Advertisements
Advertisements
Question
How is ammonia manufactured industrially?
Advertisements
Solution
Perhaps the most significant nitrogen component is ammonia, which is employed as a precursor to many other more beneficial chemicals.
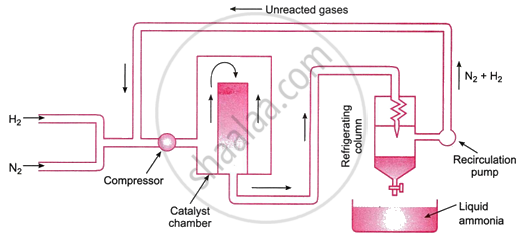
Ammonia is generally manufactured by Haber’s process. The process involves the following reaction.
\[\ce{N2 + 3H2 <=> 2NH3}\]; ΔH = −93.6 kJ mol−1
The reaction proceeds with a reduction in volume and is reversible and exothermic. The following are the ideal conditions for the highest ammonia yield, according to Le Chatelier's principle:
- Low temperature: Since the process is exothermic, a low temperature encourages the reaction to go forward, increasing the amount of ammonia produced. Experimental research has determined that the ideal temperature ranges between 720 and 770 K.
- High pressure: At high pressures, a greater yield of ammonia can be achieved since the reaction continues with a reduction in volume. The ideal pressure range is between 200 and 900 atmospheres.
- Catalyst: A suitable catalyst must also be utilised to boost the reaction's pace. Finely split iron with molybdenum as a promoter is a commonly used catalyst. These days, iron oxide combined with trace amounts of K2O and Al2O3 is also employed as a catalyst.
RELATED QUESTIONS
Describe the laboratory method of preparation of ammonia
What is the action of Excess of air on ammonia ?
What is the action of the following reagents on ammonia :
Nessler's reagent
What happens when (NH4)2Cr2O7 is heated? Write the equations.
What is the action of Excess of chlorine on ammonia?
What is the action of Na Metal on ammonia?
What is Nessler’s reagent?
In laboratory ammonia is prepared by heating:
Ammonia has a higher boiling point and is less volatile because of ____________.
Which compound is used in the preparation of caprolactam?
Ammonia act as a Lewis base because nitrogen has ____________.
Ammonia on reaction with hypochlorite anion can form:
Ammonia is generally manufactured for fertilizers by the reaction:
Which one of the following is not a use of ammonia?
In qualitative analysis when \[\ce{H2S}\] is passed through an aqueous solution of salt acidified with dil. \[\ce{HCl}\], a black precipitate is obtained. On boiling the precipitate with dil. \[\ce{HNO3}\], it forms a solution of blue colour. Addition of excess of aqueous solution of ammonia to this solution gives ______.
A brown ring is formed in the ring test for \[\ce{NO3^{-}}\] ion. It is due to the formation of ______.
\[\ce{PCl5}\] reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous \[\ce{NH3}\] solution. Write the reactions involved to explain what happens.
