Advertisements
Advertisements
प्रश्न
How is ammonia manufactured industrially?
Advertisements
उत्तर
Perhaps the most significant nitrogen component is ammonia, which is employed as a precursor to many other more beneficial chemicals.
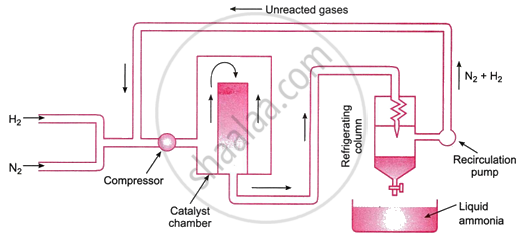
Ammonia is generally manufactured by Haber’s process. The process involves the following reaction.
\[\ce{N2 + 3H2 <=> 2NH3}\]; ΔH = −93.6 kJ mol−1
The reaction proceeds with a reduction in volume and is reversible and exothermic. The following are the ideal conditions for the highest ammonia yield, according to Le Chatelier's principle:
- Low temperature: Since the process is exothermic, a low temperature encourages the reaction to go forward, increasing the amount of ammonia produced. Experimental research has determined that the ideal temperature ranges between 720 and 770 K.
- High pressure: At high pressures, a greater yield of ammonia can be achieved since the reaction continues with a reduction in volume. The ideal pressure range is between 200 and 900 atmospheres.
- Catalyst: A suitable catalyst must also be utilised to boost the reaction's pace. Finely split iron with molybdenum as a promoter is a commonly used catalyst. These days, iron oxide combined with trace amounts of K2O and Al2O3 is also employed as a catalyst.
संबंधित प्रश्न
What is the action of Excess of air on ammonia ?
What is the action of the following reagents on ammonia :
Nessler's reagent
Mention the conditions required to maximise the yield of ammonia.
How does ammonia react with a solution of Cu2+?
What happens when (NH4)2Cr2O7 is heated? Write the equations.
What is the action of Na Metal on ammonia?
What is the action of the following reagents on ammonia :
Sodium metal
What is Nessler’s reagent?
Ammonia has a higher boiling point and is less volatile because of ____________.
Which compound is used in the preparation of caprolactam?
Which compound is used as the cooling liquid in refrigerators?
The shape of ammonia molecule is ____________.
When ammonia is heated with cupric oxide, a molecule of ammonia will ____________.
Liquid ammonia bottles are opened after cooling them in ice for some time. It is because liquid NH3 ____________.
Which one of the following is not a use of ammonia?
A brown ring is formed in the ring test for \[\ce{NO3^{-}}\] ion. It is due to the formation of ______.
What happens when reactions:
Benzylchloride is treated with ammonia followed by the reaction with Chloromethane.
