Advertisements
Advertisements
प्रश्न
What happens when (NH4)2Cr2O7 is heated? Write the equations.
Advertisements
उत्तर
When (NH4)2Cr2O7 is heated its decompose to produces Cr2O3, H2O and N2
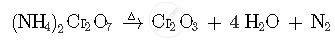
APPEARS IN
संबंधित प्रश्न
What is the action of Excess of air on ammonia ?
How is ammonia manufactured industrially?
In laboratory ammonia is prepared by heating:
Ammonia has a higher boiling point and is less volatile because of ____________.
Ammonia act as a Lewis base because nitrogen has ____________.
On heating ammonium dichromate and barium azide separately we get ______.
In Haber’s process for the manufacture of NH3:
The shape of ammonia molecule is ____________.
In the preparation of HNO3, we get NO gas by catalytic oxidation of ammonia. The moles of NO produced by the oxidation of two moles of NH3 will be ______.
What happens when reactions:
Benzylchloride is treated with ammonia followed by the reaction with Chloromethane.
