Advertisements
Advertisements
प्रश्न
What happens when (NH4)2Cr2O7 is heated? Write the equations.
Advertisements
उत्तर
When (NH4)2Cr2O7 is heated its decompose to produces Cr2O3, H2O and N2
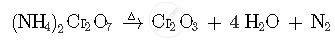
APPEARS IN
संबंधित प्रश्न
What is the action of the following reagents on ammonia :
Nessler's reagent
Mention the conditions required to maximise the yield of ammonia.
How does ammonia react with a solution of Cu2+?
What is the action of the following reagents on ammonia :
Sodium metal
What is Nessler’s reagent?
The shape of ammonia molecule is ____________.
Ammonia is generally manufactured for fertilizers by the reaction:
In qualitative analysis when \[\ce{H2S}\] is passed through an aqueous solution of salt acidified with dil. \[\ce{HCl}\], a black precipitate is obtained. On boiling the precipitate with dil. \[\ce{HNO3}\], it forms a solution of blue colour. Addition of excess of aqueous solution of ammonia to this solution gives ______.
In the preparation of HNO3, we get NO gas by catalytic oxidation of ammonia. The moles of NO produced by the oxidation of two moles of NH3 will be ______.
\[\ce{PCl5}\] reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous \[\ce{NH3}\] solution. Write the reactions involved to explain what happens.
