reagents on ammonia :
Sodium metal
Ammonia has a higher boiling point and is less volatile because of ____________.
Which compound is used as the cooling liquid in refrigerators?
Ammonia act as a Lewis base because nitrogen has ____________.
Which one of the following is not a use of ammonia?
What happens when reactions:
Benzylchloride is treated with ammonia followed by the reaction with Chloromethane.
